Hereditary Eosinophil Peroxidase Deficiency
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

MPO) in Inflammatory Communication
antioxidants Review The Enzymatic and Non-Enzymatic Function of Myeloperoxidase (MPO) in Inflammatory Communication Yulia Kargapolova * , Simon Geißen, Ruiyuan Zheng, Stephan Baldus, Holger Winkels * and Matti Adam Department III of Internal Medicine, Heart Center, Faculty of Medicine and University Hospital of Cologne, 50937 North Rhine-Westphalia, Germany; [email protected] (S.G.); [email protected] (R.Z.); [email protected] (S.B.); [email protected] (M.A.) * Correspondence: [email protected] (Y.K.); [email protected] (H.W.) Abstract: Myeloperoxidase is a signature enzyme of polymorphonuclear neutrophils in mice and humans. Being a component of circulating white blood cells, myeloperoxidase plays multiple roles in various organs and tissues and facilitates their crosstalk. Here, we describe the current knowledge on the tissue- and lineage-specific expression of myeloperoxidase, its well-studied enzymatic activity and incoherently understood non-enzymatic role in various cell types and tissues. Further, we elaborate on Myeloperoxidase (MPO) in the complex context of cardiovascular disease, innate and autoimmune response, development and progression of cancer and neurodegenerative diseases. Keywords: myeloperoxidase; oxidative burst; NETs; cellular internalization; immune response; cancer; neurodegeneration Citation: Kargapolova, Y.; Geißen, S.; Zheng, R.; Baldus, S.; Winkels, H.; Adam, M. The Enzymatic and Non-Enzymatic Function of 1. Introduction. MPO Conservation Across Species, Maturation in Myeloid Progenitors, Myeloperoxidase (MPO) in and its Role in Immune Responses Inflammatory Communication. Myeloperoxidase (MPO) is a lysosomal protein and part of the organism’s host-defense Antioxidants 2021, 10, 562. https:// system. MPOs’ pivotal function is considered to be its enzymatic activity in response to doi.org/10.3390/antiox10040562 invading pathogenic agents. -

Eosinophil-Derived Neurotoxin (EDN/Rnase 2) and the Mouse Eosinophil-Associated Rnases (Mears): Expanding Roles in Promoting Host Defense
Int. J. Mol. Sci. 2015, 16, 15442-15455; doi:10.3390/ijms160715442 OPEN ACCESS International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms Review Eosinophil-Derived Neurotoxin (EDN/RNase 2) and the Mouse Eosinophil-Associated RNases (mEars): Expanding Roles in Promoting Host Defense Helene F. Rosenberg Inflammation Immunobiology Section, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD 20892, USA; E-Mail: [email protected]; Tel.: +1-301-402-1545; Fax: +1-301-480-8384 Academic Editor: Ester Boix Received: 18 May 2015 / Accepted: 30 June 2015 / Published: 8 July 2015 Abstract: The eosinophil-derived neurotoxin (EDN/RNase2) and its divergent orthologs, the mouse eosinophil-associated RNases (mEars), are prominent secretory proteins of eosinophilic leukocytes and are all members of the larger family of RNase A-type ribonucleases. While EDN has broad antiviral activity, targeting RNA viruses via mechanisms that may require enzymatic activity, more recent studies have elucidated how these RNases may generate host defense via roles in promoting leukocyte activation, maturation, and chemotaxis. This review provides an update on recent discoveries, and highlights the versatility of this family in promoting innate immunity. Keywords: inflammation; leukocyte; evolution; chemoattractant 1. Introduction The eosinophil-derived neurotoxin (EDN/RNase 2) is one of the four major secretory proteins found in the specific granules of the human eosinophilic leukocyte (Figure 1). EDN, and its more highly charged and cytotoxic paralog, the eosinophil cationic protein (ECP/RNase 3) are released from eosinophil granules when these cells are activated by cytokines and other proinflammatory mediators [1,2]. -

Human Cathelicidin LL-37 Is a Chemoattractant for Eosinophils and Neutrophils That Acts Via Formyl-Peptide Receptors
Original Paper Int Arch Allergy Immunol 2006;140:103–112 Received: January 3, 2005 Accepted after revision: December 19, 2005 DOI: 10.1159/000092305 Published online: March 24, 2006 Human Cathelicidin LL-37 Is a Chemoattractant for Eosinophils and Neutrophils That Acts via Formyl-Peptide Receptors a a b G. Sandra Tjabringa Dennis K. Ninaber Jan Wouter Drijfhout a a Klaus F. Rabe Pieter S. Hiemstra a b Departments of Pulmonology, and Immunohematology and Blood Transfusion, Leiden University Medical Center, Leiden , The Netherlands Key Words ting using antibodies directed against phosphorylated Eosinophils Neutrophils Chemotaxis Antimicrobial ERK1/2. Results: Our results show that LL-37 chemoat- peptides Innate immunity Lung infl ammation tracts both eosinophils and neutrophils. The FPR antag- onistic peptide tBoc-MLP inhibited LL-37-induced che- motaxis. Whereas the FPR agonist fMLP activated ERK1/2 Abstract in neutrophils, LL-37 did not, indicating that fMLP and Background: Infl ammatory lung diseases such as asth- LL-37 deliver different signals through FPRs. Conclu- ma and chronic obstructive pulmonary disease (COPD) sions: LL-37 displays chemotactic activity for eosinophils are characterized by the presence of eosinophils and and neutrophils, and this activity is mediated via an FPR. neutrophils. However, the mechanisms that mediate the These results suggest that LL-37 may play a role in in- infl ux of these cells are incompletely understood. Neu- fl ammatory lung diseases such as asthma and COPD. trophil products, including neutrophil elastase and anti- Copyright © 2006 S. Karger AG, Basel microbial peptides such as neutrophil defensins and LL- 37, have been demonstrated to display chemotactic activity towards cells from both innate and adaptive im- Introduction munity. -

Independent Evolution of Four Heme Peroxidase Superfamilies
Archives of Biochemistry and Biophysics xxx (2015) xxx–xxx Contents lists available at ScienceDirect Archives of Biochemistry and Biophysics journal homepage: www.elsevier.com/locate/yabbi Independent evolution of four heme peroxidase superfamilies ⇑ Marcel Zámocky´ a,b, , Stefan Hofbauer a,c, Irene Schaffner a, Bernhard Gasselhuber a, Andrea Nicolussi a, Monika Soudi a, Katharina F. Pirker a, Paul G. Furtmüller a, Christian Obinger a a Department of Chemistry, Division of Biochemistry, VIBT – Vienna Institute of BioTechnology, University of Natural Resources and Life Sciences, Muthgasse 18, A-1190 Vienna, Austria b Institute of Molecular Biology, Slovak Academy of Sciences, Dúbravská cesta 21, SK-84551 Bratislava, Slovakia c Department for Structural and Computational Biology, Max F. Perutz Laboratories, University of Vienna, A-1030 Vienna, Austria article info abstract Article history: Four heme peroxidase superfamilies (peroxidase–catalase, peroxidase–cyclooxygenase, peroxidase–chlo- Received 26 November 2014 rite dismutase and peroxidase–peroxygenase superfamily) arose independently during evolution, which and in revised form 23 December 2014 differ in overall fold, active site architecture and enzymatic activities. The redox cofactor is heme b or Available online xxxx posttranslationally modified heme that is ligated by either histidine or cysteine. Heme peroxidases are found in all kingdoms of life and typically catalyze the one- and two-electron oxidation of a myriad of Keywords: organic and inorganic substrates. In addition to this peroxidatic activity distinct (sub)families show pro- Heme peroxidase nounced catalase, cyclooxygenase, chlorite dismutase or peroxygenase activities. Here we describe the Peroxidase–catalase superfamily phylogeny of these four superfamilies and present the most important sequence signatures and active Peroxidase–cyclooxygenase superfamily Peroxidase–chlorite dismutase superfamily site architectures. -
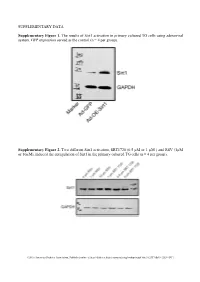
SUPPLEMENTARY DATA Supplementary Figure 1. The
SUPPLEMENTARY DATA Supplementary Figure 1. The results of Sirt1 activation in primary cultured TG cells using adenoviral system. GFP expression served as the control (n = 4 per group). Supplementary Figure 2. Two different Sirt1 activators, SRT1720 (0.5 µM or 1 µM ) and RSV (1µM or 10µM), induced the upregulation of Sirt1 in the primary cultured TG cells (n = 4 per group). ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. Primers used in qPCR Gene Name Primer Sequences Product Size (bp) Sirt1 F: tgccatcatgaagccagaga 241 (NM_001159589) R: aacatcgcagtctccaagga NOX4 F: tgtgcctttattgtgcggag 172 (NM_001285833.1) R: gctgatacactggggcaatg Supplementary Table 2. Antibodies used in Western blot or Immunofluorescence Antibody Company Cat. No Isotype Dilution Sirt1 Santa Cruz * sc-15404 Rabbit IgG 1/200 NF200 Sigma** N5389 Mouse IgG 1/500 Tubulin R&D# MAB1195 Mouse IgG 1/500 NOX4 Abcam† Ab133303 Rabbit IgG 1/500 NOX2 Abcam Ab129068 Rabbit IgG 1/500 phospho-AKT CST‡ #4060 Rabbit IgG 1/500 EGFR CST #4267 Rabbit IgG 1/500 Ki67 Santa Cruz sc-7846 Goat IgG 1/500 * Santa Cruz Biotechnology, Santa Cruz, CA, USA ** Sigma aldrich, Shanghai, China # R&D Systems Inc, Minneapolis, MN, USA † Abcam, Inc., Cambridge, MA, USA ‡ Cell Signaling Technology, Inc., Danvers, MA, USA ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary -
Instant Notes: Immunology, Second Edition
Immunology Second Edition The INSTANT NOTES series Series Editor: B.D. Hames School of Biochemistry and Molecular Biology, University of Leeds, Leeds, UK Animal Biology 2nd edition Biochemistry 2nd edition Bioinformatics Chemistry for Biologists 2nd edition Developmental Biology Ecology 2nd edition Immunology 2nd edition Genetics 2nd edition Microbiology 2nd edition Molecular Biology 2nd edition Neuroscience Plant Biology Chemistry series Consulting Editor: Howard Stanbury Analytical Chemistry Inorganic Chemistry 2nd edition Medicinal Chemistry Organic Chemistry 2nd edition Physical Chemistry Psychology series Sub-series Editor: Hugh Wagner Dept of Psychology, University of Central Lancashire, Preston, UK Psychology Cognitive Psychology Forthcoming title Physiological Psychology Immunology Second Edition P.M. Lydyard Department of Immunology and Molecular Pathology, Royal Free and University College Medical School, University College London, London, UK A. Whelan Department of Immunology, Trinity College and St James’ Hospital, Dublin, Ireland and M.W. Fanger Department of Microbiology and Immunology, Dartmouth Medical School, Lebanon, New Hampshire, USA © Garland Science/BIOS Scientific Publishers Limited, 2004 First published 2000 This edition published in the Taylor & Francis e-Library, 2005. “To purchase your own copy of this or any of Taylor & Francis or Routledge’s collection of thousands of eBooks please go to www.eBookstore.tandf.co.uk.” Second edition published 2004 All rights reserved. No part of this book may be reproduced or -

Rapid Publication
Rapid Publication Monocyte-Chemotactic Activity of Defensins from Human Neutrophils Mary C. Territo,* Tomas Ganz,** Michael E. Selsted,*1 and Robert Lehrer*II Departments of*Medicine and §Pathology, and * Will Rogers Institute Pulmonary Research Laboratory, Centerfor the Health Sciences, University ofCalifornia, Los Angeles, California 90024; and IlDepartment ofMedicine, Veterans Administration Medical Center, West Los Angeles, California 90073 Abstract Methods We investigated the monocyte-chemotactic activity of frac- Leukocytes for chemotactic studies were obtained from heparinized tionated extracts of human neutrophil granules. Monocyte- peripheral blood by Ficoll-Hypaque density separation to obtain chemotactic activity was found predominantly in the defensin- mononuclear cells, followed by dextran sedimentation to obtain neu- trophils (5). Cells were washed and resuspended at 106 monocytes or containing fraction of the neutrophil granules. Purified prepa- neutrophils/ml in HBSS containing 0.1% BSA (Calbiochem-Behring rations of each of the three human defensins (HNP-1, HNP-2, Corp., La Jolla, CA). HNP-3) were then tested. HNP-1 demonstrated significant Granule-rich fractions were prepared from neutrophils from single chemotactic activity for monocytes: Peak activity was seen at donor leukophoresis packs (Hemacare, Van Nuys, CA) containing 1-3 HNP-1 concentrations of 5 X 10' M and was 49±20% X 10'° cells, of which > 90% were viable PMN. After suspension in (mean±SE, n = 9) of that elicited by 10-8 M FMLP. HNP-2 HBSS (pH 7.4) with 2.5 mM MgCl2, the cell suspension was sealed in a (peak activity at 5 X i0' M) was somewhat less active, yield- nitrogen "bomb" (Parr Instrument Co., Moline, IL) and pressurized to ing 19±10% (n = 11). -

The Lactoferrin Receptor May Mediate the Reduction of Eosinophils in the Duodenum of Pigs Consuming Milk Containing Recombinant Human Lactoferrin
Biometals DOI 10.1007/s10534-014-9778-8 The lactoferrin receptor may mediate the reduction of eosinophils in the duodenum of pigs consuming milk containing recombinant human lactoferrin Caitlin Cooper • Eric Nonnecke • Bo Lo¨nnerdal • James Murray Received: 11 February 2014 / Accepted: 16 July 2014 Ó Springer Science+Business Media New York 2014 Abstract Lactoferrin is part of the immune system effects of consumption of milk containing recombi- and multiple tissues including the gastrointestinal (GI) nant human lactoferrin (rhLF-milk) on small intestinal tract, liver, and lung contain receptors for lactoferrin. eosinophils and expression of eosinophilic cytokines. Lactoferrin has many functions, including antimicro- In addition, LFR localization was analyzed in duode- bial, immunomodulatory, and iron binding. Addition- num and circulating eosinophils to determine if the ally, lactoferrin inhibits the migration of eosinophils, LFR could play a role in lactoferrin’s ability to inhibit which are constitutively present in the GI tract, and eosinophil migration. In the duodenum there were increase during inflammation. Lactoferrin suppresses significantly fewer eosinophils/unit area in pigs fed eosinophil infiltration into the lungs and eosinophil rhLF-milk compared to pigs fed control milk migration in -vitro. Healthy pigs have a large popu- (p = 0.025); this was not seen in the ileum lation of eosinophils in their small intestine and like (p = 0.669). In the duodenum, no differences were humans, pigs have small intestinal lactoferrin recep- observed in expression of the LFR, or any eosinophil tors (LFR); thus, pigs were chosen to investigate the migratory cytokines, and the amount of LFR protein was not different (p = 0.386). -

Specific Granule Deficiency Karen J
Selective Defect in Myeloid Cell Lactoferrin Gene Expression in Neutrophil Specific Granule Deficiency Karen J. Lomax,* John 1. Gallin,* Daniel Rotrosen,* Gordon D. Raphael,* Michael A. Kaliner,* Edward J. Benz, Jr.,* Laurence A. Boxer,§ and Harry L. Malech* *Bacterial Diseases Section and Allergic Diseases Section, Laboratory of Clinical Investigation, National Institute ofAllergy and Infectious Diseases, National Institutes ofHealth, Bethesda, Maryland 20892; tDepartment ofMedicine, Yale University, New Haven, Connecticut 06510; and §Department ofPediatrics, University ofMichigan, Ann Arbor, Michigan 48109 Abstract After subcellular fractionation of the granule components of SGD neutrophils on a sucrose gradient, the primary granule Neutrophil specific granule deficiency (SGD) is a congenital fraction is seen as a single broad band that is less dense than disorder associated with an impaired inflammatory response normal and the band of the expected density for specific gran- and a deficiency of several granule proteins. The underlying ules is absent (3-5). These abnormal banding patterns are as- abnormality causing the deficiencies is unknown. We exam- sociated with the absence or deficiency of a subset of neutro- ined mRNA transcription and protein synthesis of two neutro- phil secretory proteins that may not be limited to those usually phil granule proteins, lactoferrin and myeloperoxidase in SGD. found in specific granules such as lactoferrin and vitamin Metabolically labeled SGD nucleated marrow cells produced B- 12-binding protein. Other granule proteins, such as the pri- normal amounts of myeloperoxidase, but there was no detect- mary granule protein, defensin (6), and the tertiary granule able synthesis of lactoferrin. Transcripts of the expected size protein, gelatinase (2, 7) are also deficient. -

Fecal Eosinophil Granule-Derived Proteins Reflect Disease Activity In
THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 94, No. 12, 1999 © 1999 by Am. Coll. of Gastroenterology ISSN 0002-9270/99/$20.00 Published by Elsevier Science Inc. PII S0002-9270(99)00699-1 Fecal Eosinophil Granule-Derived Proteins Reflect Disease Activity in Inflammatory Bowel Disease Osamu Saitoh, M.D., Keishi Kojima, M.D., Kazunori Sugi, M.D., Ryoichi Matsuse, Ph.D., Kazuo Uchida, Ph.D., Kazue Tabata, Ph.D., Ken Nakagawa, M.D., Masanobu Kayazawa, M.D., Ichiro Hirata, M.D., and Ken-ichi Katsu, M.D. Second Department of Internal Medicine, Osaka Medical College, Takatsuki, and Kyoto Medical Science Laboratory, Kyoto, Japan OBJECTIVES: The aims of this study were: 1) to examine peroxidase (EPO). These proteins have potent cytotoxic whether the fecal levels of eosinophil granule-derived pro- action and are released from the cells after activation and teins reflect disease activity in inflammatory bowel disease stimulation of the cells (3). Intestinal mucosa of the patients (IBD); and 2) to examine the extracellular release of these with inflammatory bowel disease (IBD) is characterized by proteins from eosinophils and their stability in feces by an in epithelial cell damage and infiltration of various inflamma- vitro study. tory cells. The inflammatory cells include neutrophils, lym- phocytes, plasma cells, macrophages, and eosinophils. Neu- METHODS: We investigated 42 patients with ulcerative co- trophils contain various proteins such as lactoferrin, PMN litis (UC), 37 patients with Crohn’s disease (CD), and 29 (PMN)-elastase, myeloperoxidase, and lysozyme in their control subjects. The stool samples were collected at 4°C granules. We previously reported that the fecal levels of over 48 h and were homogenized. -

Comparative Properties of the Charcot-Leyden Crystal Protein and the Major Basic Protein from Human Eosinophils
Comparative properties of the Charcot-Leyden crystal protein and the major basic protein from human eosinophils. G J Gleich, … , K G Mann, J E Maldonado J Clin Invest. 1976;57(3):633-640. https://doi.org/10.1172/JCI108319. Research Article Guinea pig eosinophil granules contain a protein, the major basic protein (MBP), which accounts for more than half of the total granule protein, has a high content of arginine, and displays a remarkable tendency to form disulfide-linked aggregates. In this study we have purified a similar protein from human eosinophil granules and have compared the human MBP to the protein comprising the Charcot-Leyden crystal (CLC). Eosinophils from patients with various diseases were purified and disrupted, and the granule fraction was obtained. Examination of the granule fraction by transmission electron microscopy showed numerous typical eosinophil granules. Analyses of granule lysates by gel filtration and by polyacrylamide gel electrophoresis revealed the presence of peroxidase and MBP with properties similar to that previously found in guinea pig eosinophil granules. The human MBP had a molecular weight of 9,200, contained less than 1% carbohydrate, was rich in arginine, and readily formed disulfide-bonded aggregates. CLC were prepared from eosinophil-rich cell suspensions by homogenization in hypotonic saline. The supernates following centrifugation of cell debris spontaneously formed CLC. Analysis of CLC revealed the presence of a protein with a molecular weight of 13,000 containing 1.2% carbohydrate. The protein displayed a remarkable tendency to aggregate even in the presence of 0.2 M acetic acid. Human MBP and CLC protein differed in their molecular weights, carbohydrate compositions, and amino […] Find the latest version: https://jci.me/108319/pdf Comparative Properties of the Charcot-Leyden Crystal Protein and the Major Basic Protein from Human Eosinophils GERALD J. -

Selective Defect in Myeloid Cell Lactoferrin Gene Expression in Neutrophil Specific Granule Deficiency
Selective defect in myeloid cell lactoferrin gene expression in neutrophil specific granule deficiency. K J Lomax, … , L A Boxer, H L Malech J Clin Invest. 1989;83(2):514-519. https://doi.org/10.1172/JCI113912. Research Article Neutrophil specific granule deficiency (SGD) is a congenital disorder associated with an impaired inflammatory response and a deficiency of several granule proteins. The underlying abnormality causing the deficiencies is unknown. We examined mRNA transcription and protein synthesis of two neutrophil granule proteins, lactoferrin and myeloperoxidase in SGD. Metabolically labeled SGD nucleated marrow cells produced normal amounts of myeloperoxidase, but there was no detectable synthesis of lactoferrin. Transcripts of the expected size for lactoferrin were detectable in the nucleated marrow cells of two SGD patients, but were markedly diminished in abundance when compared with normal nucleated marrow cell RNA. Because lactoferrin is secreted by the glandular epithelia of several tissues, we also assessed lactoferrin in the nasal secretions of one SGD patient by ELISA and immunoblotting. Nasal secretory lactoferrin was the same molecular weight as neutrophil lactoferrin and was secreted in normal amounts. From these data, we conclude that lactoferrin deficiency in SGD neutrophils is tissue specific and is secondary to an abnormality of RNA production. We speculate that the deficiency of several granule proteins is due to a common defect in regulation of transcription that is responsible for the abnormal myeloid differentiation seen in SGD patients. Find the latest version: https://jci.me/113912/pdf Selective Defect in Myeloid Cell Lactoferrin Gene Expression in Neutrophil Specific Granule Deficiency Karen J. Lomax,* John 1.