A Defense Associated Peroxidase from Lemon Having Dye
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Elevated Hydrogen Peroxide and Decreased Catalase and Glutathione
Sullivan-Gunn and Lewandowski BMC Geriatrics 2013, 13:104 http://www.biomedcentral.com/1471-2318/13/104 RESEARCH ARTICLE Open Access Elevated hydrogen peroxide and decreased catalase and glutathione peroxidase protection are associated with aging sarcopenia Melanie J Sullivan-Gunn1 and Paul A Lewandowski2* Abstract Background: Sarcopenia is the progressive loss of skeletal muscle that contributes to the decline in physical function during aging. A higher level of oxidative stress has been implicated in aging sarcopenia. The current study aims to determine if the higher level of oxidative stress is a result of increased superoxide (O2‾ ) production by the NADPH oxidase (NOX) enzyme or decrease in endogenous antioxidant enzyme protection. Methods: Female Balb/c mice were assigned to 4 age groups; 6, 12, 18 and 24 months. Body weight and animal survival rates were recorded over the course of the study. Skeletal muscle tissues were collected and used to measure NOX subunit mRNA, O2‾ levels and antioxidant enzymes. Results: Key subunit components of NOX expression were elevated in skeletal muscle at 18 months, when sarcopenia was first evident. Increased superoxide dismutase 1 (SOD1) activity suggests an increase in O2‾ dismutation and this was further supported by elevated levels of hydrogen peroxide (H2O2) and decline in catalase and glutathione peroxidase (GPx) antioxidant protection in skeletal muscle at this time. NOX expression was also higher in skeletal muscle at 24 months, however this was coupled with elevated levels of O2‾ and a decline in SOD1 activity, compared to 6 and 12 months but was not associated with further loss of muscle mass. -

Catalase and Oxidase Test
CATALASE TEST Catalase is the enzyme that breaks hydrogen peroxide (H 2O2) into H 2O and O 2. Hydrogen peroxide is often used as a topical disinfectant in wounds, and the bubbling that is seen is due to the evolution of O 2 gas. H 2O2 is a potent oxidizing agent that can wreak havoc in a cell; because of this, any cell that uses O 2 or can live in the presence of O 2 must have a way to get rid of the peroxide. One of those ways is to make catalase. PROCEDURE a. Place a small amount of growth from your culture onto a clean microscope slide. If using colonies from a blood agar plate, be very careful not to scrape up any of the blood agar— blood cells are catalase positive and any contaminating agar could give a false positive. b. Add a few drops of H 2O2 onto the smear. If needed, mix with a toothpick. DO NOT use a metal loop or needle with H 2O2; it will give a false positive and degrade the metal. c. A positive result is the rapid evolution of O 2 as evidenced by bubbling. d. A negative result is no bubbles or only a few scattered bubbles. e. Dispose of your slide in the biohazard glass disposal container. Dispose of any toothpicks in the Pipet Keeper. OXIDASE TEST Basically, this is a test to see if an organism is an aerobe. It is a check for the presence of the electron transport chain that is the final phase of aerobic respiration. -

Myeloperoxidase Mediates Cell Adhesion Via the Αmβ2 Integrin (Mac-1, Cd11b/CD18)
Journal of Cell Science 110, 1133-1139 (1997) 1133 Printed in Great Britain © The Company of Biologists Limited 1997 JCS4390 Myeloperoxidase mediates cell adhesion via the αMβ2 integrin (Mac-1, CD11b/CD18) Mats W. Johansson1,*, Manuel Patarroyo2, Fredrik Öberg3, Agneta Siegbahn4 and Kenneth Nilsson3 1Department of Physiological Botany, University of Uppsala, Villavägen 6, S-75236 Uppsala, Sweden 2Microbiology and Tumour Biology Centre, Karolinska Institute, PO Box 280, S-17177 Stockholm, Sweden 3Department of Pathology, University of Uppsala, University Hospital, S-75185 Uppsala, Sweden 4Department of Clinical Chemistry, University of Uppsala, University Hospital, S-75185 Uppsala, Sweden *Author for correspondence (e-mail: [email protected]) SUMMARY Myeloperoxidase is a leukocyte component able to to αM (CD11b) or to β2 (CD18) integrin subunits, but not generate potent microbicidal substances. A homologous by antibodies to αL (CD11a), αX (CD11c), or to other invertebrate blood cell protein, peroxinectin, is not only integrins. Native myeloperoxidase mediated dose- a peroxidase but also a cell adhesion ligand. We demon- dependent cell adhesion down to relatively low concen- strate in this study that human myeloperoxidase also trations, and denaturation abolished the adhesion mediates cell adhesion. Both the human myeloid cell line activity. It is evident that myeloperoxidase supports cell HL-60, when differentiated by treatment with 12-O- adhesion, a function which may be of considerable tetradecanoyl-phorbol-13-acetate (TPA) or retinoic acid, importance for leukocyte migration and infiltration in and human blood leukocytes, adhered to myeloperoxi- inflammatory reactions, that αMβ2 integrin (Mac-1 or dase; however, undifferentiated HL-60 cells showed only CD11b/CD18) mediates this adhesion, and that the αMβ2 minimal adhesion. -

Francisella Tularensis 6/06 Tularemia Is a Commonly Acquired Laboratory Colony Morphology Infection; All Work on Suspect F
Francisella tularensis 6/06 Tularemia is a commonly acquired laboratory Colony Morphology infection; all work on suspect F. tularensis cultures .Aerobic, fastidious, requires cysteine for growth should be performed at minimum under BSL2 .Grows poorly on Blood Agar (BA) conditions with BSL3 practices. .Chocolate Agar (CA): tiny, grey-white, opaque A colonies, 1-2 mm ≥48hr B .Cysteine Heart Agar (CHA): greenish-blue colonies, 2-4 mm ≥48h .Colonies are butyrous and smooth Gram Stain .Tiny, 0.2–0.7 μm pleomorphic, poorly stained gram-negative coccobacilli .Mostly single cells Growth on BA (A) 48 h, (B) 72 h Biochemical/Test Reactions .Oxidase: Negative A B .Catalase: Weak positive .Urease: Negative Additional Information .Can be misidentified as: Haemophilus influenzae, Actinobacillus spp. by automated ID systems .Infective Dose: 10 colony forming units Biosafety Level 3 agent (once Francisella tularensis is . Growth on CA (A) 48 h, (B) 72 h suspected, work should only be done in a certified Class II Biosafety Cabinet) .Transmission: Inhalation, insect bite, contact with tissues or bodily fluids of infected animals .Contagious: No Acceptable Specimen Types .Tissue biopsy .Whole blood: 5-10 ml blood in EDTA, and/or Inoculated blood culture bottle Swab of lesion in transport media . Gram stain Sentinel Laboratory Rule-Out of Francisella tularensis Oxidase Little to no growth on BA >48 h Small, grey-white opaque colonies on CA after ≥48 h at 35/37ºC Positive Weak Negative Positive Catalase Tiny, pleomorphic, faintly stained, gram-negative coccobacilli (red, round, and random) Perform all additional work in a certified Class II Positive Biosafety Cabinet Weak Negative Positive *Oxidase: Negative Urease *Catalase: Weak positive *Urease: Negative *Oxidase, Catalase, and Urease: Appearances of test results are not agent-specific. -

Independent Evolution of Four Heme Peroxidase Superfamilies
Archives of Biochemistry and Biophysics xxx (2015) xxx–xxx Contents lists available at ScienceDirect Archives of Biochemistry and Biophysics journal homepage: www.elsevier.com/locate/yabbi Independent evolution of four heme peroxidase superfamilies ⇑ Marcel Zámocky´ a,b, , Stefan Hofbauer a,c, Irene Schaffner a, Bernhard Gasselhuber a, Andrea Nicolussi a, Monika Soudi a, Katharina F. Pirker a, Paul G. Furtmüller a, Christian Obinger a a Department of Chemistry, Division of Biochemistry, VIBT – Vienna Institute of BioTechnology, University of Natural Resources and Life Sciences, Muthgasse 18, A-1190 Vienna, Austria b Institute of Molecular Biology, Slovak Academy of Sciences, Dúbravská cesta 21, SK-84551 Bratislava, Slovakia c Department for Structural and Computational Biology, Max F. Perutz Laboratories, University of Vienna, A-1030 Vienna, Austria article info abstract Article history: Four heme peroxidase superfamilies (peroxidase–catalase, peroxidase–cyclooxygenase, peroxidase–chlo- Received 26 November 2014 rite dismutase and peroxidase–peroxygenase superfamily) arose independently during evolution, which and in revised form 23 December 2014 differ in overall fold, active site architecture and enzymatic activities. The redox cofactor is heme b or Available online xxxx posttranslationally modified heme that is ligated by either histidine or cysteine. Heme peroxidases are found in all kingdoms of life and typically catalyze the one- and two-electron oxidation of a myriad of Keywords: organic and inorganic substrates. In addition to this peroxidatic activity distinct (sub)families show pro- Heme peroxidase nounced catalase, cyclooxygenase, chlorite dismutase or peroxygenase activities. Here we describe the Peroxidase–catalase superfamily phylogeny of these four superfamilies and present the most important sequence signatures and active Peroxidase–cyclooxygenase superfamily Peroxidase–chlorite dismutase superfamily site architectures. -
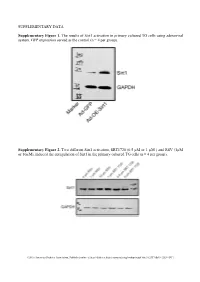
SUPPLEMENTARY DATA Supplementary Figure 1. The
SUPPLEMENTARY DATA Supplementary Figure 1. The results of Sirt1 activation in primary cultured TG cells using adenoviral system. GFP expression served as the control (n = 4 per group). Supplementary Figure 2. Two different Sirt1 activators, SRT1720 (0.5 µM or 1 µM ) and RSV (1µM or 10µM), induced the upregulation of Sirt1 in the primary cultured TG cells (n = 4 per group). ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. Primers used in qPCR Gene Name Primer Sequences Product Size (bp) Sirt1 F: tgccatcatgaagccagaga 241 (NM_001159589) R: aacatcgcagtctccaagga NOX4 F: tgtgcctttattgtgcggag 172 (NM_001285833.1) R: gctgatacactggggcaatg Supplementary Table 2. Antibodies used in Western blot or Immunofluorescence Antibody Company Cat. No Isotype Dilution Sirt1 Santa Cruz * sc-15404 Rabbit IgG 1/200 NF200 Sigma** N5389 Mouse IgG 1/500 Tubulin R&D# MAB1195 Mouse IgG 1/500 NOX4 Abcam† Ab133303 Rabbit IgG 1/500 NOX2 Abcam Ab129068 Rabbit IgG 1/500 phospho-AKT CST‡ #4060 Rabbit IgG 1/500 EGFR CST #4267 Rabbit IgG 1/500 Ki67 Santa Cruz sc-7846 Goat IgG 1/500 * Santa Cruz Biotechnology, Santa Cruz, CA, USA ** Sigma aldrich, Shanghai, China # R&D Systems Inc, Minneapolis, MN, USA † Abcam, Inc., Cambridge, MA, USA ‡ Cell Signaling Technology, Inc., Danvers, MA, USA ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary -

Peroxisomal Disorders and Their Mouse Models Point to Essential Roles of Peroxisomes for Retinal Integrity
International Journal of Molecular Sciences Review Peroxisomal Disorders and Their Mouse Models Point to Essential Roles of Peroxisomes for Retinal Integrity Yannick Das, Daniëlle Swinkels and Myriam Baes * Lab of Cell Metabolism, Department of Pharmaceutical and Pharmacological Sciences, KU Leuven, 3000 Leuven, Belgium; [email protected] (Y.D.); [email protected] (D.S.) * Correspondence: [email protected] Abstract: Peroxisomes are multifunctional organelles, well known for their role in cellular lipid homeostasis. Their importance is highlighted by the life-threatening diseases caused by peroxisomal dysfunction. Importantly, most patients suffering from peroxisomal biogenesis disorders, even those with a milder disease course, present with a number of ocular symptoms, including retinopathy. Patients with a selective defect in either peroxisomal α- or β-oxidation or ether lipid synthesis also suffer from vision problems. In this review, we thoroughly discuss the ophthalmological pathology in peroxisomal disorder patients and, where possible, the corresponding animal models, with a special emphasis on the retina. In addition, we attempt to link the observed retinal phenotype to the underlying biochemical alterations. It appears that the retinal pathology is highly variable and the lack of histopathological descriptions in patients hampers the translation of the findings in the mouse models. Furthermore, it becomes clear that there are still large gaps in the current knowledge on the contribution of the different metabolic disturbances to the retinopathy, but branched chain fatty acid accumulation and impaired retinal PUFA homeostasis are likely important factors. Citation: Das, Y.; Swinkels, D.; Baes, Keywords: peroxisome; Zellweger; metabolism; fatty acid; retina M. Peroxisomal Disorders and Their Mouse Models Point to Essential Roles of Peroxisomes for Retinal Integrity. -

Kinetic Approaches to Measuring Peroxiredoxin Reactivity
Mol. Cells 2016; 39(1): 26-30 http://dx.doi.org/10.14348/molcells.2016.2325 Molecules and Cells http://molcells.org Established in 1990 Kinetic Approaches to Measuring Peroxiredoxin Reactivity Christine C. Winterbourn*, and Alexander V. Peskin Peroxiredoxins are ubiquitous thiol proteins that catalyse demonstrated surprisingly low reactivity with thiol reagents such the breakdown of peroxides and regulate redox activity in as iodoacetamide and other oxidants such as chloramines the cell. Kinetic analysis of their reactions is required in (Peskin et al., 2007) and it is clear that the low pKa of the active order to identify substrate preferences, to understand how site thiol is insufficient to confer the high peroxide reactivity. In molecular structure affects activity and to establish their fact, typical low molecular weight and protein thiolates react -1 -1 physiological functions. Various approaches can be taken, with H2O2 with a rate constant of 20 M s whereas values of including the measurement of rates of individual steps in Prxs are 105-106 fold higher (Winterbourn and Hampton, 2008). the reaction pathway by stopped flow or competitive kinet- An elegant series of structural and mutational studies (Hall et al., ics, classical enzymatic analysis and measurement of pe- 2010; Nakamura et al., 2010; Nagy et al., 2011) have shown roxidase activity. Each methodology has its strengths and that to get sufficient rate enhancement, it is necessary to acti- they can often give complementary information. However, vate the peroxide. As discussed in detail elsewhere (Hall et al., it is important to understand the experimental conditions 2010; 2011), this involves formation of a transition state in of the assay so as to interpret correctly what parameter is which hydrogen bonding of the peroxide to conserved Arg and being measured. -

Understanding the Structure and Function of Catalases: Clues from Molecular Evolution and in Vitro Mutagenesis
PERGAMON Progress in Biophysics & Molecular Biology 72 (1999) 19±66 Understanding the structure and function of catalases: clues from molecular evolution and in vitro mutagenesis Marcel Za mocky *, Franz Koller Institut fuÈr Biochemie and Molekulare Zellbiologie and Ludwig Boltzmann Forschungsstelle fuÈr Biochemie, Vienna Biocenter, Dr. Bohr-Gasse 9, A-1030 Wien, Austria Abstract This review gives an overview about the structural organisation of dierent evolutionary lines of all enzymes capable of ecient dismutation of hydrogen peroxide. Major potential applications in biotechnology and clinical medicine justify further investigations. According to structural and functional similarities catalases can be divided in three subgroups. Typical catalases are homotetrameric haem proteins. The three-dimensional structure of six representatives has been resolved to atomic resolution. The central core of each subunit reveals a chracteristic ``catalase fold'', extremely well conserved among this group. In the native tetramer structure pairs of subunits tightly interact via exchange of their N- terminal arms. This pseudo-knot structures implies a highly ordered assembly pathway. A minor subgroup (``large catalases'') possesses an extra ¯avodoxin-like C-terminal domain. A r25AÊ long channel leads from the enzyme surface to the deeply buried active site. It enables rapid and selective diusion of the substrates to the active center. In several catalases NADPH is tightly bound close to the surface. This cofactor may prevent and reverse the formation of compound II, an inactive reaction intermediate. Bifunctional catalase-peroxidases are haem proteins which probably arose via gene duplication of an ancestral peroxidase gene. No detailed structural information is currently available. Even less is know about manganese catalases. -

Lactoperoxidase Antibacterial System: Natural Occurrence, Biological Functions and Practical Applications
724 Journal of Food Protection, Vol. 47. No. 9, Pages 724-732 (September 1984) Copyright®, International Association of Milk, Food, and Environmental Sanitarians Lactoperoxidase Antibacterial System: Natural Occurrence, Biological Functions and Practical Applications BRUNO REITER1 and GORAN HARNULV2* Downloaded from http://meridian.allenpress.com/jfp/article-pdf/47/9/724/1650811/0362-028x-47_9_724.pdf by guest on 29 September 2021 National Institute for Research in Dairying, Shinfield, Reading, RG2 9AT, England and Alfa-Laval Agri International AS, P.O. Box 39, S-I47 00 Tumba, Sweden (Received for publication January 30, 1984) ABSTRACT (37,80). The various biological functions of the LP sys tem, which have now been established, will be discussed In the present review dealing with the antibacterial lac below. toperoxidase (LP) system, it is shown that the two reactants In recent years, much work has been devoted to practi thiocyanate (SCN~) and hydrogen peroxide (H202) as well as cal applications of the LP system. The effect against oral the catalytic enzyme lactoperoxidase (LP) are widely distributed streptococci (5,19,33,101,105,107) has led to the de in nature and that evidence for the activity of the LP system velopment and marketing of a toothpaste containing nec in animals, including man, is accumulating. The in vitro effects on bacterial and animal cells are discussed and the unique ac essary ingredients to activate the LP system. Promising tion of the LP system on the bacterial cytoplasmic membrane results have also been obtained when including activating is pointed out. Some practical applications are also presented, components in the feed to calves (81,83,86) with the aim with particular emphses on the possibility of utilizing the LP of potentiating the LP system in the intestinal tract. -

Protein Engineering of a Dye Decolorizing Peroxidase from Pleurotus Ostreatus for Efficient Lignocellulose Degradation
Protein Engineering of a Dye Decolorizing Peroxidase from Pleurotus ostreatus For Efficient Lignocellulose Degradation Abdulrahman Hirab Ali Alessa A thesis submitted in partial fulfilment of the requirements for the degree of Doctor of Philosophy The University of Sheffield Faculty of Engineering Department of Chemical and Biological Engineering September 2018 ACKNOWLEDGEMENTS Firstly, I would like to express my profound gratitude to my parents, my wife, my sisters and brothers, for their continuous support and their unconditional love, without whom this would not be achieved. My thanks go to Tabuk University for sponsoring my PhD project. I would like to express my profound gratitude to Dr Wong for giving me the chance to undertake and complete my PhD project in his lab. Thank you for the continuous support and guidance throughout the past four years. I would also like to thank Dr Tee for invaluable scientific discussions and technical advices. Special thanks go to the former and current students in Wong’s research group without whom these four years would not be so special and exciting, Dr Pawel; Dr Hossam; Dr Zaki; Dr David Gonzales; Dr Inas,; Dr Yomi, Dr Miriam; Jose; Valeriane, Melvin, and Robert. ii SUMMARY Dye decolorizing peroxidases (DyPs) have received extensive attention due to their biotechnological importance and potential use in the biological treatment of lignocellulosic biomass. DyPs are haem-containing peroxidases which utilize hydrogen peroxide (H2O2) to catalyse the oxidation of a wide range of substrates. Similar to naturally occurring peroxidases, DyPs are not optimized for industrial utilization owing to their inactivation induced by excess amounts of H2O2. -

Significance of Peroxidase in Eosinophils Margaret A
University of Colorado, Boulder CU Scholar Series in Biology Ecology & Evolutionary Biology Spring 4-1-1958 Significance of peroxidase in eosinophils Margaret A. Kelsall Follow this and additional works at: http://scholar.colorado.edu/sbio Recommended Citation Kelsall, Margaret A., "Significance of peroxidase in eosinophils" (1958). Series in Biology. 14. http://scholar.colorado.edu/sbio/14 This Article is brought to you for free and open access by Ecology & Evolutionary Biology at CU Scholar. It has been accepted for inclusion in Series in Biology by an authorized administrator of CU Scholar. For more information, please contact [email protected]. SIGNIFICANCE OF PEROXIDASE IN EOSINOPHILS M a rg a ret A . K e lsa ll Peroxidase-bearing granules are the primary component and product of eosino phils. The physiological significance of eosinophils is, therefore, considered to be related to the ability of this cell to synthesize, store, and transport peroxidase and to release the peroxidase-positive granules into body fluids by a lytic process that is controlled by hormones, by variations in the histamine-epinephrine balance, and by several other stimuli. Peroxidase occurs not only in eosinophils, but also in neutrophils and blood platelets; but it is not present in most cells of animal tissues. The purpose of this work is to consider, as a working hypothesis, that the function of eosinophils is to produce, store, and transport peroxidase to catalyze oxidations. Many of the aerobic dehydrogenases that catalyze reactions in which hydrogen peroxide is produced are involved in protein catabolism. Therefore, relations between eosinophils and several normal and pathological conditions of increased protein catabolism are emphasized, and also the significance of peroxi dase in eosinophils and other leukocytes to H 20 2 produced by irradiation is considered.