Significance of Peroxidase in Eosinophils Margaret A
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Inflammation 14.11. 2004
Inflammation • Inflammation is the response of living tissue to damage. The acute inflammatory response has 3 main functions. Inflammation • The affected area is occupied by a transient material called the acute inflammatory exudate . The exudate carries proteins, fluid and cells from local blood vessels into the damaged area to mediate local defenses. • If an infective causitive agent (e.g. bacteria) is present in the damaged area, it can be destroyed and eliminated by components of the exudate . 14.11. 2004 • The damaged tissue can be broken down and partialy liquefied, and the debris removed from the site of damage. Etiology Inflammation • The cause of acute inflammation may • In all these situations, the inflammatory be due to physical damage, chemical stimulus will be met by a series of changes in substances, micro-organisms or other the human body; it will induce production of agents. The inflammatory response certain cytokines and hormones which in turn consist of changes in blood flow, will regulate haematopoiesis, protein increased permeability of blood vessels and escape of cells from the synthesis and metabolism. blood into the tissues. The changes • Most inflammatory stimuli are controlled by a are essentially the same whatever the normal immune system. The human immune cause and wherever the site. system is divided into two parts which • Acute inflammation is short-lasting, constantly and closely collaborate - the innate lasting only a few days. and the adaptive immune system. Inflammation Syst emic manifesta tion • The innate system reacts promptly without specificity and memory. Phagocytic cells are important contributors in innate of inflammation reactivity together with enzymes, complement activation and acute phase proteins. -

WHITE BLOOD CELLS Formation Function ~ TEST YOURSELF
Chapter 9 Blood, Lymph, and Immunity 231 WHITE BLOOD CELLS All white blood cells develop in the bone marrow except Any nucleated cell normally found in blood is a white blood for some lymphocytes (they start out in bone marrow but cell. White blood cells are also known as WBCs or leukocytes. develop elsewhere). At the beginning of leukopoiesis all the When white blood cells accumulate in one place, they grossly immature white blood cells look alike even though they're appear white or cream-colored. For example, pus is an accu- already committed to a specific cell line. It's not until the mulation of white blood cells. Mature white blood cells are cells start developing some of their unique characteristics larger than mature red blood cells. that we can tell them apart. There are five types of white blood cells. They are neu- Function trophils, eosinophils, basophils, monocytes and lymphocytes (Table 9-2). The function of all white blood cells is to provide a defense White blood cells can be classified in three different ways: for the body against foreign invaders. Each type of white 1. Type of defense function blood cell has its own unique role in this defense. If all the • Phagocytosis: neutrophils, eosinophils, basophils, mono- white blood cells are functioning properly, an animal has a cytes good chance of remaining healthy. Individual white blood • Antibody production and cellular immunity: lympho- cell functions will be discussed with each cell type (see cytes Table 9-2). 2. Shape of nucleus In providing defense against foreign invaders, the white • Polymorphonuclear (multilobed, segmented nucleus): blood cells do their jobs primarily out in the tissues. -

Cell Dose on Engraftment After Scts: Personalized Estimates Based on Mathematical Modeling
Bone Marrow Transplantation (2014) 49, 30–37 & 2014 Macmillan Publishers Limited All rights reserved 0268-3369/14 www.nature.com/bmt ORIGINAL ARTICLE The impact of CD34 þ cell dose on engraftment after SCTs: personalized estimates based on mathematical modeling T Stiehl1,ADHo2 and A Marciniak-Czochra1,3 It is known that the number of transplanted cells has a significant impact on the outcome after SCT. We identify issues that cannot be addressed by conventional analysis of clinical trials and ask whether it is possible to develop a refined analysis to conclude about the outcome of individual patients given clinical trial results. To accomplish this, we propose an interdisciplinary approach based on mathematical modeling. We devise and calibrate a mathematical model of short-term reconstitution and simulate treatment of large patient groups with random interindividual variation. Relating model simulations to clinical data allows quantifying the effect of transplant size on reconstitution time in the terms of patient populations and individual patients. The model confirms the existence of lower bounds on cell dose necessary for secure and efficient reconstitution but suggests that for some patient subpopulations higher thresholds might be appropriate. Simulations demonstrate that relative time gain because of increased cell dose is an ‘interpersonally stable’ parameter, in other words that slowly engrafting patients profit more from transplant enlargements than average cases. We propose a simple mathematical formula to approximate the effect of changes of transplant size on reconstitution time. Bone Marrow Transplantation (2014) 49, 30–37; doi:10.1038/bmt.2013.138; published online 23 September 2013 Keywords: SCT; hematopoiesis; mathematical modeling INTRODUCTION of benefit to individuals. -

Myeloperoxidase Mediates Cell Adhesion Via the Αmβ2 Integrin (Mac-1, Cd11b/CD18)
Journal of Cell Science 110, 1133-1139 (1997) 1133 Printed in Great Britain © The Company of Biologists Limited 1997 JCS4390 Myeloperoxidase mediates cell adhesion via the αMβ2 integrin (Mac-1, CD11b/CD18) Mats W. Johansson1,*, Manuel Patarroyo2, Fredrik Öberg3, Agneta Siegbahn4 and Kenneth Nilsson3 1Department of Physiological Botany, University of Uppsala, Villavägen 6, S-75236 Uppsala, Sweden 2Microbiology and Tumour Biology Centre, Karolinska Institute, PO Box 280, S-17177 Stockholm, Sweden 3Department of Pathology, University of Uppsala, University Hospital, S-75185 Uppsala, Sweden 4Department of Clinical Chemistry, University of Uppsala, University Hospital, S-75185 Uppsala, Sweden *Author for correspondence (e-mail: [email protected]) SUMMARY Myeloperoxidase is a leukocyte component able to to αM (CD11b) or to β2 (CD18) integrin subunits, but not generate potent microbicidal substances. A homologous by antibodies to αL (CD11a), αX (CD11c), or to other invertebrate blood cell protein, peroxinectin, is not only integrins. Native myeloperoxidase mediated dose- a peroxidase but also a cell adhesion ligand. We demon- dependent cell adhesion down to relatively low concen- strate in this study that human myeloperoxidase also trations, and denaturation abolished the adhesion mediates cell adhesion. Both the human myeloid cell line activity. It is evident that myeloperoxidase supports cell HL-60, when differentiated by treatment with 12-O- adhesion, a function which may be of considerable tetradecanoyl-phorbol-13-acetate (TPA) or retinoic acid, importance for leukocyte migration and infiltration in and human blood leukocytes, adhered to myeloperoxi- inflammatory reactions, that αMβ2 integrin (Mac-1 or dase; however, undifferentiated HL-60 cells showed only CD11b/CD18) mediates this adhesion, and that the αMβ2 minimal adhesion. -

Peroxygenase Enzymatic Activity in Plants: Ginger, Rutabaga, And
Team New Groove February 14, 2020 BIOL 495-067 Dr. Gregory Raner Research Week 2020 Abstract Title: Peroxygenase Enzymatic Activity in Plants: Ginger and Jalapeno Peppers Program of Study: Biochemistry Presentation Type: Physical Poster Subtype Oral Presentation Type: Basic Mentor(s) and Mentor Email: Dr. Gregory Raner ([email protected]) Student(s) Name(s) and Email(s): Myles Robison ([email protected]) Mason Wolk ([email protected]) Dylan Taylor ([email protected]) Abstract: Peroxidases are a ubiquitous class of enzymes found in plants fungi and other higher organisms that catalyze chemical oxidations using hydrogen peroxide as an oxidant. They are useful in a number of industrial and biotechnological applications where non-selective oxidations are required. Though a number of plant peroxidases are known, much of the focused research has occurred with a single member from this family, horseradish peroxidase. Consequently, there is an incredibly rich diversity still available for discovery in the peroxidase world, with potentially novel industrial application. The long-range objective of the research described herein is to explore a very broad range of plant sources for isolation and characterization of novel peroxidase enzymes, with enzymatic characteristics that have previously been undiscovered. Sources selected for this study include skin samples from the root of ginger, root of rutabaga, and the seeds isolated from a variety of peppers of varying pungency on the Scoville scale. Crude preparations of the peroxidases have been accomplished through crushing of the tissue with a mortar and pestle in the presence of buffer, followed by high-speed centrifugation to remove plant debris. Activity was initially screened using the enzymatic conversion of guaiacol into tetraguaiacol in the presence of H2O2. -

The Molecular Mechanism of the Catalase-Like Activity In
Article pubs.acs.org/JACS The Molecular Mechanism of the Catalase-like Activity in Horseradish Peroxidase † ∥ † ‡ ‡ § Pablo Campomanes, , Ursula Rothlisberger, Mercedes Alfonso-Prieto,*, and Carme Rovira*, , † Laboratory of Computational Chemistry and Biochemistry, Institute of Chemical Sciences and Engineering, École Polytechnique Fedéralé de Lausanne (EPFL), CH-1015 Lausanne, Switzerland ‡ Departament de Química Organicà & Institut de Química Teoricà i Computacional (IQTCUB), Universitat de Barcelona, Martí i Franques̀ 1, 08208 Barcelona, Spain § InstitucióCatalana de Recerca i Estudis Avancatş (ICREA), Passeig Lluís Companys, 23, 08018 Barcelona, Spain *S Supporting Information ABSTRACT: Horseradish peroxidase (HRP) is one of the most relevant peroxidase enzymes, used extensively in immunochemistry and biocatalysis applications. Unlike the closely related catalase enzymes, it exhibits a low activity to disproportionate hydrogen peroxide (H2O2). The origin of this disparity remains unknown due to the lack of atomistic information on the catalase-like reaction in HRP. Using QM(DFT)/MM metadynamics simulations, we uncover the mechanism for reduction of the HRP Compound I intermediate by H2O2 at atomic detail. The reaction begins with a hydrogen atom transfer, forming a peroxyl radical and a Compound II-like species. Reorientation of the peroxyl radical in the active site, concomitant with the transfer of the second hydrogen atom, is the rate-limiting step, with a computed free energy barrier (18.7 kcal/mol, ∼ 6 kcal/mol higher than the one obtained for catalase) in good agreement with experiments. Our simulations reveal the crucial role played by the distal pocket residues in accommodating H2O2, enabling formation of a Compound II-like intermediate, similar to catalases. However, out of the two pathways for Compound II reduction found in catalases, only one is operative in HRP. -
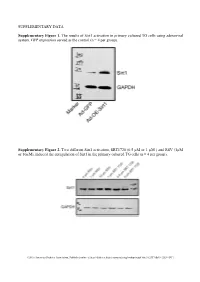
SUPPLEMENTARY DATA Supplementary Figure 1. The
SUPPLEMENTARY DATA Supplementary Figure 1. The results of Sirt1 activation in primary cultured TG cells using adenoviral system. GFP expression served as the control (n = 4 per group). Supplementary Figure 2. Two different Sirt1 activators, SRT1720 (0.5 µM or 1 µM ) and RSV (1µM or 10µM), induced the upregulation of Sirt1 in the primary cultured TG cells (n = 4 per group). ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. Primers used in qPCR Gene Name Primer Sequences Product Size (bp) Sirt1 F: tgccatcatgaagccagaga 241 (NM_001159589) R: aacatcgcagtctccaagga NOX4 F: tgtgcctttattgtgcggag 172 (NM_001285833.1) R: gctgatacactggggcaatg Supplementary Table 2. Antibodies used in Western blot or Immunofluorescence Antibody Company Cat. No Isotype Dilution Sirt1 Santa Cruz * sc-15404 Rabbit IgG 1/200 NF200 Sigma** N5389 Mouse IgG 1/500 Tubulin R&D# MAB1195 Mouse IgG 1/500 NOX4 Abcam† Ab133303 Rabbit IgG 1/500 NOX2 Abcam Ab129068 Rabbit IgG 1/500 phospho-AKT CST‡ #4060 Rabbit IgG 1/500 EGFR CST #4267 Rabbit IgG 1/500 Ki67 Santa Cruz sc-7846 Goat IgG 1/500 * Santa Cruz Biotechnology, Santa Cruz, CA, USA ** Sigma aldrich, Shanghai, China # R&D Systems Inc, Minneapolis, MN, USA † Abcam, Inc., Cambridge, MA, USA ‡ Cell Signaling Technology, Inc., Danvers, MA, USA ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary -

MOLECULAR ANALYSIS of FATTY ACID PEROXYGENASE INVOLVED in the BIOSYNTHESIS of EPOXY FATTY ACIDS in OATS (Avena Sativa)
CORE Metadata, citation and similar papers at core.ac.uk Provided by University of Saskatchewan's Research Archive MOLECULAR ANALYSIS OF FATTY ACID PEROXYGENASE INVOLVED IN THE BIOSYNTHESIS OF EPOXY FATTY ACIDS IN OATS (Avena sativa) A Thesis Submitted to the College of Graduate Studies and Research In Partial Fulfillment of the Requirements For the Degree of Master of Science In the Department of Food and Bioproduct Sciences College of Agriculture and Bioresources University of Saskatchewan Saskatoon, Saskatchewan Canada By Indika Gayani Benaragama 2015 © Indika Gayani Benaragama, October, 2015. All Rights Reserved. PERMISSION TO USE In presenting this thesis/dissertation in partial fulfillment of the requirements for a Postgraduate degree from the University of Saskatchewan, I agree that the Libraries of this University may make it freely available for inspection. I further agree that permission for copying of this thesis/dissertation in any manner, in whole or in part, for scholarly purposes may be granted by the professor or professors who supervised my thesis/dissertation work or, in their absence, by the Head of the Department or the Dean of the College in which my thesis work was done. It is understood that any copying or publication or use of this thesis/dissertation or parts thereof for financial gain shall not be allowed without my written permission. It is also understood that due recognition shall be given to me and to the University of Saskatchewan in any scholarly use which may be made of any material in my thesis/dissertation. DISCLAIMER Reference in this thesis/dissertation to any specific commercial products, process, or service by trade name, trademark, manufacturer, or otherwise, does not constitute or imply its endorsement, recommendation, or favoring by the University of Saskatchewan. -

Lactoperoxidase Antibacterial System: Natural Occurrence, Biological Functions and Practical Applications
724 Journal of Food Protection, Vol. 47. No. 9, Pages 724-732 (September 1984) Copyright®, International Association of Milk, Food, and Environmental Sanitarians Lactoperoxidase Antibacterial System: Natural Occurrence, Biological Functions and Practical Applications BRUNO REITER1 and GORAN HARNULV2* Downloaded from http://meridian.allenpress.com/jfp/article-pdf/47/9/724/1650811/0362-028x-47_9_724.pdf by guest on 29 September 2021 National Institute for Research in Dairying, Shinfield, Reading, RG2 9AT, England and Alfa-Laval Agri International AS, P.O. Box 39, S-I47 00 Tumba, Sweden (Received for publication January 30, 1984) ABSTRACT (37,80). The various biological functions of the LP sys tem, which have now been established, will be discussed In the present review dealing with the antibacterial lac below. toperoxidase (LP) system, it is shown that the two reactants In recent years, much work has been devoted to practi thiocyanate (SCN~) and hydrogen peroxide (H202) as well as cal applications of the LP system. The effect against oral the catalytic enzyme lactoperoxidase (LP) are widely distributed streptococci (5,19,33,101,105,107) has led to the de in nature and that evidence for the activity of the LP system velopment and marketing of a toothpaste containing nec in animals, including man, is accumulating. The in vitro effects on bacterial and animal cells are discussed and the unique ac essary ingredients to activate the LP system. Promising tion of the LP system on the bacterial cytoplasmic membrane results have also been obtained when including activating is pointed out. Some practical applications are also presented, components in the feed to calves (81,83,86) with the aim with particular emphses on the possibility of utilizing the LP of potentiating the LP system in the intestinal tract. -

White Blood Cells)
Lec.4 Medical Physiology – Blood Physiology Z.H.Kamil Leukocytes (White Blood Cells) Leukocytes are the only formed elements that are complete cells, with nuclei and the usual organelles. Accounting for less than 1% of total blood volume, leukocytes are far less numerous than red blood cells. On average, there are 4800–10,800 WBCs/μl of blood. Leukocytes are crucial to our defense against disease. They form a mobile army that helps protect the body from damage by bacteria, viruses, parasites, toxins, and tumor cells. As such, they have special functional characteristics. Red blood cells are kept into the bloodstream, and they carry out their functions in the blood. But white blood cells are able to slip out of the capillary blood vessels in a process called diapedesis and the circulatory system is simply their means of transport to areas of the body (mostly loose connective tissues or lymphoid tissues) where they mount inflammatory or immune responses. The signals that prompt WBCs to leave the bloodstream at specific locations are cell adhesion molecules displayed by endothelial cells forming the capillary walls at sites of inflammation. Once out of the bloodstream, leukocytes move through the tissue spaces by amoeboid motion (they form flowing cytoplasmic extensions that move them along). By following the chemical trail of molecules released by damaged cells or other leukocytes, a phenomenon called positive chemotaxis, they pinpoint areas of tissue damage and infection and gather there in large numbers to destroy foreign substances and dead cells. Whenever white blood cells are mobilized for action, the body speeds up their production and their numbers may double within a few hours. -

Blood Dyscrasias
MEDICATION-INDUCED BLOOD DYSCRASIAS Etiology And Disease Types Jassin M. Jouria, MD Dr. Jassin M. Jouria is a medical doctor, professor of academic medicine, and medical author. He graduated from Ross University School of Medicine and has completed his clinical clerkship training in various teaching hospitals throughout New York, including King’s County Hospital Center and Brookdale Medical Center, among others. Dr. Jouria has passed all USMLE medical board exams, and has served as a test prep tutor and instructor for Kaplan. He has developed several medical courses and curricula for a variety of educational institutions. Dr. Jouria has also served on multiple levels in the academic field including faculty member and Department Chair. Dr. Jouria continues to serves as a Subject Matter Expert for several continuing education organizations covering multiple basic medical sciences. He has also developed several continuing medical education courses covering various topics in clinical medicine. Recently, Dr. Jouria has been contracted by the University of Miami/Jackson Memorial Hospital’s Department of Surgery to develop an e-module training series for trauma patient management. Dr. Jouria is currently authoring an academic textbook on Human Anatomy & Physiology. Abstract Although drug-induced hematologic disorders are less common than other types of adverse reactions, they are associated with significant morbidity and mortality. Some agents, such as hemolytics, cause predictable hematologic disease, but others induce idiosyncratic reactions not directly related to the drug’s pharmacology. The most important part of managing hematologic disorders is the prompt recognition that a problem exists. The main mechanisms to manage hematologic disorders include vigilance to observe signs and symptoms indicating a blood disorder and patient education of the warning symptoms to alert them of the need to report a condition to their primary care provider or an emergency health team. -

A Strong Impact of Genetic Background on Gut Microflora in Mice
SAGE-Hindawi Access to Research International Journal of Inflammation Volume 2010, Article ID 986046, 12 pages doi:10.4061/2010/986046 Research Article A Strong Impact of Genetic Background on Gut Microflora in Mice R. Steven Esworthy,1 David D. Smith,2 and Fong-Fong Chu1 1 Department of Cancer Biology, Beckman Research Institute of the City of Hope, 1500 Duarte Road, Duarte, CA 91010-3000, USA 2 Division of Information Sciences, Beckman Research Institute of the City of Hope, 1500 Duarte Road, Duarte, CA 91010-3000, USA Correspondence should be addressed to Fong-Fong Chu, [email protected] Received 29 March 2010; Accepted 9 June 2010 Academic Editor: Gerhard Rogler Copyright © 2010 R. Steven Esworthy et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Genetic background affects susceptibility to ileocolitis in mice deficient in two intracellular glutathione peroxidases, GPx1 and GPx2. The C57BL/6 (B6) GPx1/2 double-knockout (DKO) mice have mild ileocolitis, and 129S1/Sv (129) DKO mice have severe inflammation. We used diet to modulate ileocolitis; a casein-based defined diet with AIN76A micronutrients (AIN) attenuates inflammation compared to conventional LabDiets. Because luminal microbiota induce DKO ileocolitis, we assessed bacterial composition with automated ribosomal intergenic-spacer analysis (ARISA) on cecal DNA. We found that mouse strain had the strongest impact on the composition of microbiota than diet and GPx genotypes. In comparing AIN and LabDiet, DKO mice were more resistant to change than the non-DKO or WT mice.