An in Vitro Study on the Regulation of Oxidative Protective Genes In
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Peroxiredoxins in Neurodegenerative Diseases
antioxidants Review Peroxiredoxins in Neurodegenerative Diseases Monika Szeliga Mossakowski Medical Research Centre, Department of Neurotoxicology, Polish Academy of Sciences, 5 Pawinskiego Street, 02-106 Warsaw, Poland; [email protected]; Tel.: +48-(22)-6086416 Received: 31 October 2020; Accepted: 27 November 2020; Published: 30 November 2020 Abstract: Substantial evidence indicates that oxidative/nitrosative stress contributes to the neurodegenerative diseases. Peroxiredoxins (PRDXs) are one of the enzymatic antioxidant mechanisms neutralizing reactive oxygen/nitrogen species. Since mammalian PRDXs were identified 30 years ago, their significance was long overshadowed by the other well-studied ROS/RNS defense systems. An increasing number of studies suggests that these enzymes may be involved in the neurodegenerative process. This article reviews the current knowledge on the expression and putative roles of PRDXs in neurodegenerative disorders such as Alzheimer’s disease, Parkinson’s disease and dementia with Lewy bodies, multiple sclerosis, amyotrophic lateral sclerosis and Huntington’s disease. Keywords: peroxiredoxin (PRDX); oxidative stress; nitrosative stress; neurodegenerative disease 1. Introduction Under physiological conditions, reactive oxygen species (ROS, e.g., superoxide anion, O2 -; · hydrogen peroxide, H O ; hydroxyl radical, OH; organic hydroperoxide, ROOH) and reactive nitrogen 2 2 · species (RNS, e.g., nitric oxide, NO ; peroxynitrite, ONOO-) are constantly produced as a result of normal · cellular metabolism and play a crucial role in signal transduction, enzyme activation, gene expression, and regulation of immune response [1]. The cells are endowed with several enzymatic (e.g., glutathione peroxidase (GPx); peroxiredoxin (PRDX); thioredoxin (TRX); catalase (CAT); superoxide dismutase (SOD)), and non-enzymatic (e.g., glutathione (GSH); quinones; flavonoids) antioxidant systems that minimize the levels of ROS and RNS. -

Targeted Genes and Methodology Details for Neuromuscular Genetic Panels
Targeted Genes and Methodology Details for Neuromuscular Genetic Panels Reference transcripts based on build GRCh37 (hg19) interrogated by Neuromuscular Genetic Panels Next-generation sequencing (NGS) and/or Sanger sequencing is performed Motor Neuron Disease Panel to test for the presence of a mutation in these genes. Gene GenBank Accession Number Regions of homology, high GC-rich content, and repetitive sequences may ALS2 NM_020919 not provide accurate sequence. Therefore, all reported alterations detected ANG NM_001145 by NGS are confirmed by an independent reference method based on laboratory developed criteria. However, this does not rule out the possibility CHMP2B NM_014043 of a false-negative result in these regions. ERBB4 NM_005235 Sanger sequencing is used to confirm alterations detected by NGS when FIG4 NM_014845 appropriate.(Unpublished Mayo method) FUS NM_004960 HNRNPA1 NM_031157 OPTN NM_021980 PFN1 NM_005022 SETX NM_015046 SIGMAR1 NM_005866 SOD1 NM_000454 SQSTM1 NM_003900 TARDBP NM_007375 UBQLN2 NM_013444 VAPB NM_004738 VCP NM_007126 ©2018 Mayo Foundation for Medical Education and Research Page 1 of 14 MC4091-83rev1018 Muscular Dystrophy Panel Muscular Dystrophy Panel Gene GenBank Accession Number Gene GenBank Accession Number ACTA1 NM_001100 LMNA NM_170707 ANO5 NM_213599 LPIN1 NM_145693 B3GALNT2 NM_152490 MATR3 NM_199189 B4GAT1 NM_006876 MYH2 NM_017534 BAG3 NM_004281 MYH7 NM_000257 BIN1 NM_139343 MYOT NM_006790 BVES NM_007073 NEB NM_004543 CAPN3 NM_000070 PLEC NM_000445 CAV3 NM_033337 POMGNT1 NM_017739 CAVIN1 NM_012232 POMGNT2 -

Use of Mathematical Modeling and Other Biophysical Methods For
USE OF MATHEMATICAL MODELING AND OTHER BIOPHYSICAL METHODS FOR INSIGHTS INTO IRON-RELATED PHENOMENA OF BIOLOGICAL SYSTEMS A Dissertation by JOSHUA D. WOFFORD Submitted to the Office of Graduate and Professional Studies of Texas A&M University in partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY Chair of Committee, Paul A. Lindahl Committee Members, David P. Barondeau Simon W. North Vishal M. Gohil Head of Department, Simon W. North December 2018 Major Subject: Chemistry Copyright 2018 Joshua D. Wofford ABSTRACT Iron is a crucial nutrient in most living systems. It forms the active centers of many proteins that are critical for many cellular functions, either by themselves or as Fe-S clusters and hemes. However, Fe is potentially toxic to the cell in high concentrations and must be tightly regulated. There has been much work into understanding various pieces of Fe trafficking and regulation, but integrating all of this information into a coherent model has proven difficult. Past research has focused on different Fe species, including cytosolic labile Fe or mitochondrial Fe-S clusters, as being the main regulator of Fe trafficking in yeast. Our initial modeling efforts demonstrate that both cytosolic Fe and mitochondrial ISC assembly are required for proper regulation. More recent modeling efforts involved a more rigorous multi- tiered approach. Model simulations were optimized against experimental results involving respiring wild-type and Mrs3/4-deleted yeast. Simulations from both modeling studies suggest that mitochondria possess a “respiratory shield” that prevents a vicious cycle of nanoparticle formation, ISC loss, and subsequent loading of mitochondria with iron. -

Role of Oxidative Stress in the Pathogenesis of Amyotrophic Lateral Sclerosis: Antioxidant Metalloenzymes and Therapeutic Strategies
biomolecules Review Role of Oxidative Stress in the Pathogenesis of Amyotrophic Lateral Sclerosis: Antioxidant Metalloenzymes and Therapeutic Strategies Pavlína Hemerková * and Martin Vališ Department of Neurology, Charles University, Faculty of Medicine and University Hospital Hradec Kralove, 500 05 Hradec Kralove, Czech Republic; [email protected] * Correspondence: [email protected]; Tel.: +420-731-304-371 Abstract: Amyotrophic lateral sclerosis (ALS) affects motor neurons in the cerebral cortex, brainstem and spinal cord and leads to death due to respiratory failure within three to five years. Although the clinical symptoms of this disease were first described in 1869 and it is the most common motor neuron disease and the most common neurodegenerative disease in middle-aged individuals, the exact etiopathogenesis of ALS remains unclear and it remains incurable. However, free oxygen radicals (i.e., molecules containing one or more free electrons) are known to contribute to the pathogenesis of this disease as they very readily bind intracellular structures, leading to functional impairment. Antioxidant enzymes, which are often metalloenzymes, inactivate free oxygen radicals by converting them into a less harmful substance. One of the most important antioxidant enzymes is Cu2+Zn2+ superoxide dismutase (SOD1), which is mutated in 20% of cases of the familial form of ALS (fALS) and up to 7% of sporadic ALS (sALS) cases. In addition, the proper functioning of catalase and glutathione peroxidase (GPx) is essential for antioxidant protection. In this review article, we focus on the mechanisms through which these enzymes are involved in the antioxidant response to oxidative Citation: Hemerková, P.; Vališ, M. Role of Oxidative Stress in the stress and thus the pathogenesis of ALS and their potential as therapeutic targets. -

192ICM ICBIC Abstracts
Workshop Lecture Journal of Inorganic Biochemistry 96 (2003) 3 Structural Genomics Antonio Rosato, Magnetic Resonance Center, University of Florence, Italy To realize the true value of the wealth of data provided by genome sequencing data, it is necessary to relate them to the functional properties of the proteins they encode. Since the biological function of a protein is determined by its 3D structure, the systematic determination of proteins’ structures on a genome-wide scale is a crucial step in any (post-)genomic effort, which may (or may not) provide initial hints on the function. This is what is commonly referred to as ‘Structural Genomics’ (or Structural Proteomics). Because of the huge number of systems into question, all the complex steps necessary for structure determination must be optimized, streamlined and, possibly, robotized in order to shrink the time needed to solve each protein structure. This approach is dubbed ‘high-throughput’ (HTP) and is an intrinsic feature of Structural Genomics. What can be the relationship between Biological Inorganic Chemistry and Structural Genomics? A major challenge is that to reconcile the concept of HTP with the care that metalloproteins most often require because of their metal cofactors. The identifi cation of metalloproteins is even not explicitly taken into account in purely Structural Genomics projects, nor is any methodology particularly developed for them. To create true correlations between Biological Inorganic Chemistry and Structural Genomics it is necessary to develop new computational tools (e.g. to identify metalloproteins in databanks, or to correctly model their structures), as well as new methodological approaches to HTP metalloprotein expression/purifi cation and structural characterization. -

Studies of Iron Sulfur Cluster Maturation and Transport DISSERTATION Presented in Partial Fulfillment of the Requirements for Th
Studies of Iron Sulfur Cluster Maturation and Transport DISSERTATION Presented in Partial Fulfillment of the Requirements for the Degree Doctor of Philosophy in the Graduate School of The Ohio State University By Jingwei Li Graduate Program in Chemistry The Ohio State University 2015 Dissertation Committee: Professor James A. Cowan, Advisor Professor Ross E. Dalbey Professor Claudia Turro Copyright by Jingwei Li 2015 Abstract Cellular iron homeostasis is critically dependent on sensory and regulatory mechanisms that maintain a balance of intracellular iron concentrations. Divergence from a healthy iron concentration can result in common disease states such as anemia and ataxia. With the goal of understanding the molecular basis for such health problems, and advancing the knowledge based toward potential remedies, an understanding of the molecular details of cellular iron transport and the biological chemistry of iron species is an essential prerequisite. In that regard, iron-sulfur clusters are ubiquitous iron-containing centers in a variety of proteins and serve a multitude of roles that include electron transfer, catalysis of reactions, and sensors of cellular oxygen and iron levels. Recently, a substantial body of evidence has suggested an essential role for cellular glutathione (a molecule normally implicated with eliminating reactive oxygen species from cells) in the regulation, stabilization and biosynthesis of cellular iron-sulfur clusters in humans and other complex organisms. We have demonstrated that glutathione can naturally bind to iron-sulfur cluster precursors and have isolated and characterized this species and shown it to be stable under physiological conditions. More importantly, we have demonstrated that the glutathione-bound iron-sulfur cluster can be transported by a ii critical export protein from the cellular mitochondrion. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Surface Phenotype Changes and Increased Response to Oxidative Stress in CD4+Cd25high T Cells
biomedicines Article Surface Phenotype Changes and Increased Response to Oxidative Stress in CD4+CD25high T Cells Yoshiki Yamamoto 1,*, Takaharu Negoro 2, Rui Tada 3 , Michiaki Narushima 4, Akane Hoshi 2, Yoichi Negishi 3,* and Yasuko Nakano 2 1 Department of Paediatrics, Tokyo Metropolitan Ebara Hospital, Tokyo 145-0065, Japan 2 Department of Pharmacogenomics, School of Pharmacy, Showa University, Tokyo 142-8555, Japan; [email protected] (T.N.); [email protected] (A.H.); [email protected] (Y.N.) 3 Department of Drug Delivery and Molecular Biopharmaceutics, School of Pharmacy, Tokyo University of Pharmacy and Life Sciences, Tokyo 192-0392, Japan; [email protected] 4 Department of Internal Medicine, Showa University Northern Yokohama Hospital, Kanagawa 224-8503, Japan; [email protected] * Correspondence: [email protected] (Y.Y.); [email protected] (Y.N.); Tel.: +81-3-5734-8000 (Y.Y.); +81-42-676-3182 (Y.N.) + + + + Abstract: Conversion of CD4 CD25 FOXP3 T regulatory cells (Tregs) from the immature (CD45RA ) to mature (CD45RO+) phenotype has been shown during development and allergic reactions. The relative frequencies of these Treg phenotypes and their responses to oxidative stress during devel- opment and allergic inflammation were analysed in samples from paediatric and adult subjects. The FOXP3lowCD45RA+ population was dominant in early childhood, while the percentage of high + FOXP3 CD45RO cells began increasing in the first year of life. These phenotypic changes were observed in subjects with and without asthma. Further, there was a significant increase in phospho- Citation: Yamamoto, Y.; Negoro, T.; + high rylated ERK1/2 (pERK1/2) protein in hydrogen peroxide (H2O2)-treated CD4 CD25 cells in Tada, R.; Narushima, M.; Hoshi, A.; adults with asthma compared with those without asthma. -

(ER) Membrane Contact Sites (MCS) Uses Toxic Waste to Deliver Messages Edgar Djaha Yoboue1, Roberto Sitia1 and Thomas Simmen2
Yoboue et al. Cell Death and Disease (2018) 9:331 DOI 10.1038/s41419-017-0033-4 Cell Death & Disease REVIEW ARTICLE Open Access Redox crosstalk at endoplasmic reticulum (ER) membrane contact sites (MCS) uses toxic waste to deliver messages Edgar Djaha Yoboue1, Roberto Sitia1 and Thomas Simmen2 Abstract Many cellular redox reactions housed within mitochondria, peroxisomes and the endoplasmic reticulum (ER) generate hydrogen peroxide (H2O2) and other reactive oxygen species (ROS). The contribution of each organelle to the total cellular ROS production is considerable, but varies between cell types and also over time. Redox-regulatory enzymes are thought to assemble at a “redox triangle” formed by mitochondria, peroxisomes and the ER, assembling “redoxosomes” that sense ROS accumulations and redox imbalances. The redoxosome enzymes use ROS, potentially toxic by-products made by some redoxosome members themselves, to transmit inter-compartmental signals via chemical modifications of downstream proteins and lipids. Interestingly, important components of the redoxosome are ER chaperones and oxidoreductases, identifying ER oxidative protein folding as a key ROS producer and controller of the tri-organellar membrane contact sites (MCS) formed at the redox triangle. At these MCS, ROS accumulations could directly facilitate inter-organellar signal transmission, using ROS transporters. In addition, ROS influence the flux 2+ 2+ of Ca ions, since many Ca handling proteins, including inositol 1,4,5 trisphosphate receptors (IP3Rs), SERCA pumps or regulators of the mitochondrial Ca2+ uniporter (MCU) are redox-sensitive. Fine-tuning of these redox and ion signaling pathways might be difficult in older organisms, suggesting a dysfunctional redox triangle may accompany 1234567890 1234567890 the aging process. -
![A Label-Free Cellular Proteomics Approach to Decipher the Antifungal Action of Dimiq, a Potent Indolo[2,3- B]Quinoline Agent, Against Candida Albicans Biofilms](https://docslib.b-cdn.net/cover/9827/a-label-free-cellular-proteomics-approach-to-decipher-the-antifungal-action-of-dimiq-a-potent-indolo-2-3-b-quinoline-agent-against-candida-albicans-biofilms-409827.webp)
A Label-Free Cellular Proteomics Approach to Decipher the Antifungal Action of Dimiq, a Potent Indolo[2,3- B]Quinoline Agent, Against Candida Albicans Biofilms
A Label-Free Cellular Proteomics Approach to Decipher the Antifungal Action of DiMIQ, a Potent Indolo[2,3- b]Quinoline Agent, against Candida albicans Biofilms Robert Zarnowski 1,2*, Anna Jaromin 3*, Agnieszka Zagórska 4, Eddie G. Dominguez 1,2, Katarzyna Sidoryk 5, Jerzy Gubernator 3 and David R. Andes 1,2 1 Department of Medicine, School of Medicine & Public Health, University of Wisconsin-Madison, Madison, WI 53706, USA; [email protected] (E.G.D.); [email protected] (D.R.A.) 2 Department of Medical Microbiology, School of Medicine & Public Health, University of Wisconsin-Madison, Madison, WI 53706, USA 3 Department of Lipids and Liposomes, Faculty of Biotechnology, University of Wroclaw, 50-383 Wroclaw, Poland; [email protected] 4 Department of Medicinal Chemistry, Jagiellonian University Medical College, 30-688 Cracow, Poland; [email protected] 5 Department of Pharmacy, Cosmetic Chemicals and Biotechnology, Team of Chemistry, Łukasiewicz Research Network-Industrial Chemistry Institute, 01-793 Warsaw, Poland; [email protected] * Correspondence: [email protected] (R.Z.); [email protected] (A.J.); Tel.: +1-608-265-8578 (R.Z.); +48-71-3756203 (A.J.) Label-Free Cellular Proteomics of Candida albicans biofilms treated with DiMIQ Identified Proteins Accession # Alternate ID Gene names (ORF ) WT DIMIQ Z SCORE Proteins induced by DiMIQ Arginase (EC 3.5.3.1) A0A1D8PP00 CAR1 CAALFM_C504490CA 0.000 6.648 drug induced Glucan 1,3-beta-glucosidase BGL2 (EC 3.2.1.58) (Exo-1Q5AMT2 BGL2 CAALFM_C402250CA -

Independent Evolution of Four Heme Peroxidase Superfamilies
Archives of Biochemistry and Biophysics xxx (2015) xxx–xxx Contents lists available at ScienceDirect Archives of Biochemistry and Biophysics journal homepage: www.elsevier.com/locate/yabbi Independent evolution of four heme peroxidase superfamilies ⇑ Marcel Zámocky´ a,b, , Stefan Hofbauer a,c, Irene Schaffner a, Bernhard Gasselhuber a, Andrea Nicolussi a, Monika Soudi a, Katharina F. Pirker a, Paul G. Furtmüller a, Christian Obinger a a Department of Chemistry, Division of Biochemistry, VIBT – Vienna Institute of BioTechnology, University of Natural Resources and Life Sciences, Muthgasse 18, A-1190 Vienna, Austria b Institute of Molecular Biology, Slovak Academy of Sciences, Dúbravská cesta 21, SK-84551 Bratislava, Slovakia c Department for Structural and Computational Biology, Max F. Perutz Laboratories, University of Vienna, A-1030 Vienna, Austria article info abstract Article history: Four heme peroxidase superfamilies (peroxidase–catalase, peroxidase–cyclooxygenase, peroxidase–chlo- Received 26 November 2014 rite dismutase and peroxidase–peroxygenase superfamily) arose independently during evolution, which and in revised form 23 December 2014 differ in overall fold, active site architecture and enzymatic activities. The redox cofactor is heme b or Available online xxxx posttranslationally modified heme that is ligated by either histidine or cysteine. Heme peroxidases are found in all kingdoms of life and typically catalyze the one- and two-electron oxidation of a myriad of Keywords: organic and inorganic substrates. In addition to this peroxidatic activity distinct (sub)families show pro- Heme peroxidase nounced catalase, cyclooxygenase, chlorite dismutase or peroxygenase activities. Here we describe the Peroxidase–catalase superfamily phylogeny of these four superfamilies and present the most important sequence signatures and active Peroxidase–cyclooxygenase superfamily Peroxidase–chlorite dismutase superfamily site architectures. -
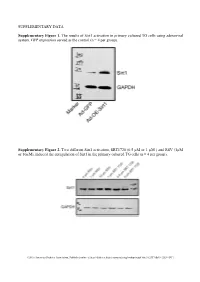
SUPPLEMENTARY DATA Supplementary Figure 1. The
SUPPLEMENTARY DATA Supplementary Figure 1. The results of Sirt1 activation in primary cultured TG cells using adenoviral system. GFP expression served as the control (n = 4 per group). Supplementary Figure 2. Two different Sirt1 activators, SRT1720 (0.5 µM or 1 µM ) and RSV (1µM or 10µM), induced the upregulation of Sirt1 in the primary cultured TG cells (n = 4 per group). ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. Primers used in qPCR Gene Name Primer Sequences Product Size (bp) Sirt1 F: tgccatcatgaagccagaga 241 (NM_001159589) R: aacatcgcagtctccaagga NOX4 F: tgtgcctttattgtgcggag 172 (NM_001285833.1) R: gctgatacactggggcaatg Supplementary Table 2. Antibodies used in Western blot or Immunofluorescence Antibody Company Cat. No Isotype Dilution Sirt1 Santa Cruz * sc-15404 Rabbit IgG 1/200 NF200 Sigma** N5389 Mouse IgG 1/500 Tubulin R&D# MAB1195 Mouse IgG 1/500 NOX4 Abcam† Ab133303 Rabbit IgG 1/500 NOX2 Abcam Ab129068 Rabbit IgG 1/500 phospho-AKT CST‡ #4060 Rabbit IgG 1/500 EGFR CST #4267 Rabbit IgG 1/500 Ki67 Santa Cruz sc-7846 Goat IgG 1/500 * Santa Cruz Biotechnology, Santa Cruz, CA, USA ** Sigma aldrich, Shanghai, China # R&D Systems Inc, Minneapolis, MN, USA † Abcam, Inc., Cambridge, MA, USA ‡ Cell Signaling Technology, Inc., Danvers, MA, USA ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary