Ceftaroline Fosamil: a Super-Cephalosporin?
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Medical Review(S) Clinical Review
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 200327 MEDICAL REVIEW(S) CLINICAL REVIEW Application Type NDA Application Number(s) 200327 Priority or Standard Standard Submit Date(s) December 29, 2009 Received Date(s) December 30, 2009 PDUFA Goal Date October 30, 2010 Division / Office Division of Anti-Infective and Ophthalmology Products Office of Antimicrobial Products Reviewer Name(s) Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD Review Completion October 29, 2010 Date Established Name Ceftaroline fosamil for injection (Proposed) Trade Name Teflaro Therapeutic Class Cephalosporin; ß-lactams Applicant Cerexa, Inc. Forest Laboratories, Inc. Formulation(s) 400 mg/vial and 600 mg/vial Intravenous Dosing Regimen 600 mg every 12 hours by IV infusion Indication(s) Acute Bacterial Skin and Skin Structure Infection (ABSSSI); Community-acquired Bacterial Pneumonia (CABP) Intended Population(s) Adults ≥ 18 years of age Template Version: March 6, 2009 Reference ID: 2857265 Clinical Review Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD NDA 200327: Teflaro (ceftaroline fosamil) Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT ......................................... 9 1.1 Recommendation on Regulatory Action ........................................................... 10 1.2 Risk Benefit Assessment.................................................................................. 10 1.3 Recommendations for Postmarketing Risk Evaluation and Mitigation Strategies ........................................................................................................................ -

Ceftazidime for Injection) PHARMACY BULK PACKAGE – NOT for DIRECT INFUSION
PRESCRIBING INFORMATION FORTAZ® (ceftazidime for injection) PHARMACY BULK PACKAGE – NOT FOR DIRECT INFUSION To reduce the development of drug-resistant bacteria and maintain the effectiveness of FORTAZ and other antibacterial drugs, FORTAZ should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. DESCRIPTION Ceftazidime is a semisynthetic, broad-spectrum, beta-lactam antibacterial drug for parenteral administration. It is the pentahydrate of pyridinium, 1-[[7-[[(2-amino-4 thiazolyl)[(1-carboxy-1-methylethoxy)imino]acetyl]amino]-2-carboxy-8-oxo-5-thia-1 azabicyclo[4.2.0]oct-2-en-3-yl]methyl]-, hydroxide, inner salt, [6R-[6α,7β(Z)]]. It has the following structure: The molecular formula is C22H32N6O12S2, representing a molecular weight of 636.6. FORTAZ is a sterile, dry-powdered mixture of ceftazidime pentahydrate and sodium carbonate. The sodium carbonate at a concentration of 118 mg/g of ceftazidime activity has been admixed to facilitate dissolution. The total sodium content of the mixture is approximately 54 mg (2.3 mEq)/g of ceftazidime activity. The Pharmacy Bulk Package vial contains 709 mg of sodium carbonate. The sodium content is approximately 54 mg (2.3mEq) per gram of ceftazidime. FORTAZ in sterile crystalline form is supplied in Pharmacy Bulk Packages equivalent to 6g of anhydrous ceftazidime. The Pharmacy Bulk Package bottle is a container of sterile preparation for parenteral use that contains many single doses. The contents are intended for use in a pharmacy admixture program and are restricted to the preparation of admixtures for intravenous use. THE PHARMACY BULK PACKAGE IS NOT FOR DIRECT INFUSION, FURTHER DILUTION IS REQUIRED BEFORE USE. -

Cefoxitin Versus Piperacillin– Tazobactam As Surgical Antibiotic Prophylaxis in Patients Undergoing Pancreatoduodenectomy: Protocol for a Randomised Controlled Trial
Open access Protocol BMJ Open: first published as 10.1136/bmjopen-2020-048398 on 4 March 2021. Downloaded from Cefoxitin versus piperacillin– tazobactam as surgical antibiotic prophylaxis in patients undergoing pancreatoduodenectomy: protocol for a randomised controlled trial Nicole M Nevarez ,1 Brian C Brajcich,2 Jason Liu,2,3 Ryan Ellis,2 Clifford Y Ko,2 Henry A Pitt,4 Michael I D'Angelica,5 Adam C Yopp1 To cite: Nevarez NM, ABSTRACT Strengths and limitations of this study Brajcich BC, Liu J, et al. Introduction Although antibiotic prophylaxis is Cefoxitin versus piperacillin– established in reducing postoperative surgical site tazobactam as surgical ► A major strength of this study is the multi- infections (SSIs), the optimal antibiotic for prophylaxis in antibiotic prophylaxis institutional, double- arm, randomised controlled in patients undergoing pancreatoduodenectomy (PD) remains unclear. The study trial design. objective is to evaluate if administration of piperacillin– pancreatoduodenectomy: ► A limitation of this study is that all perioperative care protocol for a randomised tazobactam as antibiotic prophylaxis results in decreased is at the discretion of the operating surgeon and is controlled trial. BMJ Open 30- day SSI rate compared with cefoxitin in patients not standardised. 2021;11:e048398. doi:10.1136/ undergoing elective PD. ► All data will be collected through the American bmjopen-2020-048398 Methods and analysis This study will be a multi- College of Surgeons National Surgical Quality ► Prepublication history for institution, double- arm, non- blinded randomised controlled Improvement Program, which is a strength for its this paper is available online. superiority trial. Adults ≥18 years consented to undergo PD ease of use but a limitation due to the variety of data To view these files, please visit for all indications who present to institutions participating included. -

Activity of Aztreonam in Combination with Ceftazidime-Avibactam Against
Diagnostic Microbiology and Infectious Disease 99 (2021) 115227 Contents lists available at ScienceDirect Diagnostic Microbiology and Infectious Disease journal homepage: www.elsevier.com/locate/diagmicrobio Activity of aztreonam in combination with ceftazidime–avibactam against serine- and metallo-β-lactamase–producing ☆ ☆☆ ★ Pseudomonas aeruginosa , , Michelle Lee a, Taylor Abbey a, Mark Biagi b, Eric Wenzler a,⁎ a College of Pharmacy, University of Illinois at Chicago, Chicago, IL, USA b College of Pharmacy, University of Illinois at Chicago, Rockford, IL, USA article info abstract Article history: Existing data support the combination of aztreonam and ceftazidime–avibactam against serine-β-lactamase Received 29 June 2020 (SBL)– and metallo-β-lactamase (MBL)–producing Enterobacterales, although there is a paucity of data against Received in revised form 15 September 2020 SBL- and MBL-producing Pseudomonas aeruginosa. In this study, 5 SBL- and MBL-producing P. aeruginosa (1 Accepted 19 September 2020 IMP, 4 VIM) were evaluated against aztreonam and ceftazidime–avibactam alone and in combination via broth Available online xxxx microdilution and time-kill analyses. All 5 isolates were nonsusceptible to aztreonam, aztreonam–avibactam, – – Keywords: and ceftazidime avibactam. Combining aztreonam with ceftazidime avibactam at subinhibitory concentrations Metallo-β-lactamase produced synergy and restored bactericidal activity in 4/5 (80%) isolates tested. These results suggest that the VIM combination of aztreonam and ceftazidime–avibactam may be a viable treatment option against SBL- and IMP MBL-producing P. aeruginosa. Aztreonam © 2020 Elsevier Inc. All rights reserved. Ceftazidime–avibactam Synergy 1. Introduction of SBLs and MBLs harbored by P. aeruginosa are fundamentally different than those in Enterobacterales (Alm et al., 2015; Kazmierczak et al., The rapid global dissemination of Ambler class B metallo-β- 2016; Periasamy et al., 2020; Poirel et al., 2000; Watanabe et al., lactamase (MBL) enzymes along with their increasing diversity across 1991). -

Use of Ceftaroline Fosamil in Children: Review of Current Knowledge and Its Application
Infect Dis Ther (2017) 6:57–67 DOI 10.1007/s40121-016-0144-8 REVIEW Use of Ceftaroline Fosamil in Children: Review of Current Knowledge and its Application Juwon Yim . Leah M. Molloy . Jason G. Newland Received: November 10, 2016 / Published online: December 30, 2016 Ó The Author(s) 2016. This article is published with open access at Springerlink.com ABSTRACT infections, CABP caused by penicillin- and ceftriaxone-resistant S. pneumoniae and Ceftaroline is a novel cephalosporin recently resistant Gram-positive infections that fail approved in children for treatment of acute first-line antimicrobial agents. However, bacterial skin and soft tissue infections and limited data are available on tolerability in community-acquired bacterial pneumonia neonates and infants younger than 2 months (CABP) caused by methicillin-resistant of age, and on pharmacokinetic characteristics Staphylococcus aureus, Streptococcus pneumoniae in children with chronic medical conditions and other susceptible bacteria. With a favorable and those with invasive, complicated tolerability profile and efficacy proven in infections. In this review, the microbiological pediatric patients and excellent in vitro profile of ceftaroline, its mechanism of action, activity against resistant Gram-positive and and pharmacokinetic profile will be presented. Gram-negative bacteria, ceftaroline may serve Additionally, clinical evidence for use in as a therapeutic option for polymicrobial pediatric patients and proposed place in therapy is discussed. Enhanced content To view enhanced content for this article go to http://www.medengine.com/Redeem/ 1F47F0601BB3F2DD. Keywords: Antibiotic resistance; Ceftaroline J. Yim (&) fosamil; Children; Methicillin-resistant St. John Hospital and Medical Center, Detroit, MI, Staphylococcus aureus; Streptococcus pneumoniae USA e-mail: [email protected] L. -

Global Assessment of the Antimicrobial Activity of Polymyxin B Against 54 731 Clinical Isolates of Gram-Negative Bacilli
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector ORIGINAL ARTICLE 10.1111/j.1469-0691.2005.01351.x Global assessment of the antimicrobial activity of polymyxin B against 54 731 clinical isolates of Gram-negative bacilli: report from the SENTRY antimicrobial surveillance programme (2001–2004) A. C. Gales1, R. N. Jones2,3 and H. S. Sader1,2 1Division of Infectious Diseases, Universidade Federal de Sa˜o Paulo, Sa˜o Paulo, Brazil, 2JMI Laboratories, North Liberty, IA, USA, and 3Tufts University School of Medicine, Boston, MA, USA ABSTRACT In total, 54 731 Gram-negative bacilli isolated worldwide between 2001 and 2004 from diverse sites of infection were tested for susceptibility to polymyxin B by the broth reference microdilution method, with interpretation of results according to CLSI (formerly NCCLS) guidelines. Polymyxin B showed excellent potency and spectrum against 8705 Pseudomonas aeruginosa and 2621 Acinetobacter spp. isolates (MIC50, £ ⁄ ⁄ 1mg L and MIC90,2mg L for both pathogens). Polymyxin B resistance rates were slightly higher for carbapenem-resistant P. aeruginosa (2.7%) and Acinetobacter spp. (2.8%), or multidrug-resistant (MDR) P. aeruginosa (3.3%) and Acinetobacter spp. (3.2%), when compared with the entire group (1.3% for P. aeruginosa and 2.1% for Acinetobacter spp.). Among P. aeruginosa, polymyxin B resistance rates varied from 2.9% in the Asia-Pacific region to only 1.1% in Europe, Latin America and North America, while polymyxin B resistance rates ranged from 2.7% in Europe to 1.7% in North America and Latin America £ ⁄ % among Acinetobacter spp. -

Cusumano-Et-Al-2017.Pdf
International Journal of Infectious Diseases 63 (2017) 1–6 Contents lists available at ScienceDirect International Journal of Infectious Diseases journal homepage: www.elsevier.com/locate/ijid Rapidly growing Mycobacterium infections after cosmetic surgery in medical tourists: the Bronx experience and a review of the literature a a b c b Lucas R. Cusumano , Vivy Tran , Aileen Tlamsa , Philip Chung , Robert Grossberg , b b, Gregory Weston , Uzma N. Sarwar * a Albert Einstein College of Medicine, Montefiore Medical Center, Bronx, New York, USA b Division of Infectious Diseases, Department of Medicine, Albert Einstein College of Medicine, Montefiore Medical Center, Bronx, New York, USA c Department of Pharmacy, Nebraska Medicine, Omaha, Nebraska, USA A R T I C L E I N F O A B S T R A C T Article history: Background: Medical tourism is increasingly popular for elective cosmetic surgical procedures. However, Received 10 May 2017 medical tourism has been accompanied by reports of post-surgical infections due to rapidly growing Received in revised form 22 July 2017 mycobacteria (RGM). The authors’ experience working with patients with RGM infections who have Accepted 26 July 2017 returned to the USA after traveling abroad for cosmetic surgical procedures is described here. Corresponding Editor: Eskild Petersen, Methods: Patients who developed RGM infections after undergoing cosmetic surgeries abroad and who ?Aarhus, Denmark presented at the Montefiore Medical Center (Bronx, New York, USA) between August 2015 and June 2016 were identified. A review of patient medical records was performed. Keywords: Results: Four patients who presented with culture-proven RGM infections at the sites of recent cosmetic Mycobacterium abscessus complex procedures were identified. -

Dalvance Generic Name: Dalbavancin Manufacturer1: DURATA Therapeutics Drug Class1,2,3,4: Antibiotic
Brand Name: Dalvance Generic Name: dalbavancin Manufacturer1: DURATA therapeutics Drug Class1,2,3,4: Antibiotic Uses: Labeled Uses1: Treatment of adult patients with acute bacterial skin and skin structure infections caused by susceptible isolates of the following Gram-positive microorganisms: Staphylococcus aureus (including methicillin-susceptible and methicillin-resistant strains), Streptococcus pyogenes, Streptococcus agalactiae, Streptococcus anginosus group (including S. anginosus, S. intermedius, S. constellatus) Mechanism of Action:1,2,3,4 This drug is a lipoglycopeptide which binds to the D-alanyl-D-alanine terminus of the stem pentapeptide in nascent cell wall. Through this above mechanism it prevents cross-linking and interferes with cell wall synthesis. Dalbavancin is bactericidal in vitro against Staphylococcus aureus and Streptococcus pyogenes. Pharmacokinetics1,2,3,4 Tmax End of infusion time Vd 7-13L t1/2 346 hours Clearance .0513 l/h Protein binding (albumin) 93% (primarily to albumin) Bioavailability 100% Metabolism1,2,3,4: A minor metabolite- hydroxy-dalbavancin has been observed in the urine of healthy subjects, however quantifiable plasma concentrations have not been observed. Elimination1,2,3,4: Urine (33% as unchanged drug, 12% as hydroxy metabolite) Feces (20%) Efficacy: Boucher HW, Wilcox M, Talbot GH, Puttagunta S, Das AF, Dunne MW. Once-Weekly Dalbavancin versus Daily Conventional Therapy for Skin Infection. N Engl J Med 2014; 370:2169-2179. Study Design: Double blind, double dummy, randomized controlled study Description of Study: Discover 1 and Discover 2 were international, multicenter, randomized trials conducted from 2011 through 2012 at 54 and 86 investigative sites, respectively. The studies had the same design. Patients with acute bacterial skin and skin-structure infection were stratified then randomly assigned to receive dalbavancin intravenously on days 1 and 8 or vancomycin intravenously for at least 3 days with the option to switch to oral linezolid to complete 10 to 14 days of therapy. -

Severe Sepsis and Septic Shock Antibiotic Guide
Stanford Health Issue Date: 05/2017 Stanford Antimicrobial Safety and Sustainability Program Severe Sepsis and Septic Shock Antibiotic Guide Table 1: Antibiotic selection options for healthcare associated and/or immunocompromised patients • Healthcare associated: intravenous therapy, wound care, or intravenous chemotherapy within the prior 30 days, residence in a nursing home or other long-term care facility, hospitalization in an acute care hospital for two or more days within the prior 90 days, attendance at a hospital or hemodialysis clinic within the prior 30 days • Immunocompromised: Receiving chemotherapy, known systemic cancer not in remission, ANC <500, severe cell-mediated immune deficiency Table 2: Antibiotic selection options for community acquired, immunocompetent patients Table 3: Antibiotic selection options for patients with simple sepsis, community acquired, immunocompetent patients requiring hospitalization. Risk Factors for Select Organisms P. aeruginosa MRSA Invasive Candidiasis VRE (and other resistant GNR) Community acquired: • Known colonization with MDROs • Central venous catheter • Liver transplant • Prior IV antibiotics within 90 day • Recent MRSA infection • Broad-spectrum antibiotics • Known colonization • Known colonization with MDROs • Known MRSA colonization • + 1 of the following risk factors: • Prolonged broad antibacterial • Skin & Skin Structure and/or IV access site: ♦ Parenteral nutrition therapy Hospital acquired: ♦ Purulence ♦ Dialysis • Prolonged profound • Prior IV antibiotics within 90 days ♦ Abscess -

Criteria for Use of Dalbavancin for Acute Bacterial Skin/Soft Tissue Infection (Abssti)
Criteria for Use of Dalbavancin for Acute Bacterial Skin/Soft Tissue Infection (abSSTI) 1. Patients meeting any of the following are NOT ELIGIBLE for dalbavancin therapy: a. History of hypersensitivity reaction to lipoglycopeptide antibiotics (vancomycin, televancin, dalbavancin, oritavancin). b. Patients with acute bacterial skin or skin structure infections such as superficial/simple cellulitis/erysipelas, impetiginous lesion, furuncle, or simple abscess that only requires surgical drainage for cure. c. Infection thought to be caused by gram-negative bacteria d. Infection due to an organism suspected or known to be resistant to dalbavancin or vancomycin 2. For outpatient use (i.e. ED) a. Contact infectious disease for authorization: ABX approval pager (see ON-CALL schedule) b. The following clinical criteria must be met: i. Pre-antibiotic blood cultures must be drawn. ii. Clinical condition expected to require ≥ 24 hours of IV antibiotics – must not qualify for oral antibiotic therapy. iii. Presence of cellulitis, major abscess or a wound infection associated with at least 75cm2 of erythema highly suspected or known to be caused by gram-positive bacteria. iv. The size of the infection must be clearly documented and/or outlined prior to leaving the ED, preferably with a photograph. v. Patient to be discharged to home ± home health (not to skilled nursing facility). c. Required follow up must be set up prior to leaving the ED: i. Must document patient contact info for follow up, preferably reliable cell phone number. ii. Must have follow up within 48-72H with Dr. Turnipseed (916-765-0196) or Rominski. 1. Email patient name, MRN, and phone number. -
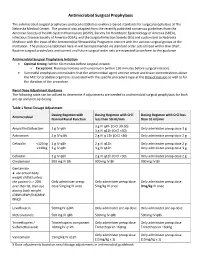
Antimicrobial Surgical Prophylaxis
Antimicrobial Surgical Prophylaxis The antimicrobial surgical prophylaxis protocol establishes evidence-based standards for surgical prophylaxis at The Nebraska Medical Center. The protocol was adapted from the recently published consensus guidelines from the American Society of Health-System Pharmacists (ASHP), Society for Healthcare Epidemiology of America (SHEA), Infectious Disease Society of America (IDSA), and the Surgical Infection Society (SIS) and customized to Nebraska Medicine with the input of the Antimicrobial Stewardship Program in concert with the various surgical groups at the institution. The protocol established here-in will be implemented via standard order sets utilized within One Chart. Routine surgical prophylaxis and current and future surgical order sets are expected to conform to this guidance. Antimicrobial Surgical Prophylaxis Initiation Optimal timing: Within 60 minutes before surgical incision o Exceptions: Fluoroquinolones and vancomycin (within 120 minutes before surgical incision) Successful prophylaxis necessitates that the antimicrobial agent achieve serum and tissue concentrations above the MIC for probable organisms associated with the specific procedure type at the time of incision as well as for the duration of the procedure. Renal Dose Adjustment Guidance The following table can be utilized to determine if adjustments are needed to antimicrobial surgical prophylaxis for both pre-op and post-op dosing. Table 1 Renal Dosage Adjustment Dosing Regimen with Dosing Regimen with CrCl Dosing Regimen with -

Dalbavancin (Dalvance®)
DalbavancinDalbavancin (Dalvance (Dalvance®)®) IV Only Use requires formal ID Consult Activity: Coverage against Staphylococcus aureus (including MSSA and MRSA), Streptococcus pyogenes, Streptococcus agalactiae (Group B Strep.) and Streptococcus anginosus group (including S. anginosus, S. intermedius, S. constellatus) No clinical data, but activity in vitro vs. Enterococcus faecalis (vancomycin-susceptible strains only), Enterococcus faecium (vancomycin-susceptible strains only), vancomycin-intermediate S. aureus (not vancomycin-resistant strains) Criteria for Use: Treatment of adult patients with acute bacterial skin and skin structure infections (ABSSSI) caused by susceptible gram-positive isolates Unable to use vancomycin (due to intolerance, MIC >2mg/L, or infection unresponsive to vancomycin despite therapeutic concentrations) Unable to use other agents (refer to empiric therapy for ABSSSI) Unacceptable Uses: Infections due to vancomycin-resistant enterococci Contraindicated in patients with known hypersensitivity to dalbavancin. Due to the possibility of cross-reactivity to glycopeptide, avoid in patients with previous glycopeptide hypersensitivity due to long half-life Dosing in Adults: Standard dose: Administration should be over 30 minutes 1 Dose Regimen: 1500mg IV once 2 Dose Regimen: 1000mg IV once, then 500mg IV on day 8 Renal dose adjustment: 1 Dose Regimen CrCl <30 mL/min 1125 mg IV 2 Dose Regimen CrCl <30 mL/min: 750mg IV once, then 325mg IV day 8 If receiving regularly scheduled hemodialysis: No dosage adjustment No hepatic dose adjustment anticipated Monitoring: Baseline BUN/Scr, AST/ALT/bili, CBC w/ diff, infusion-related reactions Considerations for Use: In clinical trials, 6 (0.9%) patients in the dalbavancin arm had ALT elevations greater than 5x ULN including 3 with ALT >10x ULN.