Guidelines for Treatment of Intra-Abdominal Infections in Adults
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Print This Article
International Surgery Journal Lew D et al. Int Surg J. 2021 May;8(5):1575-1578 http://www.ijsurgery.com pISSN 2349-3305 | eISSN 2349-2902 DOI: https://dx.doi.org/10.18203/2349-2902.isj20211831 Case Report Acute gangrenous appendicitis and acute gangrenous cholecystitis in a pregnant patient, a difficult diagnosis: a case report David Lew, Jane Tian*, Martine A. Louis, Darshak Shah Department of Surgery, Flushing Hospital Medical Center, Flushing, New York, USA Received: 26 February 2021 Accepted: 02 April 2021 *Correspondence: Dr. Jane Tian, E-mail: [email protected] Copyright: © the author(s), publisher and licensee Medip Academy. This is an open-access article distributed under the terms of the Creative Commons Attribution Non-Commercial License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT Abdominal pain is a common complaint in pregnancy, especially given the physiological and anatomical changes that occur as the pregnancy progresses. The diagnosis and treatment of common surgical pathologies can therefore be difficult and limited by the special considerations for the fetus. While uncommon in the general population, concurrent or subsequent disease processes should be considered in the pregnant patient. We present the case of a 36 year old, 13 weeks pregnant female who presented with both acute appendicitis and acute cholecystitis. Keywords: Appendicitis, Cholecystitis, Pregnancy, Pregnant INTRODUCTION population is rare.5 Here we report a case of concurrent appendicitis and cholecystitis in a pregnant woman. General surgeons are often called to evaluate patients with abdominal pain. The differential diagnosis list must CASE REPORT be expanded in pregnant woman and the approach to diagnosing and treating certain diseases must also be A 36 year old, 13 weeks pregnant female (G2P1001) adjusted to prevent harm to the fetus. -

Ceftazidime for Injection) PHARMACY BULK PACKAGE – NOT for DIRECT INFUSION
PRESCRIBING INFORMATION FORTAZ® (ceftazidime for injection) PHARMACY BULK PACKAGE – NOT FOR DIRECT INFUSION To reduce the development of drug-resistant bacteria and maintain the effectiveness of FORTAZ and other antibacterial drugs, FORTAZ should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. DESCRIPTION Ceftazidime is a semisynthetic, broad-spectrum, beta-lactam antibacterial drug for parenteral administration. It is the pentahydrate of pyridinium, 1-[[7-[[(2-amino-4 thiazolyl)[(1-carboxy-1-methylethoxy)imino]acetyl]amino]-2-carboxy-8-oxo-5-thia-1 azabicyclo[4.2.0]oct-2-en-3-yl]methyl]-, hydroxide, inner salt, [6R-[6α,7β(Z)]]. It has the following structure: The molecular formula is C22H32N6O12S2, representing a molecular weight of 636.6. FORTAZ is a sterile, dry-powdered mixture of ceftazidime pentahydrate and sodium carbonate. The sodium carbonate at a concentration of 118 mg/g of ceftazidime activity has been admixed to facilitate dissolution. The total sodium content of the mixture is approximately 54 mg (2.3 mEq)/g of ceftazidime activity. The Pharmacy Bulk Package vial contains 709 mg of sodium carbonate. The sodium content is approximately 54 mg (2.3mEq) per gram of ceftazidime. FORTAZ in sterile crystalline form is supplied in Pharmacy Bulk Packages equivalent to 6g of anhydrous ceftazidime. The Pharmacy Bulk Package bottle is a container of sterile preparation for parenteral use that contains many single doses. The contents are intended for use in a pharmacy admixture program and are restricted to the preparation of admixtures for intravenous use. THE PHARMACY BULK PACKAGE IS NOT FOR DIRECT INFUSION, FURTHER DILUTION IS REQUIRED BEFORE USE. -

Use of Ceftaroline Fosamil in Children: Review of Current Knowledge and Its Application
Infect Dis Ther (2017) 6:57–67 DOI 10.1007/s40121-016-0144-8 REVIEW Use of Ceftaroline Fosamil in Children: Review of Current Knowledge and its Application Juwon Yim . Leah M. Molloy . Jason G. Newland Received: November 10, 2016 / Published online: December 30, 2016 Ó The Author(s) 2016. This article is published with open access at Springerlink.com ABSTRACT infections, CABP caused by penicillin- and ceftriaxone-resistant S. pneumoniae and Ceftaroline is a novel cephalosporin recently resistant Gram-positive infections that fail approved in children for treatment of acute first-line antimicrobial agents. However, bacterial skin and soft tissue infections and limited data are available on tolerability in community-acquired bacterial pneumonia neonates and infants younger than 2 months (CABP) caused by methicillin-resistant of age, and on pharmacokinetic characteristics Staphylococcus aureus, Streptococcus pneumoniae in children with chronic medical conditions and other susceptible bacteria. With a favorable and those with invasive, complicated tolerability profile and efficacy proven in infections. In this review, the microbiological pediatric patients and excellent in vitro profile of ceftaroline, its mechanism of action, activity against resistant Gram-positive and and pharmacokinetic profile will be presented. Gram-negative bacteria, ceftaroline may serve Additionally, clinical evidence for use in as a therapeutic option for polymicrobial pediatric patients and proposed place in therapy is discussed. Enhanced content To view enhanced content for this article go to http://www.medengine.com/Redeem/ 1F47F0601BB3F2DD. Keywords: Antibiotic resistance; Ceftaroline J. Yim (&) fosamil; Children; Methicillin-resistant St. John Hospital and Medical Center, Detroit, MI, Staphylococcus aureus; Streptococcus pneumoniae USA e-mail: [email protected] L. -

Cefalexin in the WHO Essential Medicines List for Children Reject
Reviewer No. 1: checklist for application of: Cefalexin In the WHO Essential Medicines List for Children (1) Have all important studies that you are aware of been included? Yes No 9 (2) Is there adequate evidence of efficacy for the proposed use? Yes 9 No (3) Is there evidence of efficacy in diverse settings and/or populations? Yes 9 No (4) Are there adverse effects of concern? Yes 9 No (5) Are there special requirements or training needed for safe/effective use? Yes No 9 (6) Is this product needed to meet the majority health needs of the population? Yes No 9 (7) Is the proposed dosage form registered by a stringent regulatory authority? Yes 9 No (8) What action do you propose for the Committee to take? Reject the application for inclusion of the following presentations of cefalexin: • Tablets/ capsules 250mg • Oral suspensions 125mg/5ml and 125mg/ml (9) Additional comment, if any. In order to identify any additional literature, the following broad and sensitive search was conducted using the PubMed Clinical Query application: (cephalexin OR cefalexin AND - 1 - pediatr*) AND ((clinical[Title/Abstract] AND trial[Title/Abstract]) OR clinical trials[MeSH Terms] OR clinical trial[Publication Type] OR random*[Title/Abstract] OR random allocation[MeSH Terms] OR therapeutic use[MeSH Subheading]) Only one small additional study was identified, which looked at the provision of prophylactic antibiotics in patients presenting to an urban children's hospital with trauma to the distal fingertip, requiring repair.1 In a prospective randomised control trial, 146 patients were enrolled, of which 69 were randomised to the no-antibiotic group, and 66 were randomised to the antibiotic (cefalexin) group. -

General Items
Essential Medicines List (EML) 2019 Application for the inclusion of imipenem/cilastatin, meropenem and amoxicillin/clavulanic acid in the WHO Model List of Essential Medicines, as reserve second-line drugs for the treatment of multidrug-resistant tuberculosis (complementary lists of anti-tuberculosis drugs for use in adults and children) General items 1. Summary statement of the proposal for inclusion, change or deletion This application concerns the updating of the forthcoming WHO Model List of Essential Medicines (EML) and WHO Model List of Essential Medicines for Children (EMLc) to include the following medicines: 1) Imipenem/cilastatin (Imp-Cln) to the main list but NOT the children’s list (it is already mentioned on both lists as an option in section 6.2.1 Beta Lactam medicines) 2) Meropenem (Mpm) to both the main and the children’s lists (it is already on the list as treatment for meningitis in section 6.2.1 Beta Lactam medicines) 3) Clavulanic acid to both the main and the children’s lists (it is already listed as amoxicillin/clavulanic acid (Amx-Clv), the only commercially available preparation of clavulanic acid, in section 6.2.1 Beta Lactam medicines) This application makes reference to amendments recommended in particular to section 6.2.4 Antituberculosis medicines in the latest editions of both the main EML (20th list) and the EMLc (6th list) released in 2017 (1),(2). On the basis of the most recent Guideline Development Group advising WHO on the revision of its guidelines for the treatment of multidrug- or rifampicin-resistant (MDR/RR-TB)(3), the applicant considers that the three agents concerned be viewed as essential medicines for these forms of TB in countries. -

12. What's Really New in Antibiotic Therapy Print
What’s really new in antibiotic therapy? Martin J. Hug Freiburg University Medical Center EAHP Academy Seminars 20-21 September 2019 Newsweek, May 24-31 2019 Disclosures There are no conflicts of interest to declare EAHP Academy Seminars 20-21 September 2019 Antiinfectives and Resistance EAHP Academy Seminars 20-21 September 2019 Resistance of Klebsiella pneumoniae to Pip.-Taz. olates) EAHP Academy Seminars 20-21 September 2019 https://resistancemap.cddep.org/AntibioticResistance.php Multiresistant Pseudomonas Aeruginosa Combined resistance against at least three different types of antibiotics, 2017 EAHP Academy Seminars 20-21 September 2019 https://atlas.ecdc.europa.eu/public/index.aspx Distribution of ESBL producing Enterobacteriaceae EAHP Academy Seminars 20-21 September 2019 Rossolini GM. Global threat of Gram-negative antimicrobial resistance. 27th ECCMID, Vienna, 2017, IS07 Priority Pathogens Defined by the World Health Organisation Critical Priority High Priority Medium Priority Acinetobacter baumanii Enterococcus faecium Streptococcus pneumoniae carbapenem-resistant vancomycin-resistant penicillin-non-susceptible Pseudomonas aeruginosa Helicobacter pylori Haemophilus influenzae carbapenem-resistant clarithromycin-resistant ampicillin-resistant Enterobacteriaceae Salmonella species Shigella species carbapenem-resistant fluoroquinolone-resistant fluoroquinolone-resistant Staphylococcus aureus vancomycin or methicillin -resistant Campylobacter species fluoroquinolone-resistant Neisseria gonorrhoae 3rd gen. cephalosporin-resistant -

Severe Sepsis and Septic Shock Antibiotic Guide
Stanford Health Issue Date: 05/2017 Stanford Antimicrobial Safety and Sustainability Program Severe Sepsis and Septic Shock Antibiotic Guide Table 1: Antibiotic selection options for healthcare associated and/or immunocompromised patients • Healthcare associated: intravenous therapy, wound care, or intravenous chemotherapy within the prior 30 days, residence in a nursing home or other long-term care facility, hospitalization in an acute care hospital for two or more days within the prior 90 days, attendance at a hospital or hemodialysis clinic within the prior 30 days • Immunocompromised: Receiving chemotherapy, known systemic cancer not in remission, ANC <500, severe cell-mediated immune deficiency Table 2: Antibiotic selection options for community acquired, immunocompetent patients Table 3: Antibiotic selection options for patients with simple sepsis, community acquired, immunocompetent patients requiring hospitalization. Risk Factors for Select Organisms P. aeruginosa MRSA Invasive Candidiasis VRE (and other resistant GNR) Community acquired: • Known colonization with MDROs • Central venous catheter • Liver transplant • Prior IV antibiotics within 90 day • Recent MRSA infection • Broad-spectrum antibiotics • Known colonization • Known colonization with MDROs • Known MRSA colonization • + 1 of the following risk factors: • Prolonged broad antibacterial • Skin & Skin Structure and/or IV access site: ♦ Parenteral nutrition therapy Hospital acquired: ♦ Purulence ♦ Dialysis • Prolonged profound • Prior IV antibiotics within 90 days ♦ Abscess -

The Antibacterial Activity of Cefpodoxime and the Novel Β
Session-198 b SATURDAY-279 The Antibacterial Activity of Cefpodoxime and the Novel -lactamase Inhibitor ETX1317 Against Recent Clinical Isolates of b-lactamase-producing Enterobacteriaceae Entasis Therapeutics S. McLeod1, M. Hackel2, R. Badal2, A. Shapiro1, J. Mueller1, R. Tommasi1, and A. Miller1 1-781-810-0120 1Entasis Therapeutics, Waltham, MA and 2IHMA, International Health Management Associates, Schaumburg, IL USA www.entasistx.com Abstract In vitro activity of ETX1317 vs. comparator BLIs Cumulative activity vs. 911 ESBL-enriched Enterobacteriaceae from 2013-2015 Activity vs. 35 sequenced KPC+ and/or MBL+ Enterobacteriaceae MIC (mg/L) Background β-lactamase inhibition (IC50, µM) Strain ID Species encoded bla genes IPM CAZ-AVI CPD-ETX1317 ETX1317 ETX1317 is a novel, diazabicyclooctenone inhibitor of serine β-lactamases that Number (cumulative %) of isolates inhibited at MIC (mg/L) Class A Class C Class D Drug %S* ARC3528*¥ E. cloacae AmpC [∆308-313(SKVALA)] 0.25 32 0.125 4 restores β-lactam activity against multidrug-resistant Enterobacteriaceae. ETX0282, < 0.03 0.06 0.12 0.25 0.5 1 2 4 8 16 32 > 32 ARC4165*¥ C. freundii CMY-2; AmpC [N366Y]; TEM-1; SHV-5 2 32 ≤0.03 4 the oral prodrug of ETX1317, is currently under investigation in combination with BLI CTX-M-15 SHV-5 KPC-2 TEM-1 AmpC* P99 OXA-24/40 OXA-48 0 5 6 14 19 34 20 27 17 18 18 733 3 CPD 10.8 ARC6479* K. oxytoca NDM-1 16 >32 32 32 cefpodoxime proxetil (CPDP), which is hydrolyzed in vivo to release cefpodoxime 0% 0.5% 1% 3% 5% 9% 11% 14% 16% 18% 20% 100% (CPD). -
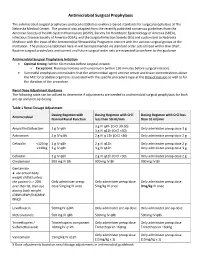
Antimicrobial Surgical Prophylaxis
Antimicrobial Surgical Prophylaxis The antimicrobial surgical prophylaxis protocol establishes evidence-based standards for surgical prophylaxis at The Nebraska Medical Center. The protocol was adapted from the recently published consensus guidelines from the American Society of Health-System Pharmacists (ASHP), Society for Healthcare Epidemiology of America (SHEA), Infectious Disease Society of America (IDSA), and the Surgical Infection Society (SIS) and customized to Nebraska Medicine with the input of the Antimicrobial Stewardship Program in concert with the various surgical groups at the institution. The protocol established here-in will be implemented via standard order sets utilized within One Chart. Routine surgical prophylaxis and current and future surgical order sets are expected to conform to this guidance. Antimicrobial Surgical Prophylaxis Initiation Optimal timing: Within 60 minutes before surgical incision o Exceptions: Fluoroquinolones and vancomycin (within 120 minutes before surgical incision) Successful prophylaxis necessitates that the antimicrobial agent achieve serum and tissue concentrations above the MIC for probable organisms associated with the specific procedure type at the time of incision as well as for the duration of the procedure. Renal Dose Adjustment Guidance The following table can be utilized to determine if adjustments are needed to antimicrobial surgical prophylaxis for both pre-op and post-op dosing. Table 1 Renal Dosage Adjustment Dosing Regimen with Dosing Regimen with CrCl Dosing Regimen with -

In Vitro Susceptibilities of Escherichia Coli and Klebsiella Spp. To
Jpn. J. Infect. Dis., 60, 227-229, 2007 Short Communication In Vitro Susceptibilities of Escherichia coli and Klebsiella Spp. to Ampicillin-Sulbactam and Amoxicillin-Clavulanic Acid Birgul Kacmaz* and Nedim Sultan1 Department of Central Microbiology and 1Department of Microbiology, Faculty of Medicine, Gazi University, Ankara, Turkey (Received January 30, 2007. Accepted April 13, 2007) SUMMARY: Ampicillin-sulbactam (A/S) and amoxicillin-clavulanic acid (AUG) are thought to be equally efficacious clinically against the Enterobacteriaceae family. In this study, the in vitro activities of the A/S and AUG were evaluated and compared against Escherichia coli and Klebsiella spp. Antimicrobial susceptibility tests were performed by standard agar dilution and disc diffusion techniques according to the Clinical and Laboratory Standards Institute (CLSI). During the study period, 973 strains were isolated. Of the 973 bacteria isolated, 823 were E. coli and 150 Klebsiella spp. More organisms were found to be susceptible to AUG than A/S, regardless of the susceptibility testing methodology. The agar dilution results of the isolates that were found to be sensitive or resistant were also compatible with the disc diffusion results. However, some differences were seen in the agar dilution results of some isolates that were found to be intermediately resistant with disc diffusion. In E. coli isolates, 17 of the 76 AUG intermediately resistant isolates (by disc diffusion), and 17 of the 63 A/S intermediately resistant isolates (by disc diffusion) showed different resistant patterns by agar dilution. When the CLSI breakpoint criteria are applied it should be considered that AUG and A/S sensitivity in E. coli and Klebsiella spp. -

Antimicrobial Stewardship Guidance
Antimicrobial Stewardship Guidance Federal Bureau of Prisons Clinical Practice Guidelines March 2013 Clinical guidelines are made available to the public for informational purposes only. The Federal Bureau of Prisons (BOP) does not warrant these guidelines for any other purpose, and assumes no responsibility for any injury or damage resulting from the reliance thereof. Proper medical practice necessitates that all cases are evaluated on an individual basis and that treatment decisions are patient-specific. Consult the BOP Clinical Practice Guidelines Web page to determine the date of the most recent update to this document: http://www.bop.gov/news/medresources.jsp Federal Bureau of Prisons Antimicrobial Stewardship Guidance Clinical Practice Guidelines March 2013 Table of Contents 1. Purpose ............................................................................................................................................. 3 2. Introduction ...................................................................................................................................... 3 3. Antimicrobial Stewardship in the BOP............................................................................................ 4 4. General Guidance for Diagnosis and Identifying Infection ............................................................. 5 Diagnosis of Specific Infections ........................................................................................................ 6 Upper Respiratory Infections (not otherwise specified) .............................................................................. -

Burkholderia Pseudomallei Clinical Isolates Are Highly Susceptible in Vitro To
bioRxiv preprint doi: https://doi.org/10.1101/2020.03.26.009134; this version posted March 27, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. 1 Burkholderia pseudomallei clinical isolates are highly susceptible in vitro to 2 cefiderocol, a novel siderophore cephalosporin 3 Delaney Burnard1,6, Gemma Robertson1,2,4, Andrew Henderson1,5, Caitlin Falconer1, Michelle 4 Bauer-Leo1, Kyra Cottrell1, Ian Gassiep2,3, Robert Norton8,9, David L. Paterson1,3, Patrick N. 5 A. Harris1,2* 6 7 1University of Queensland Centre for Clinical Research, Herston, Queensland, Australia 8 2Pathology Queensland, Queensland Health, Herston, Queensland, Australia 9 3Royal Brisbane and Women’s Hospital, Queensland Health, Herston, Queensland, Australia 10 4Forensic and Scientific Services, Queensland Health, Coopers Plains, Queensland, Australia 11 5Princess Alexandra Hospital, Queensland Health, Woolloongabba, Queensland, Australia 12 6Genecology Research Centre, University of the Sunshine Coast, Sippy Downs, Queensland, Australia 13 8Townsville Hospital and Health Service, Townsville, Queensland, Australia 14 9School of Medicine, James cook university, Townsville, Queensland, Australia 15 16 *Corresponding author: [email protected] 17 University of Queensland, Faculty of Medicine, UQ Centre for Clinical Research, Royal Brisbane & Women’s 18 Hospital, Herston, QLD, 4029, Australia 19 20 Running title: B. pseudomallei in vitro susceptibility to cefiderocol 21 Keywords: melioidosis, Burkholderia pseudomallei, cefiderocol, antimicrobial resistance, 22 AMR, minimum inhibitory concentration, MIC 1 bioRxiv preprint doi: https://doi.org/10.1101/2020.03.26.009134; this version posted March 27, 2020.