Cefalexin in the WHO Essential Medicines List for Children Reject
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Medical Review(S) Clinical Review
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 200327 MEDICAL REVIEW(S) CLINICAL REVIEW Application Type NDA Application Number(s) 200327 Priority or Standard Standard Submit Date(s) December 29, 2009 Received Date(s) December 30, 2009 PDUFA Goal Date October 30, 2010 Division / Office Division of Anti-Infective and Ophthalmology Products Office of Antimicrobial Products Reviewer Name(s) Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD Review Completion October 29, 2010 Date Established Name Ceftaroline fosamil for injection (Proposed) Trade Name Teflaro Therapeutic Class Cephalosporin; ß-lactams Applicant Cerexa, Inc. Forest Laboratories, Inc. Formulation(s) 400 mg/vial and 600 mg/vial Intravenous Dosing Regimen 600 mg every 12 hours by IV infusion Indication(s) Acute Bacterial Skin and Skin Structure Infection (ABSSSI); Community-acquired Bacterial Pneumonia (CABP) Intended Population(s) Adults ≥ 18 years of age Template Version: March 6, 2009 Reference ID: 2857265 Clinical Review Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD NDA 200327: Teflaro (ceftaroline fosamil) Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT ......................................... 9 1.1 Recommendation on Regulatory Action ........................................................... 10 1.2 Risk Benefit Assessment.................................................................................. 10 1.3 Recommendations for Postmarketing Risk Evaluation and Mitigation Strategies ........................................................................................................................ -

Use of Ceftaroline Fosamil in Children: Review of Current Knowledge and Its Application
Infect Dis Ther (2017) 6:57–67 DOI 10.1007/s40121-016-0144-8 REVIEW Use of Ceftaroline Fosamil in Children: Review of Current Knowledge and its Application Juwon Yim . Leah M. Molloy . Jason G. Newland Received: November 10, 2016 / Published online: December 30, 2016 Ó The Author(s) 2016. This article is published with open access at Springerlink.com ABSTRACT infections, CABP caused by penicillin- and ceftriaxone-resistant S. pneumoniae and Ceftaroline is a novel cephalosporin recently resistant Gram-positive infections that fail approved in children for treatment of acute first-line antimicrobial agents. However, bacterial skin and soft tissue infections and limited data are available on tolerability in community-acquired bacterial pneumonia neonates and infants younger than 2 months (CABP) caused by methicillin-resistant of age, and on pharmacokinetic characteristics Staphylococcus aureus, Streptococcus pneumoniae in children with chronic medical conditions and other susceptible bacteria. With a favorable and those with invasive, complicated tolerability profile and efficacy proven in infections. In this review, the microbiological pediatric patients and excellent in vitro profile of ceftaroline, its mechanism of action, activity against resistant Gram-positive and and pharmacokinetic profile will be presented. Gram-negative bacteria, ceftaroline may serve Additionally, clinical evidence for use in as a therapeutic option for polymicrobial pediatric patients and proposed place in therapy is discussed. Enhanced content To view enhanced content for this article go to http://www.medengine.com/Redeem/ 1F47F0601BB3F2DD. Keywords: Antibiotic resistance; Ceftaroline J. Yim (&) fosamil; Children; Methicillin-resistant St. John Hospital and Medical Center, Detroit, MI, Staphylococcus aureus; Streptococcus pneumoniae USA e-mail: [email protected] L. -

Empiric Antimicrobial Therapy for Diabetic Foot Infection
Empiric Antimicrobial Therapy for Diabetic Foot Infection (NB Provincial Health Authorities Anti-Infective Stewardship Committee, September 2019) Infection Severity Preferred Empiric Regimens Alternative Regimens Comments Mild Wound less than 4 weeks duration:d Wound less than 4 weeks duration:e • Outpatient management • Cellulitis less than 2 cm and • cephalexin 500 – 1000 mg PO q6h*,a OR • clindamycin 300 – 450 mg PO q6h (only if recommended ,a • cefadroxil 500 – 1000 mg PO q12h* severe delayed reaction to a beta-lactam) without involvement of deeper • Tailor regimen based on culture tissues and susceptibility results and True immediate allergy to a beta-lactam at MRSA Suspected: • Non-limb threatening patient response risk of cross reactivity with cephalexin or • doxycycline 200 mg PO for 1 dose then • No signs of sepsis cefadroxil: 100 mg PO q12h OR • cefuroxime 500 mg PO q8–12h*,b • sulfamethoxazole+trimethoprim 800+160 mg to 1600+320 mg PO q12h*,f Wound greater than 4 weeks duration:d Wound greater than 4 weeks duratione • amoxicillin+clavulanate 875/125 mg PO and MRSA suspected: q12h*,c OR • doxycycline 200 mg PO for 1 dose then • cefuroxime 500 mg PO q8–12h*,b AND 100 mg PO q12h AND metroNIDAZOLE metroNIDAZOLE 500 mg PO q12h 500 mg PO q12h OR • sulfamethoxazole+trimethoprim 800+160 mg to 1600/320 mg PO q12h*,f AND metroNIDAZOLE 500 mg PO q12h Moderate Wound less than 4 weeks duration:d Wound less than 4 weeks duration:e • Initial management with • Cellulitis greater than 2 cm or • ceFAZolin 2 g IV q8h*,b OR • levoFLOXacin 750 -

Cefixime (Suprax) Division of Disease Control What Do I Need to Know?
Division of Disease Control What Do I Need To Know? Cefixime (Suprax) About your medicine: Cefixime is used to treat bacterial infections in many different parts of the body. This medicine will not work for colds, flu, or other viral infections. It may be used to other problems as determined by your healthcare provider. These instructions will help you receive the optimum benefits from Cefixime. Before taking this medication: Tell your doctor, nurse or healthcare provider if you… . Have any allergies to medications (especially cephalosporins and penicillin) . Take any other medications . Have medical condition (especially kidney or eye problems) . Are pregnant, breast-feeding or trying to become pregnant . History of convulsions Possible side effects of this medicine: If these symptoms continue or become severe, call your health care provider… . Nausea, vomiting, diarrhea . Gas . Indigestion . Dyspepsia (upset stomach) . Abdominal pain . Muscle cramps, stiffness, spasms . If your symptoms do not improve within a few days, notify your healthcare provider How to take this medicine: . This is the only dose of this medicine you will receive . Take this medication with a full glass (8 ounces) of water, preferably after a meal . DO NOT take with milk or other dairy products, or any products containing magnesium or calcium (such as antacids), iron or aluminum within 2 hours of the time you take this medicine . Drink several additional glasses of water today Warning: Call a healthcare professional immediately if you develop any of the following: . Difficulty breathing . Hives/skin rash Sexual Behavior: Abstain from any type of sexual activity for 7 days after treatment of self and partner For more information, contact the NDDoH STD Program at 701.328.2378 or toll-free 800.472.2180 Page 1 of 1 Last Updated: 4/3/2012 . -

The Antibacterial Activity of Cefpodoxime and the Novel Β
Session-198 b SATURDAY-279 The Antibacterial Activity of Cefpodoxime and the Novel -lactamase Inhibitor ETX1317 Against Recent Clinical Isolates of b-lactamase-producing Enterobacteriaceae Entasis Therapeutics S. McLeod1, M. Hackel2, R. Badal2, A. Shapiro1, J. Mueller1, R. Tommasi1, and A. Miller1 1-781-810-0120 1Entasis Therapeutics, Waltham, MA and 2IHMA, International Health Management Associates, Schaumburg, IL USA www.entasistx.com Abstract In vitro activity of ETX1317 vs. comparator BLIs Cumulative activity vs. 911 ESBL-enriched Enterobacteriaceae from 2013-2015 Activity vs. 35 sequenced KPC+ and/or MBL+ Enterobacteriaceae MIC (mg/L) Background β-lactamase inhibition (IC50, µM) Strain ID Species encoded bla genes IPM CAZ-AVI CPD-ETX1317 ETX1317 ETX1317 is a novel, diazabicyclooctenone inhibitor of serine β-lactamases that Number (cumulative %) of isolates inhibited at MIC (mg/L) Class A Class C Class D Drug %S* ARC3528*¥ E. cloacae AmpC [∆308-313(SKVALA)] 0.25 32 0.125 4 restores β-lactam activity against multidrug-resistant Enterobacteriaceae. ETX0282, < 0.03 0.06 0.12 0.25 0.5 1 2 4 8 16 32 > 32 ARC4165*¥ C. freundii CMY-2; AmpC [N366Y]; TEM-1; SHV-5 2 32 ≤0.03 4 the oral prodrug of ETX1317, is currently under investigation in combination with BLI CTX-M-15 SHV-5 KPC-2 TEM-1 AmpC* P99 OXA-24/40 OXA-48 0 5 6 14 19 34 20 27 17 18 18 733 3 CPD 10.8 ARC6479* K. oxytoca NDM-1 16 >32 32 32 cefpodoxime proxetil (CPDP), which is hydrolyzed in vivo to release cefpodoxime 0% 0.5% 1% 3% 5% 9% 11% 14% 16% 18% 20% 100% (CPD). -

'Cephalosporin Allergy' Label Is Misleading
VOLUME 41 : NUMBER 2 : APRIL 2018 ARTICLE ‘Cephalosporin allergy’ label is misleading Carlo L Yuson SUMMARY Immunology registrar1 Constance H Katelaris Penicillins and cephalosporins can cause a similar spectrum of allergic reactions at a similar rate. Immunologist2 Cross-reactive allergy between penicillins and cephalosporins is rare, as is cross-reaction within William B Smith 1 the cephalosporin group. Patients should therefore not be labelled ‘cephalosporin-allergic’. Immunologist Cross-reactive allergy may occur between cephalosporins (and penicillins) which share similar 1 Clinical Immunology and side chains. Allergy Royal Adelaide Hospital Generally, a history of a penicillin allergy should not rule out the use of cephalosporins, and a 2 Immunology and Allergy history of a specific cephalosporin allergy should not rule out the use of other cephalosporins. Unit Campbelltown Hospital Specialist advice or further investigations may be required when the index reaction was New South Wales anaphylaxis or a severe cutaneous adverse reaction, or when the antibiotics in question share common side chains. Keywords When recording a drug allergy in the patient’s records, it is important to identify the specific drug cephalosporin allergy, hypersensitivity, penicillin suspected (or confirmed), along with the date and nature of the adverse reaction. Records need allergy to be updated after a negative drug challenge. Aust Prescr 2018;41:37–41 as Stevens-Johnson syndrome, toxic epidermal Introduction https://doi.org/10.18773/ necrolysis or acute generalised exanthematous To label an individual with a ‘cephalosporin allergy’ austprescr.2018.008 pustulosis or organ hypersensitivity). is misleading. Given the structural diversity of the cephalosporin family, hypersensitivity is seldom a Structural chemistry and allergy Corrected 3 December 2018 class effect but is much more likely to relate to the Immunological reactivity to small molecules such This is the corrected individual drug. -

Medicines to Treat Bacterial Infections
Government of Western Australia North Metropolitan Health Service Women and Newborn Health Service Medicines to treat bacterial infections This brochure contains some information on Important: Antibiotic resistance can the medicines you may have been prescribed affect us all. to treat a bacterial infection either in hospital or on discharge. Please talk to your doctor or Help limit the development of antibiotic pharmacist if you would like more information resistance by using antibiotics correctly. on a specific antibiotic. Make sure you: Take antibiotics exactly as prescribed. What is an antibiotic? • Antibiotics are medicines used to treat • Follow instructions on how many times a infections caused by bacteria. They are not day and for how long to take them. effective against viral infections such as the • Do not stop treatment early, even if you common cold and the ‘flu’. feel better. Medicine Other information Amoxicillin May be taken with or without food. Amoxicillin/ Take with the first bite of a meal. clavulanic acid Cefalexin May be taken with or without food. Take on an empty stomach with a glass of water, 1 hour before or 2 hours after food. Ciprofloxacin Do not take dairy products, antacids, iron, zinc or calcium within 2 hours of the dose. Clindamycin Take with a full glass of water. May be taken with or without food. Take with food or milk. Remain upright for an hour after dose to prevent damage to the Doxycycline lining of your throat. Do not take dairy products, antacids, iron, zinc or calcium within 2 hours of the dose. Take on an empty stomach, 1 hour before or 2 hours after food. -

Clavulanic Acid, and Cefaclor Against Experimental Streptococcus Pneumoniae Respiratory Infections in Mice JOHN GISBY,* BARBARA J
ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, May 1991, p. 831-836 Vol. 35, No. 5 0066-4804/91/050831-06$02.00/0 Copyright © 1991, American Society for Microbiology Comparative Efficacies of Ciprofloxacin, Amoxicillin, Amoxicillin- Clavulanic Acid, and Cefaclor against Experimental Streptococcus pneumoniae Respiratory Infections in Mice JOHN GISBY,* BARBARA J. WIGHTMAN, AND ANGELA S. BEALE SmithKline Beecham Pharmaceuticals, Brockham Park, Betchworth, Surrey RH3 7AJ, England Received 24 October 1990/Accepted 13 February 1991 Experimental respiratory infections were established in mice by intranasal inoculation of Streptococcus pneumoniae. Inoculation of 107 CFU of either S. pneumoniae 1629 or S. pneumoniae 7 produced a fatal pneumonia in nontreated mice 2 to 3 days after infection. Oral therapy was commenced 1 h after infection and was continued three times a day for 2 days. The doses used in mice produced peak concentrations in serum and lung tissue similar to those measured in humans. Ciprofloxacin failed to eliminate either strain of pneumo- coccus from mouse lungs at any of the doses tested (40, 80, or 160 mg/kg of body weight) by the end of therapy (33 h). Mice that received ciprofloxacin at 160 mg/kg were clear of S. pneumoniae 7 5 days later, whereas persistence and regrowth of S. pneumoniae 1629 resulted in the death of 20% of animals treated with ciprofloxacin. Therapy with cefaclor (20 mg/kg) produced an effect similar to that of ciprofloxacin. In contrast, amoxicillin (10 and 20 mg/kg) and amoxicillin-clavulanic acid (10/5 and 20/10 mg/kg) were significantly (P < 0.05) more effective in eliminating both strains of S. -

Antimicrobial Stewardship Guidance
Antimicrobial Stewardship Guidance Federal Bureau of Prisons Clinical Practice Guidelines March 2013 Clinical guidelines are made available to the public for informational purposes only. The Federal Bureau of Prisons (BOP) does not warrant these guidelines for any other purpose, and assumes no responsibility for any injury or damage resulting from the reliance thereof. Proper medical practice necessitates that all cases are evaluated on an individual basis and that treatment decisions are patient-specific. Consult the BOP Clinical Practice Guidelines Web page to determine the date of the most recent update to this document: http://www.bop.gov/news/medresources.jsp Federal Bureau of Prisons Antimicrobial Stewardship Guidance Clinical Practice Guidelines March 2013 Table of Contents 1. Purpose ............................................................................................................................................. 3 2. Introduction ...................................................................................................................................... 3 3. Antimicrobial Stewardship in the BOP............................................................................................ 4 4. General Guidance for Diagnosis and Identifying Infection ............................................................. 5 Diagnosis of Specific Infections ........................................................................................................ 6 Upper Respiratory Infections (not otherwise specified) .............................................................................. -

Sexually Transmitted Diseases Treatment Options
Sexually transmitted disease (STD) treatment options PREFERRED & ALTERNATIVE OPTIONS Many clinical partners are operating in a limited capacity during the COVID-19 pandemic. Below are preferred (in clinic or other location where injections can be given) and alternative (when only oral medicines are available 1) treatments for STDs. Syndrome Preferred Treatments Alternative Treatments Follow-up Male urethritis syndrome Ceftriaxone 250mg intramuscular (IM) x 1 PLUS Men who have sex with men (MSM) and transgender women2: Patients should be counseled to azithromycin 1g PO x 1 Cefixime 800 mg PO x 1 PLUS doxycycline 100 mg PO BID x 7 days be tested for STDs once clinical Presumptively treating: care is resumed in the local If azithromycin is not available: doxycycline 100 Men who have sex with women only: gonorrhea clinics. Clients who have been mg PO BID for 7 days (except in pregnancy3) Cefixime 800mg PO x 1 PLUS azithromycin 1g PO x 1 referred for oral treatment If cephalosporin allergy5 is reported, gentamicin If cefixime is unavailable, substitute cefpodoxime 400mg PO q12h should return for 240mg IM x 1 PLUS azithromycin 2g PO x 1 x 2 for cefixime in above regimens4 comprehensive testing and screening and linked to services If oral cephalosporin not available or history of cephalosporin at that time. allergy5: azithromycin 2g PO x 1 If azithromycin is not available: doxycycline 100 mg PO BID for 7 days (except in pregnancy3) Patients should be advised to abstain from sex for 7 days Treatment typically guided by examination and For presumptive therapy when examination and laboratory following completion of Vaginal discharge syndrome treatment. -

Antibiotic Use Guidelines for Companion Animal Practice (2Nd Edition) Iii
ii Antibiotic Use Guidelines for Companion Animal Practice (2nd edition) iii Antibiotic Use Guidelines for Companion Animal Practice, 2nd edition Publisher: Companion Animal Group, Danish Veterinary Association, Peter Bangs Vej 30, 2000 Frederiksberg Authors of the guidelines: Lisbeth Rem Jessen (University of Copenhagen) Peter Damborg (University of Copenhagen) Anette Spohr (Evidensia Faxe Animal Hospital) Sandra Goericke-Pesch (University of Veterinary Medicine, Hannover) Rebecca Langhorn (University of Copenhagen) Geoffrey Houser (University of Copenhagen) Jakob Willesen (University of Copenhagen) Mette Schjærff (University of Copenhagen) Thomas Eriksen (University of Copenhagen) Tina Møller Sørensen (University of Copenhagen) Vibeke Frøkjær Jensen (DTU-VET) Flemming Obling (Greve) Luca Guardabassi (University of Copenhagen) Reproduction of extracts from these guidelines is only permitted in accordance with the agreement between the Ministry of Education and Copy-Dan. Danish copyright law restricts all other use without written permission of the publisher. Exception is granted for short excerpts for review purposes. iv Foreword The first edition of the Antibiotic Use Guidelines for Companion Animal Practice was published in autumn of 2012. The aim of the guidelines was to prevent increased antibiotic resistance. A questionnaire circulated to Danish veterinarians in 2015 (Jessen et al., DVT 10, 2016) indicated that the guidelines were well received, and particularly that active users had followed the recommendations. Despite a positive reception and the results of this survey, the actual quantity of antibiotics used is probably a better indicator of the effect of the first guidelines. Chapter two of these updated guidelines therefore details the pattern of developments in antibiotic use, as reported in DANMAP 2016 (www.danmap.org). -
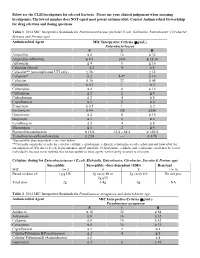
Below Are the CLSI Breakpoints for Selected Bacteria. Please Use Your Clinical Judgement When Assessing Breakpoints
Below are the CLSI breakpoints for selected bacteria. Please use your clinical judgement when assessing breakpoints. The lowest number does NOT equal most potent antimicrobial. Contact Antimicrobial Stewardship for drug selection and dosing questions. Table 1: 2014 MIC Interpretive Standards for Enterobacteriaceae (includes E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia and Proteus spp) Antimicrobial Agent MIC Interpretive Criteria (g/mL) Enterobacteriaceae S I R Ampicillin ≤ 8 16 ≥ 32 Ampicillin-sulbactam ≤ 8/4 16/8 ≥ 32/16 Aztreonam ≤ 4 8 ≥ 16 Cefazolin (blood) ≤ 2 4 ≥ 8 Cefazolin** (uncomplicated UTI only) ≤ 16 ≥ 32 Cefepime* ≤ 2 4-8* ≥ 16 Cefotetan ≤ 16 32 ≥ 64 Ceftaroline ≤ 0.5 1 ≥ 2 Ceftazidime ≤ 4 8 ≥ 16 Ceftriaxone ≤ 1 2 ≥ 4 Cefpodoxime ≤ 2 4 ≥ 8 Ciprofloxacin ≤ 1 2 ≥ 4 Ertapenem ≤ 0.5 1 ≥ 2 Fosfomycin ≤ 64 128 ≥256 Gentamicin ≤ 4 8 ≥ 16 Imipenem ≤ 1 2 ≥ 4 Levofloxacin ≤ 2 4 ≥ 8 Meropenem ≤ 1 2 ≥ 4 Piperacillin-tazobactam ≤ 16/4 32/4 – 64/4 ≥ 128/4 Trimethoprim-sulfamethoxazole ≤ 2/38 --- ≥ 4/76 *Susceptibile dose-dependent – see chart below **Cefazolin can predict results for cefaclor, cefdinir, cefpodoxime, cefprozil, cefuroxime axetil, cephalexin and loracarbef for uncomplicated UTIs due to E.coli, K.pneumoniae, and P.mirabilis. Cefpodoxime, cefinidir, and cefuroxime axetil may be tested individually because some isolated may be susceptible to these agents while testing resistant to cefazolin. Cefepime dosing for Enterobacteriaceae ( E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia & Proteus spp) Susceptible Susceptible –dose-dependent (SDD) Resistant MIC </= 2 4 8 >/= 16 Based on dose of: 1g q12h 1g every 8h or 2g every 8 h Do not give 2g q12 Total dose 2g 3-4g 6g NA Table 2: 2014 MIC Interpretive Standards for Pseudomonas aeruginosa and Acinetobacter spp.