Fosfomycin Resistance in Escherichia Coli Isolates from South Korea and in Vitro Activity of Fosfomycin Alone and in Combination with Other Antibiotics
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Medical Review(S) Clinical Review
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 200327 MEDICAL REVIEW(S) CLINICAL REVIEW Application Type NDA Application Number(s) 200327 Priority or Standard Standard Submit Date(s) December 29, 2009 Received Date(s) December 30, 2009 PDUFA Goal Date October 30, 2010 Division / Office Division of Anti-Infective and Ophthalmology Products Office of Antimicrobial Products Reviewer Name(s) Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD Review Completion October 29, 2010 Date Established Name Ceftaroline fosamil for injection (Proposed) Trade Name Teflaro Therapeutic Class Cephalosporin; ß-lactams Applicant Cerexa, Inc. Forest Laboratories, Inc. Formulation(s) 400 mg/vial and 600 mg/vial Intravenous Dosing Regimen 600 mg every 12 hours by IV infusion Indication(s) Acute Bacterial Skin and Skin Structure Infection (ABSSSI); Community-acquired Bacterial Pneumonia (CABP) Intended Population(s) Adults ≥ 18 years of age Template Version: March 6, 2009 Reference ID: 2857265 Clinical Review Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD NDA 200327: Teflaro (ceftaroline fosamil) Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT ......................................... 9 1.1 Recommendation on Regulatory Action ........................................................... 10 1.2 Risk Benefit Assessment.................................................................................. 10 1.3 Recommendations for Postmarketing Risk Evaluation and Mitigation Strategies ........................................................................................................................ -

Cefoxitin Versus Piperacillin– Tazobactam As Surgical Antibiotic Prophylaxis in Patients Undergoing Pancreatoduodenectomy: Protocol for a Randomised Controlled Trial
Open access Protocol BMJ Open: first published as 10.1136/bmjopen-2020-048398 on 4 March 2021. Downloaded from Cefoxitin versus piperacillin– tazobactam as surgical antibiotic prophylaxis in patients undergoing pancreatoduodenectomy: protocol for a randomised controlled trial Nicole M Nevarez ,1 Brian C Brajcich,2 Jason Liu,2,3 Ryan Ellis,2 Clifford Y Ko,2 Henry A Pitt,4 Michael I D'Angelica,5 Adam C Yopp1 To cite: Nevarez NM, ABSTRACT Strengths and limitations of this study Brajcich BC, Liu J, et al. Introduction Although antibiotic prophylaxis is Cefoxitin versus piperacillin– established in reducing postoperative surgical site tazobactam as surgical ► A major strength of this study is the multi- infections (SSIs), the optimal antibiotic for prophylaxis in antibiotic prophylaxis institutional, double- arm, randomised controlled in patients undergoing pancreatoduodenectomy (PD) remains unclear. The study trial design. objective is to evaluate if administration of piperacillin– pancreatoduodenectomy: ► A limitation of this study is that all perioperative care protocol for a randomised tazobactam as antibiotic prophylaxis results in decreased is at the discretion of the operating surgeon and is controlled trial. BMJ Open 30- day SSI rate compared with cefoxitin in patients not standardised. 2021;11:e048398. doi:10.1136/ undergoing elective PD. ► All data will be collected through the American bmjopen-2020-048398 Methods and analysis This study will be a multi- College of Surgeons National Surgical Quality ► Prepublication history for institution, double- arm, non- blinded randomised controlled Improvement Program, which is a strength for its this paper is available online. superiority trial. Adults ≥18 years consented to undergo PD ease of use but a limitation due to the variety of data To view these files, please visit for all indications who present to institutions participating included. -

Cefalexin in the WHO Essential Medicines List for Children Reject
Reviewer No. 1: checklist for application of: Cefalexin In the WHO Essential Medicines List for Children (1) Have all important studies that you are aware of been included? Yes No 9 (2) Is there adequate evidence of efficacy for the proposed use? Yes 9 No (3) Is there evidence of efficacy in diverse settings and/or populations? Yes 9 No (4) Are there adverse effects of concern? Yes 9 No (5) Are there special requirements or training needed for safe/effective use? Yes No 9 (6) Is this product needed to meet the majority health needs of the population? Yes No 9 (7) Is the proposed dosage form registered by a stringent regulatory authority? Yes 9 No (8) What action do you propose for the Committee to take? Reject the application for inclusion of the following presentations of cefalexin: • Tablets/ capsules 250mg • Oral suspensions 125mg/5ml and 125mg/ml (9) Additional comment, if any. In order to identify any additional literature, the following broad and sensitive search was conducted using the PubMed Clinical Query application: (cephalexin OR cefalexin AND - 1 - pediatr*) AND ((clinical[Title/Abstract] AND trial[Title/Abstract]) OR clinical trials[MeSH Terms] OR clinical trial[Publication Type] OR random*[Title/Abstract] OR random allocation[MeSH Terms] OR therapeutic use[MeSH Subheading]) Only one small additional study was identified, which looked at the provision of prophylactic antibiotics in patients presenting to an urban children's hospital with trauma to the distal fingertip, requiring repair.1 In a prospective randomised control trial, 146 patients were enrolled, of which 69 were randomised to the no-antibiotic group, and 66 were randomised to the antibiotic (cefalexin) group. -

Cefixime (Suprax) Division of Disease Control What Do I Need to Know?
Division of Disease Control What Do I Need To Know? Cefixime (Suprax) About your medicine: Cefixime is used to treat bacterial infections in many different parts of the body. This medicine will not work for colds, flu, or other viral infections. It may be used to other problems as determined by your healthcare provider. These instructions will help you receive the optimum benefits from Cefixime. Before taking this medication: Tell your doctor, nurse or healthcare provider if you… . Have any allergies to medications (especially cephalosporins and penicillin) . Take any other medications . Have medical condition (especially kidney or eye problems) . Are pregnant, breast-feeding or trying to become pregnant . History of convulsions Possible side effects of this medicine: If these symptoms continue or become severe, call your health care provider… . Nausea, vomiting, diarrhea . Gas . Indigestion . Dyspepsia (upset stomach) . Abdominal pain . Muscle cramps, stiffness, spasms . If your symptoms do not improve within a few days, notify your healthcare provider How to take this medicine: . This is the only dose of this medicine you will receive . Take this medication with a full glass (8 ounces) of water, preferably after a meal . DO NOT take with milk or other dairy products, or any products containing magnesium or calcium (such as antacids), iron or aluminum within 2 hours of the time you take this medicine . Drink several additional glasses of water today Warning: Call a healthcare professional immediately if you develop any of the following: . Difficulty breathing . Hives/skin rash Sexual Behavior: Abstain from any type of sexual activity for 7 days after treatment of self and partner For more information, contact the NDDoH STD Program at 701.328.2378 or toll-free 800.472.2180 Page 1 of 1 Last Updated: 4/3/2012 . -

Efficacy of Fosfomycin Trometamol and Cefuroxime Axetil in the Treatment of Kashmir, India Urinary Tract Infections During Pregnancy
International Journal of Surgery Science 2019; 3(2): 33-35 E-ISSN: 2616-3470 P-ISSN: 2616-3462 © Surgery Science Efficacy of fosfomycin trometamol and cefuroxime axetil www.surgeryscience.com 2019; 3(2): 33-35 in the treatment of UTI during pregnancy: A Received: 18-11-2018 Accepted: 22-12-2018 comparative study Dr. Robindera Kour In Charge Consultant Surgery, Dr. Robindera Kour, Dr. Gurpreet Kour, Dr. Iqbal Singh, Dr. KK Gupta Govt. Hospital Sarwal, Jammu, and Dr. Rajiv Sharma Jammu and Kashmir, India Dr. Gurpreet Kour DOI: https://doi.org/10.33545/surgery.2019.v3.i2a.373 Medical Officer, Acharya Shri Chander College of Medical Abstract Sciences, Jammu, Jammu and Objective: To compare the efficacy of fosfomycin trometamol and cefuroxime axetil in the treatment of Kashmir, India urinary tract infections during pregnancy. Methods: Prospective clinical study was conducted among pregnant women who were followed as part of Dr. Iqbal Singh routine prenatal care at Government Hospital Sarwal, Jammu, India with complaints of lower UTI were Assistant Professor, Public Health assessed. 60 patients were enrolled, divided into 2 equal groups for treatment with single-dose fosfomycin Dentistry, Indira Gandhi Government Dental College, trometamol and 5-day courses of cefuroxime axetil. Follow-up was carried out after 5 weeks. Jammu, Jammu and Kashmir, Results: The treatment groups did not differ significantly in terms of demographics, clinical compliance India rate and adverse effects. But the patients who were taking 5-day courses of cefuroxime axetil showed statistically significant higher cure rate as well as lower recurrence rate as compared to fosfomycin Dr. -

Severe Sepsis and Septic Shock Antibiotic Guide
Stanford Health Issue Date: 05/2017 Stanford Antimicrobial Safety and Sustainability Program Severe Sepsis and Septic Shock Antibiotic Guide Table 1: Antibiotic selection options for healthcare associated and/or immunocompromised patients • Healthcare associated: intravenous therapy, wound care, or intravenous chemotherapy within the prior 30 days, residence in a nursing home or other long-term care facility, hospitalization in an acute care hospital for two or more days within the prior 90 days, attendance at a hospital or hemodialysis clinic within the prior 30 days • Immunocompromised: Receiving chemotherapy, known systemic cancer not in remission, ANC <500, severe cell-mediated immune deficiency Table 2: Antibiotic selection options for community acquired, immunocompetent patients Table 3: Antibiotic selection options for patients with simple sepsis, community acquired, immunocompetent patients requiring hospitalization. Risk Factors for Select Organisms P. aeruginosa MRSA Invasive Candidiasis VRE (and other resistant GNR) Community acquired: • Known colonization with MDROs • Central venous catheter • Liver transplant • Prior IV antibiotics within 90 day • Recent MRSA infection • Broad-spectrum antibiotics • Known colonization • Known colonization with MDROs • Known MRSA colonization • + 1 of the following risk factors: • Prolonged broad antibacterial • Skin & Skin Structure and/or IV access site: ♦ Parenteral nutrition therapy Hospital acquired: ♦ Purulence ♦ Dialysis • Prolonged profound • Prior IV antibiotics within 90 days ♦ Abscess -

In Vitro Susceptibility of Multi-Drug Resistant Klebsiella Pneumoniae Strains Causing Nosocomial Infections to Fosfomycin. a Comparison of Determination Methods
pathogens Article In Vitro Susceptibility of Multi-Drug Resistant Klebsiella pneumoniae Strains Causing Nosocomial Infections to Fosfomycin. A Comparison of Determination Methods Beata M ˛aczy´nska 1,* , Justyna Paleczny 1 , Monika Oleksy-Wawrzyniak 1 , Irena Choroszy-Król 2 and Marzenna Bartoszewicz 1 1 Department of Pharmaceutical Microbiology and Parasitology, Faculty of Pharmacy, Medical University, 50-367 Wroclaw, Poland; [email protected] (J.P.); [email protected] (M.O.-W.); [email protected] (M.B.) 2 Department of Basic Sciences, Faculty of Health Sciences, Medical University, 50-367 Wroclaw, Poland; [email protected] * Correspondence: [email protected] Abstract: Introduction: Over the past few decades, Klebsiella pneumoniae strains increased their pathogenicity and antibiotic resistance, thereby becoming a major therapeutic challenge. One of the few available therapeutic options seems to be intravenous fosfomycin. Unfortunately, the determination of sensitivity to fosfomycin performed in hospital laboratories can pose a significant problem. Therefore, the aim of the present research was to evaluate the activity of fosfomycin against clinical, multidrug-resistant Klebsiella pneumoniae strains isolated from nosocomial infections between Citation: M ˛aczy´nska,B.; Paleczny, J.; 2011 and 2020, as well as to evaluate the methods routinely used in hospital laboratories to assess Oleksy-Wawrzyniak, M.; bacterial susceptibility to this antibiotic. Materials and Methods: 43 multidrug-resistant Klebsiella Choroszy-Król, I.; Bartoszewicz, M. strains isolates from various infections were tested. All the strains had ESBL enzymes, and 20 In Vitro Susceptibility of Multi-Drug also showed the presence of carbapenemases. Susceptibility was determined using the diffusion Resistant Klebsiella pneumoniae Strains method (E-test) and the automated system (Phoenix), which were compared with the reference Causing Nosocomial Infections to method (agar dilution). -

Sexually Transmitted Diseases Treatment Options
Sexually transmitted disease (STD) treatment options PREFERRED & ALTERNATIVE OPTIONS Many clinical partners are operating in a limited capacity during the COVID-19 pandemic. Below are preferred (in clinic or other location where injections can be given) and alternative (when only oral medicines are available 1) treatments for STDs. Syndrome Preferred Treatments Alternative Treatments Follow-up Male urethritis syndrome Ceftriaxone 250mg intramuscular (IM) x 1 PLUS Men who have sex with men (MSM) and transgender women2: Patients should be counseled to azithromycin 1g PO x 1 Cefixime 800 mg PO x 1 PLUS doxycycline 100 mg PO BID x 7 days be tested for STDs once clinical Presumptively treating: care is resumed in the local If azithromycin is not available: doxycycline 100 Men who have sex with women only: gonorrhea clinics. Clients who have been mg PO BID for 7 days (except in pregnancy3) Cefixime 800mg PO x 1 PLUS azithromycin 1g PO x 1 referred for oral treatment If cephalosporin allergy5 is reported, gentamicin If cefixime is unavailable, substitute cefpodoxime 400mg PO q12h should return for 240mg IM x 1 PLUS azithromycin 2g PO x 1 x 2 for cefixime in above regimens4 comprehensive testing and screening and linked to services If oral cephalosporin not available or history of cephalosporin at that time. allergy5: azithromycin 2g PO x 1 If azithromycin is not available: doxycycline 100 mg PO BID for 7 days (except in pregnancy3) Patients should be advised to abstain from sex for 7 days Treatment typically guided by examination and For presumptive therapy when examination and laboratory following completion of Vaginal discharge syndrome treatment. -
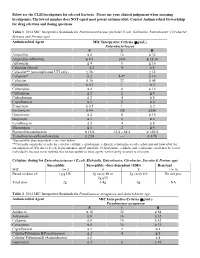
Below Are the CLSI Breakpoints for Selected Bacteria. Please Use Your Clinical Judgement When Assessing Breakpoints
Below are the CLSI breakpoints for selected bacteria. Please use your clinical judgement when assessing breakpoints. The lowest number does NOT equal most potent antimicrobial. Contact Antimicrobial Stewardship for drug selection and dosing questions. Table 1: 2014 MIC Interpretive Standards for Enterobacteriaceae (includes E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia and Proteus spp) Antimicrobial Agent MIC Interpretive Criteria (g/mL) Enterobacteriaceae S I R Ampicillin ≤ 8 16 ≥ 32 Ampicillin-sulbactam ≤ 8/4 16/8 ≥ 32/16 Aztreonam ≤ 4 8 ≥ 16 Cefazolin (blood) ≤ 2 4 ≥ 8 Cefazolin** (uncomplicated UTI only) ≤ 16 ≥ 32 Cefepime* ≤ 2 4-8* ≥ 16 Cefotetan ≤ 16 32 ≥ 64 Ceftaroline ≤ 0.5 1 ≥ 2 Ceftazidime ≤ 4 8 ≥ 16 Ceftriaxone ≤ 1 2 ≥ 4 Cefpodoxime ≤ 2 4 ≥ 8 Ciprofloxacin ≤ 1 2 ≥ 4 Ertapenem ≤ 0.5 1 ≥ 2 Fosfomycin ≤ 64 128 ≥256 Gentamicin ≤ 4 8 ≥ 16 Imipenem ≤ 1 2 ≥ 4 Levofloxacin ≤ 2 4 ≥ 8 Meropenem ≤ 1 2 ≥ 4 Piperacillin-tazobactam ≤ 16/4 32/4 – 64/4 ≥ 128/4 Trimethoprim-sulfamethoxazole ≤ 2/38 --- ≥ 4/76 *Susceptibile dose-dependent – see chart below **Cefazolin can predict results for cefaclor, cefdinir, cefpodoxime, cefprozil, cefuroxime axetil, cephalexin and loracarbef for uncomplicated UTIs due to E.coli, K.pneumoniae, and P.mirabilis. Cefpodoxime, cefinidir, and cefuroxime axetil may be tested individually because some isolated may be susceptible to these agents while testing resistant to cefazolin. Cefepime dosing for Enterobacteriaceae ( E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia & Proteus spp) Susceptible Susceptible –dose-dependent (SDD) Resistant MIC </= 2 4 8 >/= 16 Based on dose of: 1g q12h 1g every 8h or 2g every 8 h Do not give 2g q12 Total dose 2g 3-4g 6g NA Table 2: 2014 MIC Interpretive Standards for Pseudomonas aeruginosa and Acinetobacter spp. -

Guidelines on Urinary and Male Genital Tract Infections
European Association of Urology GUIDELINES ON URINARY AND MALE GENITAL TRACT INFECTIONS K.G. Naber, B. Bergman, M.C. Bishop, T.E. Bjerklund Johansen, H. Botto, B. Lobel, F. Jimenez Cruz, F.P. Selvaggi TABLE OF CONTENTS PAGE 1. INTRODUCTION 5 1.1 Classification 5 1.2 References 6 2. UNCOMPLICATED UTIS IN ADULTS 7 2.1 Summary 7 2.2 Background 8 2.3 Definition 8 2.4 Aetiological spectrum 9 2.5 Acute uncomplicated cystitis in pre-menopausal, non-pregnant women 9 2.5.1 Diagnosis 9 2.5.2 Treatment 10 2.5.3 Post-treatment follow-up 11 2.6 Acute uncomplicated pyelonephritis in pre-menopausal, non-pregnant women 11 2.6.1 Diagnosis 11 2.6.2 Treatment 12 2.6.3 Post-treatment follow-up 12 2.7 Recurrent (uncomplicated) UTIs in women 13 2.7.1 Background 13 2.7.2 Prophylactic antimicrobial regimens 13 2.7.3 Alternative prophylactic methods 14 2.8 UTIs in pregnancy 14 2.8.1 Epidemiology 14 2.8.2 Asymptomatic bacteriuria 15 2.8.3 Acute cystitis during pregnancy 15 2.8.4 Acute pyelonephritis during pregnancy 15 2.9 UTIs in post-menopausal women 15 2.10 Acute uncomplicated UTIs in young men 16 2.10.1 Pathogenesis and risk factors 16 2.10.2 Diagnosis 16 2.10.3 Treatment 16 2.11 References 16 3. UTIs IN CHILDREN 20 3.1 Summary 20 3.2 Background 20 3.3 Aetiology 20 3.4 Pathogenesis 20 3.5 Signs and symptoms 21 3.5.1 New-borns 21 3.5.2 Children < 6 months of age 21 3.5.3 Pre-school children (2-6 years of age) 21 3.5.4 School-children and adolescents 21 3.5.5 Severity of a UTI 21 3.5.6 Severe UTIs 21 3.5.7 Simple UTIs 21 3.5.8 Epididymo orchitis 22 3.6 Diagnosis 22 3.6.1 Physical examination 22 3.6.2 Laboratory tests 22 3.6.3 Imaging of the urinary tract 23 3.7 Schedule of investigation 24 3.8 Treatment 24 3.8.1 Severe UTIs 25 3.8.2 Simple UTIs 25 3.9 References 26 4. -

Piperacillin/Tazobactam Drug Class1 Antibiotic – Penicillin with Β-Lactamase Inhibitor
Monographs for Commonly Administered Intravenous Medications in Home and Community Care Piperacillin/Tazobactam Drug Class1 Antibiotic – penicillin with β-lactamase inhibitor Spectrum1 Refer to product monograph for complete spectrum For β-lactamase producing bacteria strains (e.g., Haemophilus influenza, Escherichia coli, and Staphylococcus aureus). Also to susceptible Acinetobacter species, Klebsiella pneumonia, Pseudomonas aeruginosa often combined with aminoglycoside). Cross Sensitivities / Allergies1 Cross sensitivities with penicillin and possibly cephalosporin and/or beta-lactam inhibitors Indications1,2 Intra-abdominal Respiratory tract Skin and skin structure Septicemia Gynecological Urinary tract Other conditions based on culture and sensitivity results Outpatient Considerations1 For patients with a documented allergy to penicillin, cephalosporin or beta-lactam inhibitor, the first dose should be administered in a hospital or clinic setting. Must be able to access laboratory monitoring (either at outpatient laboratory or by arranging in-home lab) if using an interacting oral medication (see Potential Drug Interactions section) Prescribing Considerations At time of ordering please provide the following to the pharmacist: and Dosage in Adults1,2 Height, weight Most recent serum creatinine with date obtained Indication (infection being treated) Usually dosed every 6 to 8 hours. Available as 2 g/0.25 g, 3 g/0.375 g, 4 g/0.5 g piperacillin/tazobactam (dose selected based on indication) Maximum 18 g/ 2.25 g piperacillin/tazobactam daily Dose and administration interval require adjustment for renal impairment Errors have occurred due to unusual ordering as combined dose of piperacillin and tazobactam. Order may be expressed only as piperacillin component or as a total of piperacillin + tazobactam (8:1 ratio). -

Gonorrhea, Chlamydia, and Syphilis
2019 GONORRHEA, CHLAMYDIA, AND SYPHILIS AND CHLAMYDIA, GONORRHEA, Dedication TAG would like to thank the National Coalition of STD Directors for funding and input on the report. THE PIPELINE REPORT Pipeline for Gonorrhea, Chlamydia, and Syphilis By Jeremiah Johnson Introduction The current toolbox for addressing gonorrhea, chlamydia, and syphilis is inadequate. At a time where all three epidemics are dramatically expanding in locations all around the globe, including record-breaking rates of new infections in the United States, stakeholders must make do with old tools, inadequate systems for addressing sexual health, and a sparse research pipeline of new treatment, prevention, and diagnostic options. Lack of investment in sexual health research has left the field with inadequate prevention options, and limited access to infrastructure for testing and treatment have allowed sexually transmitted infections (STIs) to flourish. The consequences of this underinvestment are large: according to the World Health Organization (WHO), in 2012 there were an estimated 357 million new infections (roughly 1 million per day) of the four curable STIs: gonorrhea, chlamydia, syphilis, and trichomoniasis.1 In the United States, the three reportable STIs that are the focus of this report—gonorrhea, chlamydia, and syphilis—are growing at record paces. In 2017, a total of 30,644 cases of primary and secondary (P&S) syphilis—the most infectious stages of the disease—were reported in the United States. Since reaching a historic low in 2000 and 2001, the rate of P&S syphilis has increased almost every year, increasing 10.5% during 2016–2017. Also in 2017, 555,608 cases of gonorrhea were reported to the U.S.