The Antibacterial Activity of Cefpodoxime and the Novel Β
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Cefalexin in the WHO Essential Medicines List for Children Reject
Reviewer No. 1: checklist for application of: Cefalexin In the WHO Essential Medicines List for Children (1) Have all important studies that you are aware of been included? Yes No 9 (2) Is there adequate evidence of efficacy for the proposed use? Yes 9 No (3) Is there evidence of efficacy in diverse settings and/or populations? Yes 9 No (4) Are there adverse effects of concern? Yes 9 No (5) Are there special requirements or training needed for safe/effective use? Yes No 9 (6) Is this product needed to meet the majority health needs of the population? Yes No 9 (7) Is the proposed dosage form registered by a stringent regulatory authority? Yes 9 No (8) What action do you propose for the Committee to take? Reject the application for inclusion of the following presentations of cefalexin: • Tablets/ capsules 250mg • Oral suspensions 125mg/5ml and 125mg/ml (9) Additional comment, if any. In order to identify any additional literature, the following broad and sensitive search was conducted using the PubMed Clinical Query application: (cephalexin OR cefalexin AND - 1 - pediatr*) AND ((clinical[Title/Abstract] AND trial[Title/Abstract]) OR clinical trials[MeSH Terms] OR clinical trial[Publication Type] OR random*[Title/Abstract] OR random allocation[MeSH Terms] OR therapeutic use[MeSH Subheading]) Only one small additional study was identified, which looked at the provision of prophylactic antibiotics in patients presenting to an urban children's hospital with trauma to the distal fingertip, requiring repair.1 In a prospective randomised control trial, 146 patients were enrolled, of which 69 were randomised to the no-antibiotic group, and 66 were randomised to the antibiotic (cefalexin) group. -

Antimicrobial Stewardship Guidance
Antimicrobial Stewardship Guidance Federal Bureau of Prisons Clinical Practice Guidelines March 2013 Clinical guidelines are made available to the public for informational purposes only. The Federal Bureau of Prisons (BOP) does not warrant these guidelines for any other purpose, and assumes no responsibility for any injury or damage resulting from the reliance thereof. Proper medical practice necessitates that all cases are evaluated on an individual basis and that treatment decisions are patient-specific. Consult the BOP Clinical Practice Guidelines Web page to determine the date of the most recent update to this document: http://www.bop.gov/news/medresources.jsp Federal Bureau of Prisons Antimicrobial Stewardship Guidance Clinical Practice Guidelines March 2013 Table of Contents 1. Purpose ............................................................................................................................................. 3 2. Introduction ...................................................................................................................................... 3 3. Antimicrobial Stewardship in the BOP............................................................................................ 4 4. General Guidance for Diagnosis and Identifying Infection ............................................................. 5 Diagnosis of Specific Infections ........................................................................................................ 6 Upper Respiratory Infections (not otherwise specified) .............................................................................. -
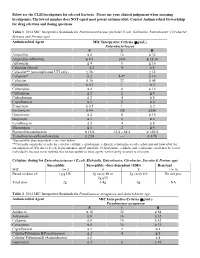
Below Are the CLSI Breakpoints for Selected Bacteria. Please Use Your Clinical Judgement When Assessing Breakpoints
Below are the CLSI breakpoints for selected bacteria. Please use your clinical judgement when assessing breakpoints. The lowest number does NOT equal most potent antimicrobial. Contact Antimicrobial Stewardship for drug selection and dosing questions. Table 1: 2014 MIC Interpretive Standards for Enterobacteriaceae (includes E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia and Proteus spp) Antimicrobial Agent MIC Interpretive Criteria (g/mL) Enterobacteriaceae S I R Ampicillin ≤ 8 16 ≥ 32 Ampicillin-sulbactam ≤ 8/4 16/8 ≥ 32/16 Aztreonam ≤ 4 8 ≥ 16 Cefazolin (blood) ≤ 2 4 ≥ 8 Cefazolin** (uncomplicated UTI only) ≤ 16 ≥ 32 Cefepime* ≤ 2 4-8* ≥ 16 Cefotetan ≤ 16 32 ≥ 64 Ceftaroline ≤ 0.5 1 ≥ 2 Ceftazidime ≤ 4 8 ≥ 16 Ceftriaxone ≤ 1 2 ≥ 4 Cefpodoxime ≤ 2 4 ≥ 8 Ciprofloxacin ≤ 1 2 ≥ 4 Ertapenem ≤ 0.5 1 ≥ 2 Fosfomycin ≤ 64 128 ≥256 Gentamicin ≤ 4 8 ≥ 16 Imipenem ≤ 1 2 ≥ 4 Levofloxacin ≤ 2 4 ≥ 8 Meropenem ≤ 1 2 ≥ 4 Piperacillin-tazobactam ≤ 16/4 32/4 – 64/4 ≥ 128/4 Trimethoprim-sulfamethoxazole ≤ 2/38 --- ≥ 4/76 *Susceptibile dose-dependent – see chart below **Cefazolin can predict results for cefaclor, cefdinir, cefpodoxime, cefprozil, cefuroxime axetil, cephalexin and loracarbef for uncomplicated UTIs due to E.coli, K.pneumoniae, and P.mirabilis. Cefpodoxime, cefinidir, and cefuroxime axetil may be tested individually because some isolated may be susceptible to these agents while testing resistant to cefazolin. Cefepime dosing for Enterobacteriaceae ( E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia & Proteus spp) Susceptible Susceptible –dose-dependent (SDD) Resistant MIC </= 2 4 8 >/= 16 Based on dose of: 1g q12h 1g every 8h or 2g every 8 h Do not give 2g q12 Total dose 2g 3-4g 6g NA Table 2: 2014 MIC Interpretive Standards for Pseudomonas aeruginosa and Acinetobacter spp. -

Antibiotic Review
8/28/19 Disclosure 2 E. Monee’ Carter-Griffin DNP, MA, RN, ACNP-BC Antibiotic has no financial relationships with commercial Review interests to disclose. Any unlaBeled and/or E. Monee’ Carter-Griffin, DNP, MA, RN, ACNP-BC unapproved uses of drugs or products will Be Associate Chair for Advanced Practice Nursing disclosed. University of Texas at Arlington, College of Nursing & Health Innovation Dallas Pulmonary & Critical Care, PA • Identify the general characteristics of • SusceptiBility à determine which antibiotics. antibiotics a bacteria is sensitive to • Discuss the mechanism of action, • à pharmacokinetics, and spectrum of activity MIC lowest concentration of drug that inhiBits growth of the Bacteria for the most common antiBiotic drug classes. Antibiotic Objectives • Identify commonly prescriBed antiBiotics • Trough à lowest concentration of within the drug classes including dosage Lingo drug in bloodstream range and indication for prescribing. – Collect prior to administration of • Identify special considerations for specific drug antibiotics and/or drug classes. • Peak à highest concentration of • Practice appropriate prescriBing for common drug in bloodstream bacterial infections. • Review Basic antiBiotic stewardship 3 4 principles. • Penicillins • Bactericidal or bacteriostatic • Cephalosporins • Narrow or Broad spectrum • Carbapenems General • Can elicit allergic responses Antibiotic • MonoBactam Characteristics • Affect normal Body flora Drug Classes • Macrolides • Fluoroquinolones • Sulfonamides • Tetracyclines 5 6 1 8/28/19 • Penicillins – Natural penicillins 8 – Aminopenicillins Penicillins Penicillins – Anti-staphylococcal penicillins • Mechanism of action àbactericidal à – Anti-pseudomonal penicillins interrupts cell wall synthesis 7 • Pharmacokinetics • Spectrum of Activity – Gram positive organisms Natural – Penicillin G à mostly given IM, But Natural • Streptococcus species (e.g. S. Penicillins one variation can Be given IV Penicillins pyogenes) • Poorly aBsorBed via PO • Some enterococcus species (e.g. -

Cephalosporins Can Be Prescribed Safely for Penicillin-Allergic Patients ▲
JFP_0206_AE_Pichichero.Final 1/23/06 1:26 PM Page 106 APPLIED EVIDENCE New research findings that are changing clinical practice Michael E. Pichichero, MD University of Rochester Cephalosporins can be Medical Center, Rochester, NY prescribed safely for penicillin-allergic patients Practice recommendations an allergic reaction to cephalosporins, ■ The widely quoted cross-allergy risk compared with the incidence of a primary of 10% between penicillin and (and unrelated) cephalosporin allergy. cephalosporins is a myth (A). Most people produce IgG and IgM antibodies in response to exposure to ■ Cephalothin, cephalexin, cefadroxil, penicillin1 that may cross-react with and cefazolin confer an increased risk cephalosporin antigens.2 The presence of of allergic reaction among patients these antibodies does not predict allergic, with penicillin allergy (B). IgE cross-sensitivity to a cephalosporin. ■ Cefprozil, cefuroxime, cefpodoxime, Even penicillin skin testing is generally not ceftazidime, and ceftriaxone do not predictive of cephalosporin allergy.3 increase risk of an allergic reaction (B). Reliably predicting cross-reactivity ndoubtedly you have patients who A comprehensive review of the evidence say they are allergic to penicillin shows that the attributable risk of a cross- U but have difficulty recalling details reactive allergic reaction varies and is of the reactions they experienced. To be strongest when the chemical side chain of safe, we often label these patients as peni- the specific cephalosporin is similar to that cillin-allergic without further questioning of penicillin or amoxicillin. and withhold not only penicillins but Administration of cephalothin, cepha- cephalosporins due to concerns about lexin, cefadroxil, and cefazolin in penicillin- potential cross-reactivity and resultant IgE- allergic patients is associated with a mediated, type I reactions. -

Technical Appendix
Article DOI: http://dx.doi.org/10.3201/eid2405.171830 Antimicrobial Resistance in Invasive Bacterial Infections in Hospitalized Children, Cambodia, 2007–2016 Technical Appendix Methods Classification of blood culture isolates Coagulase-negative Staphylococci, Micrococcus species, and unspecified Gram-positive bacilli were classified as contaminants. Non-pathogenic environmental non-fermenting Gram- negatives were classified as of uncertain significance. All other bacterial species were considered as pathogens. Management of Antimicrobial Susceptibility Testing data Antimicrobial susceptibility testing (AST) results for BC isolates from 2007 to mid-2011 were interpreted as previously described (1). For BC isolates from mid-2011 onwards and CSFC isolates, disk-diffusion zone sizes and MIC results were available and re-interpreted using CLSI 2017 performance standards (2). Isolates classified as resistant, intermediate, or non-susceptible were analyzed as resistant. Changes in microbiology practice and guidelines meant species- specific AST panels varied over the study period. To account for this, pragmatic definitions of class resistance were used, and isolates only considered tested for multidrug resistant (MDR) status if they had undergone an appropriate minimum MDR test panel (Technical Appendix Table 1). Technical Appendix Table 1. Definitions of antimicrobial resistance to be reported and minimum multidrug resistance (MDR) test panels for each organism/organism group* MDR test panel - minimum antimicrobial Organism / Organism Resistance -

IDSA/ATS Consensus Guidelines on The
SUPPLEMENT ARTICLE Infectious Diseases Society of America/American Thoracic Society Consensus Guidelines on the Management of Community-Acquired Pneumonia in Adults Lionel A. Mandell,1,a Richard G. Wunderink,2,a Antonio Anzueto,3,4 John G. Bartlett,7 G. Douglas Campbell,8 Nathan C. Dean,9,10 Scott F. Dowell,11 Thomas M. File, Jr.12,13 Daniel M. Musher,5,6 Michael S. Niederman,14,15 Antonio Torres,16 and Cynthia G. Whitney11 1McMaster University Medical School, Hamilton, Ontario, Canada; 2Northwestern University Feinberg School of Medicine, Chicago, Illinois; 3University of Texas Health Science Center and 4South Texas Veterans Health Care System, San Antonio, and 5Michael E. DeBakey Veterans Affairs Medical Center and 6Baylor College of Medicine, Houston, Texas; 7Johns Hopkins University School of Medicine, Baltimore, Maryland; 8Division of Pulmonary, Critical Care, and Sleep Medicine, University of Mississippi School of Medicine, Jackson; 9Division of Pulmonary and Critical Care Medicine, LDS Hospital, and 10University of Utah, Salt Lake City, Utah; 11Centers for Disease Control and Prevention, Atlanta, Georgia; 12Northeastern Ohio Universities College of Medicine, Rootstown, and 13Summa Health System, Akron, Ohio; 14State University of New York at Stony Brook, Stony Brook, and 15Department of Medicine, Winthrop University Hospital, Mineola, New York; and 16Cap de Servei de Pneumologia i Alle`rgia Respirato`ria, Institut Clı´nic del To`rax, Hospital Clı´nic de Barcelona, Facultat de Medicina, Universitat de Barcelona, Institut d’Investigacions Biome`diques August Pi i Sunyer, CIBER CB06/06/0028, Barcelona, Spain. EXECUTIVE SUMMARY priate starting point for consultation by specialists. Substantial overlap exists among the patients whom Improving the care of adult patients with community- these guidelines address and those discussed in the re- acquired pneumonia (CAP) has been the focus of many cently published guidelines for health care–associated different organizations, and several have developed pneumonia (HCAP). -

Vs. Long-Course Antibiotic Treatment for Acute Streptococcal Pharyngitis: Systematic Review and Meta-Analysis of Randomized Controlled Trials
antibiotics Review Short- vs. Long-Course Antibiotic Treatment for Acute Streptococcal Pharyngitis: Systematic Review and Meta-Analysis of Randomized Controlled Trials Anna Engell Holm * , Carl Llor, Lars Bjerrum and Gloria Cordoba Research Unit for General Practice and Section of General Practice, Department of Public Health, Øster Farimagsgade 5, 1014 Copenhagen, Denmark; [email protected] (C.L.); [email protected] (L.B.); [email protected] (G.C.) * Correspondence: [email protected]; Tel.: +45-261-110-54 Received: 9 September 2020; Accepted: 21 October 2020; Published: 26 October 2020 Abstract: BACKGROUND: To evaluate the effectiveness of short courses of antibiotic therapy for patients with acute streptococcal pharyngitis. METHODS: Randomized controlled trials comparing short-course antibiotic therapy ( 5 days) with long-course antibiotic therapy ( 7 days) for patients with ≤ ≥ streptococcal pharyngitis were included. Two primary outcomes: early clinical cure and early bacterial eradication. RESULTS: Fifty randomized clinical trials were included. Overall, short-course antibiotic treatment was as effective as long-course antibiotic treatment for early clinical cure (odds ratio (OR) 0.85; 95% confidence interval (CI) 0.79 to 1.15). Subgroup analysis showed that short-course penicillin was less effective for early clinical cure (OR 0.43; 95% CI, 0.23 to 0.82) and bacteriological eradication (OR 0.34; 95% CI, 0.19 to 0.61) in comparison to long-course penicillin. Short-course macrolides were equally effective, compared to long-course penicillin. Finally, short-course cephalosporin was more effective for early clinical cure (OR 1.48; 95% CI, 1.11 to 1.96) and early microbiological cure (OR 1.60; 95% CI, 1.13 to 2.27) in comparison to long-course penicillin. -

A Review of Recommended Antibiotic Therapies with Impact on Outcomes in Acute Otitis Media and Acute Bacterial Sinusitis
REPORTS A Review of Recommended Antibiotic Therapies With Impact on Outcomes in Acute Otitis Media and Acute Bacterial Sinusitis Michael E. Pichichero, MD, and Diana I. Brixner, RPh, PhD cute otitis media (AOM) and acute efficacy and adherence in the treatment of bacterial sinusitis (ABS) manage- AOM and ABS. A ment is becoming increasingly com- plex, with continual changes in the Direct and Indirect Costs pathogens responsible for these conditions. of AOM and ABS Treatment involves the selection of an effi- The total costs of both AOM and ABS can cacious agent displaying a favorable adher- be high. Direct AOM and ABS costs are typ- ence profile. Although antibiotic efficacy is ically based on medication use and physi- critical to treatment successes, therapy cian visits. Annual estimates of direct AOM adherence is arguably of equal importance, costs range from $1.96 billion to $4.1 bil- because failure to adhere to even the most lion,2,3 whereas direct sinusitis costs (as a effective therapy can ultimately lead to clin- primary diagnosis) are estimated at $1.8 bil- ical failure and undermine treatment efforts. lion.4 However, direct costs represent only a The dual importance of efficacy and adher- portion of the total economic burden of ill- ence requires additional consideration when ness. Indirect costs can include the expense treating pediatric infections. Managed care of care for sick children, transportation organizations (MCOs) are increasingly costs, the value of work time lost, baby-sit- aware that a balance is needed when weigh- ting fees, ancillary medication costs, and ing efficacy and adherence issues with expenditures for treatment of adverse antibiotics. -

Antimicrobial Stewardship in LTCF
Antimicrobial Stewardship in Long-Term Care Facilities Maryland Train the Trainer Program A collaboration between University of Maryland School of Pharmacy, Peter Lamy Center on Drug Therapy and Aging, and Maryland Department of Mental Health and Hygiene Nicole J. Brandt, PharmD, MBA, BCGP, BCPP , FASCP Professor, Geriatric Pharmacotherapy, Pharmacy Practice and Science University of Maryland School of Pharmacy Executive Director, Peter Lamy Center Drug Therapy and Aging Email: [email protected] •In Progress •Completed Activities Grant Activities •Remaining Activities Gap Survey Analysis of IRB Approval Analysis Completion Results ABX Summit CE Summit CE ABX Summit Activity 1 Coordination Processing Educational Educational Activity 2 Theme 1: Theme 2: ASB & UTIs Antibiograms ADE Tool ADE Toolkit Prototype Prototype Feedback & Activity 3 Mock-Up Demonstration Approval Development Marketing, Website Website Website Design & Feedback & Marketing Des. & Dev. Development Launch Faculty Design Roll-Out Office Hours TEAM MEMBERS • Special THANK you to: . Maryland Department of Health . Centers for Disease Control . Health Quality Innovators . Think Research . University of Maryland Team & . All of You ….. 4 Gap Analysis Overview provided by Rachyl Fornaro, PharmD Candidate 2019 Respondent Demographics Director of Nursing Nursing Home 3% Administrator 3% Medical Director Director/Assistant 1% Director of Clinical Services 4% Nurse Practitioner Infection Prevention 6% and Control Officer Other 45% 7% Pharmacist 31% Respondent Demographics: -

Guidelines on Urological Infections
Guidelines on Urological Infections M. Grabe (Chairman), T.E. Bjerklund-Johansen, H. Botto, M. Çek, K.G. Naber, P. Tenke, F. Wagenlehner © European Association of Urology 2010 TABLE OF CONTENTS PAGE 1. INTRODUCTION 7 1.1 Pathogenesis of urinary tract infections 7 1.2 Microbiological and other laboratory findings 7 1.3 Classification of urological infections 8 1.4 Aim of guidelines 8 1.5 Methods 9 1.6 Level of evidence and grade of guideline recommendations 9 1.7 References 9 2. UNCOMPLICATED URINARY TRACT INFECTIONS IN ADULTS 11 2.1 Definition 11 2.1.1 Aetiological spectrum 11 2.2 Acute uncomplicated cystitis in premenopausal, non-pregnant women 11 2.2.1 Diagnosis 11 2.2.1.1 Clinical diagnosis 11 2.2.1.2 Laboratory diagnosis 11 2.2.2 Therapy 11 2.2.3 Follow up 12 2.3 Acute uncomplicated pyelonephritis in premenopausal, non-pregnant women 12 2.3.1 Diagnosis 12 2.3.1.1 Clinical diagnosis 12 2.3.1.2 Laboratory diagnosis 12 2.3.1.3 Imaging diagnosis 13 2.3.2 Therapy 13 2.3.2.1 Mild and moderate cases of acute uncomplicated pyelonephritis 13 2.3.2.2 Severe cases of acute uncomplicated pyelonephritis 13 2.3.3 Follow-up 14 2.4 Recurrent (uncomplicated) UTIs in women 16 2.4.1 Diagnosis 16 2.4.2 Prevention 16 2.4.2.1 Antimicrobial prophylaxis 16 2.4.2.2 Immunoactive prophylaxis 16 2.4.2.3 Prophylaxis with probiotics 17 2.4.2.4 Prophylaxis with cranberry 17 2.5 Urinary tract infections in pregnancy 17 2.5.1 Definition of significant bacteriuria 17 2.5.2 Screening 17 2.5.3 Treatment of asymptomatic bacteriuria 17 2.5.4 Duration of therapy 18 2.5.5 Follow-up 18 2.5.6 Prophylaxis 18 2.5.7 Treatment of pyelonephritis 18 2.5.8 Complicated UTI 18 2.6 UTIs in postmenopausal women 18 2.6.1 Risk factors 18 2.6.2 Diagnosis 18 2.6.3 Treatment 18 2.7 Acute uncomplicated UTIs in young men 19 2.7.1 Men with acute uncomplicated UTI 19 2.7.2 Men with UTI and concomitant prostate infection 19 2.8 Asymptomatic bacteriuria 19 2.8.1 Diagnosis 19 2.8.2 Screening 19 2.9 References 26 3. -

Guidelines for Treatment of Intra-Abdominal Infections in Adults
GUIDELINES FOR TREATMENT OF INTRA-ABDOMINAL INFECTIONS IN ADULTS Table of Contents Appendicitis Cholangitis & Cholecystitis Diverticulitis Esophageal Perforation Secondary Peritonitis Tertiary Peritonitis (Infection associated with perforation or spillage of GI pathogens into (Persistent infection associated with recurring GI perforation and/or the peritoneal cavity) anastomotic leakage after initial treatment for secondary peritonitis) Spontaneous Bacterial Peritonitis (SBP) Treatment and Prophylaxis Pancreatitis Footnotes References Table of Contents Appendicitis Empiric Therapy Duration Community Acquired, No Severe Sepsis/Shock Non-perforated: 1st line: Discontinue after appendectomy. If no appendectomy performed a 10-day duration is Cefuroxime* 1.5 g IV q8h recommended ref1 + Metronidazole 500 mg PO/IV q8h High-risk allergy3/contraindications4 to beta-lactams: Perforated: Ciprofloxacin* 400 mg IV q8h 4 full days after source control ref 3 + Metronidazole 500 mg PO/IV q8h Duration of therapy may be extended with inadequate source control or persistent Community Acquired with Severe Sepsis/Shock OR MDR-GNR Risk: clinical symptoms or signs of infection. st 1 line: Piperacillin-tazobactam*4.5 g IV q6h Patients with bacteremia: 2 Low/medium-risk allergy to penicillins: 7-14 days Cefepime* 2 g IV q8h + Metronidazole 500 mg PO/IV q8h For patients with secondary gram-negative bacteremia, a 7-day duration of IV therapy Consider the addition of vancomycin to cefepime for Enterococcus (or oral quinolone at discharge) may be appropriate ref5