Cephalosporins Can Be Prescribed Safely for Penicillin-Allergic Patients ▲
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Cefditoren Pivoxil) Tablets 200 and 400 Mg
SPECTRACEF® (cefditoren pivoxil) Tablets 200 and 400 mg. To reduce the development of drug-resistant bacteria and maintain the effectiveness of SPECTRACEF® and other antibacterial drugs, SPECTRACEF® should be used only to treat infections that are proven or strongly suspected to be caused by bacteria. DESCRIPTION SPECTRACEF® tablets contain cefditoren pivoxil, a semi-synthetic cephalosporin antibiotic for oral administration. It is a prodrug which is hydrolyzed by esterases during absorption, and the drug is distributed in the circulating blood as active cefditoren. Chemically, cefditoren pivoxil is (-)-(6R,7R)-2,2-dimethylpropionyloxymethyl 7-[(Z)-2-(2-aminothiazol-4-yl)-2-methoxy iminoacetamido]-3-[(Z)-2-(4-methylthiazol-5-yl)ethenyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate. The empirical formula is C25H28N6O7S3 and the molecular weight is 620.73. The structural formula of cefditoren pivoxil is shown below: cefditoren pivoxil The amorphous form of cefditoren pivoxil developed for clinical use is a light yellow powder. It is freely soluble in dilute hydrochloric acid and soluble at levels equal to 6.06 mg/mL in ethanol and <0.1 mg/mL in water. SPECTRACEF® (cefditoren pivoxil) tablets contain 200 mg or 400 mg of cefditoren as cefditoren pivoxil and the following inactive ingredients: croscarmellose sodium, D-mannitol, hydroxypropyl cellulose, hypromellose, magnesium stearate, sodium caseinate (a milk protein), and sodium tripolyphosphate. The tablet coating contains carnauba wax, hypromellose, polyethylene glycol, and titanium dioxide. Tablets are printed with ink containing D&C Red No. 27, FD&C Blue No. 1, propylene glycol, and shellac. CLINICAL PHARMACOLOGY Pharmacokinetics Absorption Oral Bioavailability Following oral administration, cefditoren pivoxil is absorbed from the gastrointestinal tract and hydrolyzed to cefditoren by esterases. -

Medical Review(S) Clinical Review
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 200327 MEDICAL REVIEW(S) CLINICAL REVIEW Application Type NDA Application Number(s) 200327 Priority or Standard Standard Submit Date(s) December 29, 2009 Received Date(s) December 30, 2009 PDUFA Goal Date October 30, 2010 Division / Office Division of Anti-Infective and Ophthalmology Products Office of Antimicrobial Products Reviewer Name(s) Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD Review Completion October 29, 2010 Date Established Name Ceftaroline fosamil for injection (Proposed) Trade Name Teflaro Therapeutic Class Cephalosporin; ß-lactams Applicant Cerexa, Inc. Forest Laboratories, Inc. Formulation(s) 400 mg/vial and 600 mg/vial Intravenous Dosing Regimen 600 mg every 12 hours by IV infusion Indication(s) Acute Bacterial Skin and Skin Structure Infection (ABSSSI); Community-acquired Bacterial Pneumonia (CABP) Intended Population(s) Adults ≥ 18 years of age Template Version: March 6, 2009 Reference ID: 2857265 Clinical Review Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD NDA 200327: Teflaro (ceftaroline fosamil) Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT ......................................... 9 1.1 Recommendation on Regulatory Action ........................................................... 10 1.2 Risk Benefit Assessment.................................................................................. 10 1.3 Recommendations for Postmarketing Risk Evaluation and Mitigation Strategies ........................................................................................................................ -

“Ceftriaxone– Sulbactam–EDTA” and Various Antibiotics Against Gram
ORIGINAL ARTICLE A Comparative In Vitro Sensitivity Study of “Ceftriaxone– Sulbactam–EDTA” and Various Antibiotics against Gram- negative Bacterial Isolates from Intensive Care Unit Sweta Singh1, Chinmoy Sahu2, Sangram Singh Patel3, Abhay Singh4, Nidhi Yaduvanshi5 ABSTRACT Introduction: A rapid increase in multidrug-resistant (MDR) strains is being seen across the globe especially in the Southeast Asian region, including India. Carbapenems and colistin form the mainstay of treatment against gram-negative pathogens, especially extended-spectrum beta-lactamase (ESBL)- and metallo-beta-lactamse (MBL)-producing isolates. However, due to increased resistance to carbapenems and toxicity of colistin, especially in intensive care units (ICUs), carbapenem-sparing antibiotics like ceftriaxone–sulbactam–EDTA (CSE) combination needs to be evaluated. Materials and methods: Bacterial isolates cultured from various clinical samples from all ICUs for a period of 9 months were evaluated. Bacterial identification was performed by matrix assisted laser desorption ionization time of flight mass spectrometry (MALDI-TOF MS) and antibiotic susceptibility testing were performed by disk diffusion and E test method. Antibiogram of various antibiotics was noted. Extended-spectrum beta-lactamase- and MBL-producing bacteria were identified by phenotypic methods. Antibiotic sensitivity results of CSE were compared with the comparator drugs like colistin, carbapenems, and tigecycline in Enterobacteriaceae, Acinetobacter spp., and Pseudomonas spp. along with ESBL and MBL producers. Results: A total of 2,760 samples of blood, cerebrospinal fluid (CSF), respiratory samples, tissue, and pus were collected from ICUs with maximum isolates from pus (37%) followed by respiratory samples (31%) and blood (27%). Escherichia coli and Klebsiella pneumoniae were the predominant gram-negative pathogens accounting for 56% of the isolates followed by Acinetobacter spp. -

Surgical Decolonization and Prophylaxis
Surgical Decolonization and Prophylaxis SurgicalSurgical Decolonization Decolonization and Prophylaxis and Prophylaxis DECOLONIZATION DECOLONIZATION Nasal Screening Result Recommended Intervention Nasal Screening Result Recommended Intervention MRSA Negative • No decolonization required MSSAMRSA NegativeNegative • No decolonization required MSSA PositiveNegative • Intranasal mupirocin twice daily x 5 days MSSA Positive • Intranasal mupirocin twice daily x 5 days MRSA Positive • Intranasal mupirocin twice daily x 5 days, MRSA Positive AND• Intranasal mupirocin twice daily x 5 days, •ANDChlorhexidine bathing one day prior to surgery ANTIMICROBIAL• ChlorhexidinePROPHYLAXISbathing one day prior to surgery ANTIMICROBIAL PROPHYLAXIS CLINICAL CONSIDERATIONS • Preoperative dose-timing CLINICAL CONSIDERATIONS • PreoperativeWithin 60 minutesdose-timing of surgical incision WithinExceptions: 60 minutes vancomycin of surgicaland fluoroquinolones incision within 120 minutes of surgical incisionExceptions: vancomycin and fluoroquinolones within 120 minutes of surgical • Weightincision-based dosing • WeightCefazolin-based: 2 gm dosingfor patients <120 kg, and 3 gm for patients ≥120 kg Vancomycin:Cefazolin: 2 gm usefor ABW patients <120 kg, and 3 gm for patients ≥120 kg Gentamicin:Vancomycin: use use ABW ABW unless ABW is >120% of their IBW, in which case use AdjBWGentamicin:(see below use ABW for equation)unless ABW is >120% of their IBW, in which case use • DurationAdjBW of(see prophylaxis below for equation) • DurationA single of dose, prophylaxis -

Use of Ceftaroline Fosamil in Children: Review of Current Knowledge and Its Application
Infect Dis Ther (2017) 6:57–67 DOI 10.1007/s40121-016-0144-8 REVIEW Use of Ceftaroline Fosamil in Children: Review of Current Knowledge and its Application Juwon Yim . Leah M. Molloy . Jason G. Newland Received: November 10, 2016 / Published online: December 30, 2016 Ó The Author(s) 2016. This article is published with open access at Springerlink.com ABSTRACT infections, CABP caused by penicillin- and ceftriaxone-resistant S. pneumoniae and Ceftaroline is a novel cephalosporin recently resistant Gram-positive infections that fail approved in children for treatment of acute first-line antimicrobial agents. However, bacterial skin and soft tissue infections and limited data are available on tolerability in community-acquired bacterial pneumonia neonates and infants younger than 2 months (CABP) caused by methicillin-resistant of age, and on pharmacokinetic characteristics Staphylococcus aureus, Streptococcus pneumoniae in children with chronic medical conditions and other susceptible bacteria. With a favorable and those with invasive, complicated tolerability profile and efficacy proven in infections. In this review, the microbiological pediatric patients and excellent in vitro profile of ceftaroline, its mechanism of action, activity against resistant Gram-positive and and pharmacokinetic profile will be presented. Gram-negative bacteria, ceftaroline may serve Additionally, clinical evidence for use in as a therapeutic option for polymicrobial pediatric patients and proposed place in therapy is discussed. Enhanced content To view enhanced content for this article go to http://www.medengine.com/Redeem/ 1F47F0601BB3F2DD. Keywords: Antibiotic resistance; Ceftaroline J. Yim (&) fosamil; Children; Methicillin-resistant St. John Hospital and Medical Center, Detroit, MI, Staphylococcus aureus; Streptococcus pneumoniae USA e-mail: [email protected] L. -

Cefoperazone Sodium
Disclaimer While these labels are the most current Drug Control Authority (DCA) approved versions of content, these are not necessarily reflective of final printed labeling currently in distribution LPD Title : Cefoperazone Sodium LPD Date : 07 February 2017 Country : Malaysia Reference : CDS version 5.0 dated 07 January 2017 Reason : Addition of adverse drug reactions (ADRs) Haemorrhage and Coagulopathy to Section 4.8 Undesirable effects, and also, addition of serious hemorrhage and coagulopathy to the warning about vitamin K deficiency in Section 4.4 Special warnings and precautions for use Addition of ADRs Anaphylactic reaction and Anaphylactic shock to Section 4.8 Undesirable effects Addition of ADR Dermatitis exfoliative to Section 4.8 Undesirable Effects and a warning statement on severe cutaneous adverse reactions (SCARs) to Section 4.4, Special warnings and precautions for use 1. NAME OF THE MEDICINAL PRODUCT CEFOBID 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Cefoperazone sodium is a semisynthetic broad-spectrum cephalosporin antibiotic for parenteral use only. Cefoperazone contains 34 mg sodium (1.5 mEq) per gram. Cefoperazone is a white crystalline powder which is freely soluble in water. The pH of a 25% aqueous solution is 5.0 to 6.5 and the solution is colorless to straw-yellow, depending on the concentration. The empirical formula is C25H26N9NaO8S2. 3. PHARMACEUTICAL FORM Cefoperazone sodium is available in vials containing 1 and 2 grams. Not For Distribution PFIZER CONFIDENTIAL Not all product strengths or pack sizes -

Empiric Antimicrobial Therapy for Diabetic Foot Infection
Empiric Antimicrobial Therapy for Diabetic Foot Infection (NB Provincial Health Authorities Anti-Infective Stewardship Committee, September 2019) Infection Severity Preferred Empiric Regimens Alternative Regimens Comments Mild Wound less than 4 weeks duration:d Wound less than 4 weeks duration:e • Outpatient management • Cellulitis less than 2 cm and • cephalexin 500 – 1000 mg PO q6h*,a OR • clindamycin 300 – 450 mg PO q6h (only if recommended ,a • cefadroxil 500 – 1000 mg PO q12h* severe delayed reaction to a beta-lactam) without involvement of deeper • Tailor regimen based on culture tissues and susceptibility results and True immediate allergy to a beta-lactam at MRSA Suspected: • Non-limb threatening patient response risk of cross reactivity with cephalexin or • doxycycline 200 mg PO for 1 dose then • No signs of sepsis cefadroxil: 100 mg PO q12h OR • cefuroxime 500 mg PO q8–12h*,b • sulfamethoxazole+trimethoprim 800+160 mg to 1600+320 mg PO q12h*,f Wound greater than 4 weeks duration:d Wound greater than 4 weeks duratione • amoxicillin+clavulanate 875/125 mg PO and MRSA suspected: q12h*,c OR • doxycycline 200 mg PO for 1 dose then • cefuroxime 500 mg PO q8–12h*,b AND 100 mg PO q12h AND metroNIDAZOLE metroNIDAZOLE 500 mg PO q12h 500 mg PO q12h OR • sulfamethoxazole+trimethoprim 800+160 mg to 1600/320 mg PO q12h*,f AND metroNIDAZOLE 500 mg PO q12h Moderate Wound less than 4 weeks duration:d Wound less than 4 weeks duration:e • Initial management with • Cellulitis greater than 2 cm or • ceFAZolin 2 g IV q8h*,b OR • levoFLOXacin 750 -

Cefalexin in the WHO Essential Medicines List for Children Reject
Reviewer No. 1: checklist for application of: Cefalexin In the WHO Essential Medicines List for Children (1) Have all important studies that you are aware of been included? Yes No 9 (2) Is there adequate evidence of efficacy for the proposed use? Yes 9 No (3) Is there evidence of efficacy in diverse settings and/or populations? Yes 9 No (4) Are there adverse effects of concern? Yes 9 No (5) Are there special requirements or training needed for safe/effective use? Yes No 9 (6) Is this product needed to meet the majority health needs of the population? Yes No 9 (7) Is the proposed dosage form registered by a stringent regulatory authority? Yes 9 No (8) What action do you propose for the Committee to take? Reject the application for inclusion of the following presentations of cefalexin: • Tablets/ capsules 250mg • Oral suspensions 125mg/5ml and 125mg/ml (9) Additional comment, if any. In order to identify any additional literature, the following broad and sensitive search was conducted using the PubMed Clinical Query application: (cephalexin OR cefalexin AND - 1 - pediatr*) AND ((clinical[Title/Abstract] AND trial[Title/Abstract]) OR clinical trials[MeSH Terms] OR clinical trial[Publication Type] OR random*[Title/Abstract] OR random allocation[MeSH Terms] OR therapeutic use[MeSH Subheading]) Only one small additional study was identified, which looked at the provision of prophylactic antibiotics in patients presenting to an urban children's hospital with trauma to the distal fingertip, requiring repair.1 In a prospective randomised control trial, 146 patients were enrolled, of which 69 were randomised to the no-antibiotic group, and 66 were randomised to the antibiotic (cefalexin) group. -
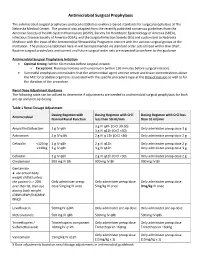
Antimicrobial Surgical Prophylaxis
Antimicrobial Surgical Prophylaxis The antimicrobial surgical prophylaxis protocol establishes evidence-based standards for surgical prophylaxis at The Nebraska Medical Center. The protocol was adapted from the recently published consensus guidelines from the American Society of Health-System Pharmacists (ASHP), Society for Healthcare Epidemiology of America (SHEA), Infectious Disease Society of America (IDSA), and the Surgical Infection Society (SIS) and customized to Nebraska Medicine with the input of the Antimicrobial Stewardship Program in concert with the various surgical groups at the institution. The protocol established here-in will be implemented via standard order sets utilized within One Chart. Routine surgical prophylaxis and current and future surgical order sets are expected to conform to this guidance. Antimicrobial Surgical Prophylaxis Initiation Optimal timing: Within 60 minutes before surgical incision o Exceptions: Fluoroquinolones and vancomycin (within 120 minutes before surgical incision) Successful prophylaxis necessitates that the antimicrobial agent achieve serum and tissue concentrations above the MIC for probable organisms associated with the specific procedure type at the time of incision as well as for the duration of the procedure. Renal Dose Adjustment Guidance The following table can be utilized to determine if adjustments are needed to antimicrobial surgical prophylaxis for both pre-op and post-op dosing. Table 1 Renal Dosage Adjustment Dosing Regimen with Dosing Regimen with CrCl Dosing Regimen with -

Product Monograph
Product Monograph PrORB-CEFUROXIME Cefuroxime Axetil Tablets, USP 250 mg and 500 mg cefuroxime/tablet Antibiotic Orbus Pharma Inc. Date of Preparation: February 25, 2009 20 Konrad Crescent Markham, Ontario Control #: 117041 L3R8T4 1 Product Monograph PrORB-CEFUROXIME Cefuroxime Axetil Tablets, USP 250 mg and 500 mg cefuroxime/tablet Antibiotic Actions and Clinical Pharmacology Cefuroxime axetil is an orally active prodrug of cefuroxime. After oral administration, cefuroxime axetil is absorbed from the gastrointestinal tract and rapidly hydrolyzed by nonspecific esterases in the intestinal mucosa and blood to release cefuroxime into the blood stream. Conversion to cefuroxime, the microbiologically active form, occurs rapidly. The inherent properties of cefuroxime are unaltered after its administration as cefuroxime axetil. Cefuroxime exerts its bactericidal effect by binding to an enzyme or enzymes referred to as penicillin-binding proteins (PBPs) involved in bacterial cell wall synthesis. This binding results in inhibition of bacterial cell wall synthesis and subsequent cell death. Specifically, cefuroxime shows high affinity for PBP 3, a primary target for cefuroxime in gram- negative organisms such as E. coli. Comparative Bioavailability Studies A two-way crossover, randomized, blinded, single-dose bioequivalence study was performed on 22 normal, healthy, non-smoking male subjects under fasting conditions. The rate and extent of absorption of cefuroxime axetil was measured and compared following a single oral dose (1 x 500 mg tablet) -

The Efficacy of Cefmetazole Against Pyelonephritis Caused by Extended
International Journal of Infectious Diseases 17 (2013) e159–e163 Contents lists available at SciVerse ScienceDirect International Journal of Infectious Diseases jou rnal homepage: www.elsevier.com/locate/ijid The efficacy of cefmetazole against pyelonephritis caused by extended-spectrum beta-lactamase-producing Enterobacteriaceae a, b c d a,e Asako Doi *, Toshihiko Shimada , Sohei Harada , Kentaro Iwata , Toru Kamiya a Department of Infectious Diseases, Rakuwakai Otowa Hospital, Otowachinji-cho 2, Yamashina-ku, Kyoto, Japan b Department of General Internal Medicine, Nara City Hospital, Nara, Japan c Department of Microbiology and Infectious Diseases, Toho University School of Medicine, Ota-ku, Tokyo, Japan d Division of Infectious Diseases, Kobe University Hospital, Chuo-ku, Kobe, Hyogo, Japan e Department of General Internal Medicine, Rakuwakai Otowa Hospital, Otowachinji-cho 2, Yamashina-ku, Kyoto, Japan A R T I C L E I N F O S U M M A R Y Article history: Objectives: Urinary tract infections (UTIs) caused by extended-spectrum beta-lactamase (ESBL)- Received 3 April 2012 producing Enterobacteriaceae are on the increase. Although cefmetazole is stable in vitro against the Accepted 26 September 2012 hydrolyzing activity of ESBLs, no clinical study has ever evaluated its role in infections caused by these Corresponding Editor: William Cameron, organisms. We therefore evaluated the efficacy of cefmetazole compared to carbapenems against Ottawa, Canada pyelonephritis caused by ESBL-producing Enterobacteriaceae. Methods: A retrospective chart review was conducted at a tertiary care hospital from August 2008 to July Keywords: 2010. Chart reviews were done for patients with ESBL-producing organisms in urine identified in the Extended-spectrum beta-lactamase (ESBL) microbiology database. -

Antimicrobial Stewardship Guidance
Antimicrobial Stewardship Guidance Federal Bureau of Prisons Clinical Practice Guidelines March 2013 Clinical guidelines are made available to the public for informational purposes only. The Federal Bureau of Prisons (BOP) does not warrant these guidelines for any other purpose, and assumes no responsibility for any injury or damage resulting from the reliance thereof. Proper medical practice necessitates that all cases are evaluated on an individual basis and that treatment decisions are patient-specific. Consult the BOP Clinical Practice Guidelines Web page to determine the date of the most recent update to this document: http://www.bop.gov/news/medresources.jsp Federal Bureau of Prisons Antimicrobial Stewardship Guidance Clinical Practice Guidelines March 2013 Table of Contents 1. Purpose ............................................................................................................................................. 3 2. Introduction ...................................................................................................................................... 3 3. Antimicrobial Stewardship in the BOP............................................................................................ 4 4. General Guidance for Diagnosis and Identifying Infection ............................................................. 5 Diagnosis of Specific Infections ........................................................................................................ 6 Upper Respiratory Infections (not otherwise specified) ..............................................................................