“Ceftriaxone– Sulbactam–EDTA” and Various Antibiotics Against Gram
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Medical Review(S) Clinical Review
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 200327 MEDICAL REVIEW(S) CLINICAL REVIEW Application Type NDA Application Number(s) 200327 Priority or Standard Standard Submit Date(s) December 29, 2009 Received Date(s) December 30, 2009 PDUFA Goal Date October 30, 2010 Division / Office Division of Anti-Infective and Ophthalmology Products Office of Antimicrobial Products Reviewer Name(s) Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD Review Completion October 29, 2010 Date Established Name Ceftaroline fosamil for injection (Proposed) Trade Name Teflaro Therapeutic Class Cephalosporin; ß-lactams Applicant Cerexa, Inc. Forest Laboratories, Inc. Formulation(s) 400 mg/vial and 600 mg/vial Intravenous Dosing Regimen 600 mg every 12 hours by IV infusion Indication(s) Acute Bacterial Skin and Skin Structure Infection (ABSSSI); Community-acquired Bacterial Pneumonia (CABP) Intended Population(s) Adults ≥ 18 years of age Template Version: March 6, 2009 Reference ID: 2857265 Clinical Review Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD NDA 200327: Teflaro (ceftaroline fosamil) Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT ......................................... 9 1.1 Recommendation on Regulatory Action ........................................................... 10 1.2 Risk Benefit Assessment.................................................................................. 10 1.3 Recommendations for Postmarketing Risk Evaluation and Mitigation Strategies ........................................................................................................................ -

Cefoperazone Sodium
Disclaimer While these labels are the most current Drug Control Authority (DCA) approved versions of content, these are not necessarily reflective of final printed labeling currently in distribution LPD Title : Cefoperazone Sodium LPD Date : 07 February 2017 Country : Malaysia Reference : CDS version 5.0 dated 07 January 2017 Reason : Addition of adverse drug reactions (ADRs) Haemorrhage and Coagulopathy to Section 4.8 Undesirable effects, and also, addition of serious hemorrhage and coagulopathy to the warning about vitamin K deficiency in Section 4.4 Special warnings and precautions for use Addition of ADRs Anaphylactic reaction and Anaphylactic shock to Section 4.8 Undesirable effects Addition of ADR Dermatitis exfoliative to Section 4.8 Undesirable Effects and a warning statement on severe cutaneous adverse reactions (SCARs) to Section 4.4, Special warnings and precautions for use 1. NAME OF THE MEDICINAL PRODUCT CEFOBID 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Cefoperazone sodium is a semisynthetic broad-spectrum cephalosporin antibiotic for parenteral use only. Cefoperazone contains 34 mg sodium (1.5 mEq) per gram. Cefoperazone is a white crystalline powder which is freely soluble in water. The pH of a 25% aqueous solution is 5.0 to 6.5 and the solution is colorless to straw-yellow, depending on the concentration. The empirical formula is C25H26N9NaO8S2. 3. PHARMACEUTICAL FORM Cefoperazone sodium is available in vials containing 1 and 2 grams. Not For Distribution PFIZER CONFIDENTIAL Not all product strengths or pack sizes -

Antimicrobial Susceptibility Among Colistin, Sulbactam, And
Hindawi Journal of Pathogens Volume 2018, Article ID 3893492, 5 pages https://doi.org/10.1155/2018/3893492 Research Article Antimicrobial Susceptibility among Colistin, Sulbactam, and Fosfomycin and a Synergism Study of Colistin in Combination with Sulbactam or Fosfomycin against Clinical Isolates of Carbapenem-Resistant Acinetobacter baumannii Sombat Leelasupasri,1 Wichai Santimaleeworagun ,2,3 and Tossawan Jitwasinkul3,4 1 Internal Medicine Unit, Phyathai II International Hospital, Bangkok, Tailand 2Department of Pharmacy, Faculty of Pharmacy, Silpakorn University, Nakhon Pathom, Tailand 3Antibiotic Optimization and Patient Care Project by Pharmaceutical Initiative for Resistant Bacteria and Infectious Diseases Working Group (PIRBIG), Silpakorn University, Nakhon Pathom, Tailand 4Department of Biopharmacy, Faculty of Pharmacy, Silpakorn University, Nakhon Pathom, Tailand Correspondence should be addressed to Wichai Santimaleeworagun; [email protected] Received 28 August 2017; Revised 9 December 2017; Accepted 25 December 2017; Published 18 January 2018 Academic Editor: Jose Yuste Copyright © 2018 Sombat Leelasupasri et al. Tis is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Tis in vitro study aimed to determine the activity of colistin plus sulbactam and colistin plus fosfomycin against carbapenem- resistant A. baumannii (CRAB). Fifeen clinical isolates were obtained from patients admitted to Phyathai II International Hospital, Bangkok, Tailand, from August 2014 to April 2015. Te antimicrobial susceptibilities of colistin, sulbactam, and fosfomycin were evaluated using the E-test or broth microdilution and the synergistic activity of the antibacterial combinations (colistin plus sulbactam or fosfomycin) was determined using the chequerboard method. Clonal relationships were explored using repetitive element palindromic- (REP-) PCR. -

Lactamase-Producing Antibiotic-Resistant Escherichia Coli and Klebsiella Pneumoniae in Ready-To-Eat Street Foods
antibiotics Article Prevalence and Characterization of Extended-Spectrum β-Lactamase-Producing Antibiotic-Resistant Escherichia coli and Klebsiella pneumoniae in Ready-to-Eat Street Foods Shobha Giri 1 , Vaishnavi Kudva 1, Kalidas Shetty 2 and Veena Shetty 1,* 1 Department of Microbiology, KS Hegde Medical Academy, Nitte (Deemed to be University), Mangalore 575018, India; [email protected] (S.G.); [email protected] (V.K.) 2 Department of Plant Sciences, North Dakota State University, Fargo, ND 58102, USA; [email protected] * Correspondence: [email protected] Abstract: As the global urban populations increase with rapid migration from rural areas, ready-to- eat (RTE) street foods are posing food safety challenges where street foods are prepared with less structured food safety guidelines in small and roadside outlets. The increased presence of extended- spectrum-β-lactamase (ESBL) producing bacteria in street foods is a significant risk for human health because of its epidemiological significance. Escherichia coli and Klebsiella pneumoniae have become important and dangerous foodborne pathogens globally for their relevance to antibiotic resistance. The present study was undertaken to evaluate the potential burden of antibiotic-resistant E. coli and K. pneumoniae contaminating RTE street foods and to assess the microbiological quality of foods in a Citation: Giri, S.; Kudva, V.; Shetty, typical emerging and growing urban suburb of India where RTE street foods are rapidly establishing K.; Shetty, V. Prevalence and with public health implications. A total of 100 RTE food samples were collected of which, 22.88% Characterization of were E. coli and 27.12% K. pneumoniae. The prevalence of ESBL-producing E. -

Activity of Imipenem, Meropenem, Cefepime, and Sulbactam in Combination with the Β-Lactamase Inhibitor LN-1-255 Against Acinetobacter Spp
antibiotics Article Activity of Imipenem, Meropenem, Cefepime, and Sulbactam in Combination with the β-Lactamase Inhibitor LN-1-255 against Acinetobacter spp. Cristina Lasarte-Monterrubio 1,† , Juan C. Vázquez-Ucha 1,† , Maria Maneiro 2, Jorge Arca-Suárez 1,*, Isaac Alonso 3, Paula Guijarro-Sánchez 1 , John D. Buynak 4, Germán Bou 1, Concepción González-Bello 2 and Alejandro Beceiro 1,* 1 Servicio de Microbiología, Instituto de Investigación Biomédica de A Coruña (INIBIC-CICA), Complejo Hospitalario Universitario A Coruña (CHUAC), As Xubias 84, 15006 A Coruña, Spain; [email protected] (C.L.-M.); [email protected] (J.C.V.-U.); [email protected] (P.G.-S.); [email protected] (G.B.) 2 Centro Singular de Investigación en Química Biolóxica e Materiais Moleculares (CIQUS), Departamento de Química Orgánica, Universidade de Santiago de Compostela, Jenaro de la Fuente s/n, 15782 Santiago de Compostela, Spain; [email protected] (M.M.); [email protected] (C.G.-B.) 3 Servicio de Microbiología, Hospital Provincial Pontevedra, Loureiro Crespo 2, 36002 Pontevedra, Spain; Citation: Lasarte-Monterrubio, C.; [email protected] 4 Vázquez-Ucha, J.C.; Maneiro, M.; Department of Chemistry, Southern Methodist University, Dallas, TX 75275, USA; [email protected] Arca-Suárez, J.; Alonso, I.; * Correspondence: [email protected] (J.A.-S.); [email protected] (A.B.); Tel.: +34-981-176-087 (J.A.-S.); +34-981-176-087 (A.B.) Guijarro-Sánchez, P.; Buynak, J.D.; † Cristina Lasarte-Monterrubio and Juan C. Vázquez-Ucha contributed equally to the study. Bou, G.; González-Bello, C.; Beceiro, A. -

In Vitro Susceptibilities of Escherichia Coli and Klebsiella Spp. To
Jpn. J. Infect. Dis., 60, 227-229, 2007 Short Communication In Vitro Susceptibilities of Escherichia coli and Klebsiella Spp. to Ampicillin-Sulbactam and Amoxicillin-Clavulanic Acid Birgul Kacmaz* and Nedim Sultan1 Department of Central Microbiology and 1Department of Microbiology, Faculty of Medicine, Gazi University, Ankara, Turkey (Received January 30, 2007. Accepted April 13, 2007) SUMMARY: Ampicillin-sulbactam (A/S) and amoxicillin-clavulanic acid (AUG) are thought to be equally efficacious clinically against the Enterobacteriaceae family. In this study, the in vitro activities of the A/S and AUG were evaluated and compared against Escherichia coli and Klebsiella spp. Antimicrobial susceptibility tests were performed by standard agar dilution and disc diffusion techniques according to the Clinical and Laboratory Standards Institute (CLSI). During the study period, 973 strains were isolated. Of the 973 bacteria isolated, 823 were E. coli and 150 Klebsiella spp. More organisms were found to be susceptible to AUG than A/S, regardless of the susceptibility testing methodology. The agar dilution results of the isolates that were found to be sensitive or resistant were also compatible with the disc diffusion results. However, some differences were seen in the agar dilution results of some isolates that were found to be intermediately resistant with disc diffusion. In E. coli isolates, 17 of the 76 AUG intermediately resistant isolates (by disc diffusion), and 17 of the 63 A/S intermediately resistant isolates (by disc diffusion) showed different resistant patterns by agar dilution. When the CLSI breakpoint criteria are applied it should be considered that AUG and A/S sensitivity in E. coli and Klebsiella spp. -

Antimicrobial Stewardship Guidance
Antimicrobial Stewardship Guidance Federal Bureau of Prisons Clinical Practice Guidelines March 2013 Clinical guidelines are made available to the public for informational purposes only. The Federal Bureau of Prisons (BOP) does not warrant these guidelines for any other purpose, and assumes no responsibility for any injury or damage resulting from the reliance thereof. Proper medical practice necessitates that all cases are evaluated on an individual basis and that treatment decisions are patient-specific. Consult the BOP Clinical Practice Guidelines Web page to determine the date of the most recent update to this document: http://www.bop.gov/news/medresources.jsp Federal Bureau of Prisons Antimicrobial Stewardship Guidance Clinical Practice Guidelines March 2013 Table of Contents 1. Purpose ............................................................................................................................................. 3 2. Introduction ...................................................................................................................................... 3 3. Antimicrobial Stewardship in the BOP............................................................................................ 4 4. General Guidance for Diagnosis and Identifying Infection ............................................................. 5 Diagnosis of Specific Infections ........................................................................................................ 6 Upper Respiratory Infections (not otherwise specified) .............................................................................. -
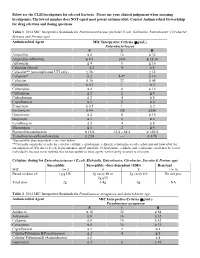
Below Are the CLSI Breakpoints for Selected Bacteria. Please Use Your Clinical Judgement When Assessing Breakpoints
Below are the CLSI breakpoints for selected bacteria. Please use your clinical judgement when assessing breakpoints. The lowest number does NOT equal most potent antimicrobial. Contact Antimicrobial Stewardship for drug selection and dosing questions. Table 1: 2014 MIC Interpretive Standards for Enterobacteriaceae (includes E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia and Proteus spp) Antimicrobial Agent MIC Interpretive Criteria (g/mL) Enterobacteriaceae S I R Ampicillin ≤ 8 16 ≥ 32 Ampicillin-sulbactam ≤ 8/4 16/8 ≥ 32/16 Aztreonam ≤ 4 8 ≥ 16 Cefazolin (blood) ≤ 2 4 ≥ 8 Cefazolin** (uncomplicated UTI only) ≤ 16 ≥ 32 Cefepime* ≤ 2 4-8* ≥ 16 Cefotetan ≤ 16 32 ≥ 64 Ceftaroline ≤ 0.5 1 ≥ 2 Ceftazidime ≤ 4 8 ≥ 16 Ceftriaxone ≤ 1 2 ≥ 4 Cefpodoxime ≤ 2 4 ≥ 8 Ciprofloxacin ≤ 1 2 ≥ 4 Ertapenem ≤ 0.5 1 ≥ 2 Fosfomycin ≤ 64 128 ≥256 Gentamicin ≤ 4 8 ≥ 16 Imipenem ≤ 1 2 ≥ 4 Levofloxacin ≤ 2 4 ≥ 8 Meropenem ≤ 1 2 ≥ 4 Piperacillin-tazobactam ≤ 16/4 32/4 – 64/4 ≥ 128/4 Trimethoprim-sulfamethoxazole ≤ 2/38 --- ≥ 4/76 *Susceptibile dose-dependent – see chart below **Cefazolin can predict results for cefaclor, cefdinir, cefpodoxime, cefprozil, cefuroxime axetil, cephalexin and loracarbef for uncomplicated UTIs due to E.coli, K.pneumoniae, and P.mirabilis. Cefpodoxime, cefinidir, and cefuroxime axetil may be tested individually because some isolated may be susceptible to these agents while testing resistant to cefazolin. Cefepime dosing for Enterobacteriaceae ( E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia & Proteus spp) Susceptible Susceptible –dose-dependent (SDD) Resistant MIC </= 2 4 8 >/= 16 Based on dose of: 1g q12h 1g every 8h or 2g every 8 h Do not give 2g q12 Total dose 2g 3-4g 6g NA Table 2: 2014 MIC Interpretive Standards for Pseudomonas aeruginosa and Acinetobacter spp. -

UNASYN® (Ampicillin Sodium/Sulbactam Sodium)
NDA 50-608/S-029 Page 3 UNASYN® (ampicillin sodium/sulbactam sodium) PHARMACY BULK PACKAGE NOT FOR DIRECT INFUSION To reduce the development of drug-resistant bacteria and maintain the effectiveness of UNASYN® and other antibacterial drugs, UNASYN should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. DESCRIPTION UNASYN is an injectable antibacterial combination consisting of the semisynthetic antibiotic ampicillin sodium and the beta-lactamase inhibitor sulbactam sodium for intravenous and intramuscular administration. Ampicillin sodium is derived from the penicillin nucleus, 6-aminopenicillanic acid. Chemically, it is monosodium (2S, 5R, 6R)-6-[(R)-2-amino-2-phenylacetamido]- 3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate and has a molecular weight of 371.39. Its chemical formula is C16H18N3NaO4S. The structural formula is: COONa O CH3 N CH O 3 NH S NH2 Sulbactam sodium is a derivative of the basic penicillin nucleus. Chemically, sulbactam sodium is sodium penicillinate sulfone; sodium (2S, 5R)-3,3-dimethyl-7-oxo-4-thia 1-azabicyclo [3.2.0] heptane-2-carboxylate 4,4-dioxide. Its chemical formula is C8H10NNaO5S with a molecular weight of 255.22. The structural formula is: NDA 50-608/S-029 Page 4 COONa CH3 O N CH3 S O O UNASYN, ampicillin sodium/sulbactam sodium parenteral combination, is available as a white to off-white dry powder for reconstitution. UNASYN dry powder is freely soluble in aqueous diluents to yield pale yellow to yellow solutions containing ampicillin sodium and sulbactam sodium equivalent to 250 mg ampicillin per mL and 125 mg sulbactam per mL. -

Consideration of Antibacterial Medicines As Part Of
Consideration of antibacterial medicines as part of the revisions to 2019 WHO Model List of Essential Medicines for adults (EML) and Model List of Essential Medicines for children (EMLc) Section 6.2 Antibacterials including Access, Watch and Reserve Lists of antibiotics This summary has been prepared by the Health Technologies and Pharmaceuticals (HTP) programme at the WHO Regional Office for Europe. It is intended to communicate changes to the 2019 WHO Model List of Essential Medicines for adults (EML) and Model List of Essential Medicines for children (EMLc) to national counterparts involved in the evidence-based selection of medicines for inclusion in national essential medicines lists (NEMLs), lists of medicines for inclusion in reimbursement programs, and medicine formularies for use in primary, secondary and tertiary care. This document does not replace the full report of the WHO Expert Committee on Selection and Use of Essential Medicines (see The selection and use of essential medicines: report of the WHO Expert Committee on Selection and Use of Essential Medicines, 2019 (including the 21st WHO Model List of Essential Medicines and the 7th WHO Model List of Essential Medicines for Children). Geneva: World Health Organization; 2019 (WHO Technical Report Series, No. 1021). Licence: CC BY-NC-SA 3.0 IGO: https://apps.who.int/iris/bitstream/handle/10665/330668/9789241210300-eng.pdf?ua=1) and Corrigenda (March 2020) – TRS1021 (https://www.who.int/medicines/publications/essentialmedicines/TRS1021_corrigenda_March2020. pdf?ua=1). Executive summary of the report: https://apps.who.int/iris/bitstream/handle/10665/325773/WHO- MVP-EMP-IAU-2019.05-eng.pdf?ua=1. -

Ampicillin/Sulbactam Vs. Cefoxitin for the Treatment of Pelvic Inflammatory Disease
Infectious Diseases in Obstetrics and Gynecology 5:319–325 (1997) © 1998 Wiley-Liss, Inc. Ampicillin/Sulbactam Vs. Cefoxitin for the Treatment of Pelvic Inflammatory Disease Joseph G. Jemsek* and Frank Harrison Department of Obstetrics and Gynecology, Charlotte Memorial Hospital, Charlotte, NC ABSTRACT Objective: The safety and efficacy of ampicillin plus sulbactam were compared with those of ce- foxitin in the treatment of women with pelvic inflammatory disease (PID). Methods: This single-site, randomized, prospective, third-party-blinded, comparative, parallel- treatment study enrolled 93 women with a diagnosis of PID. Patients were treated with either ampicillin/sulbactam (2 g/1 g, administered intravenously [IV], every 6 h) or cefoxitin (2 g, admin- istered IV, every 6 h) for a minimum of 12 doses. Patients with cultures positive for Chlamydia trachomatis also received concurrent oral or IV doxycycline (100 mg twice daily). Patients with cultures negative for C. trachomatis received prophylactic oral doxycycline (100 mg twice daily) for 10–14 days after treatment with either ampicillin/sulbactam or cefoxitin was completed. Results: Ninety-three patients were entered in the study: 47 in the ampicillin/sulbactam arm and 46 in the cefoxitin arm. All 93 patients were evaluable for safety; 61 (66%) were evaluable for efficacy. Demographic characteristics were similar for the groups. Of the 27 evaluable ampicillin/ sulbactam-treated patients, 67% experienced clinical cure, 30% improved, and 4% failed treatment. Respective values for the 34 cefoxitin-treated patients were 68%, 24%, and 9% (P = 0.67). Patho- gens were eradicated in 70% of the women given ampicillin/sulbactam vs. 56% of those who re- ceived cefoxitin (P = 0.64). -

Cephalosporins Can Be Prescribed Safely for Penicillin-Allergic Patients ▲
JFP_0206_AE_Pichichero.Final 1/23/06 1:26 PM Page 106 APPLIED EVIDENCE New research findings that are changing clinical practice Michael E. Pichichero, MD University of Rochester Cephalosporins can be Medical Center, Rochester, NY prescribed safely for penicillin-allergic patients Practice recommendations an allergic reaction to cephalosporins, ■ The widely quoted cross-allergy risk compared with the incidence of a primary of 10% between penicillin and (and unrelated) cephalosporin allergy. cephalosporins is a myth (A). Most people produce IgG and IgM antibodies in response to exposure to ■ Cephalothin, cephalexin, cefadroxil, penicillin1 that may cross-react with and cefazolin confer an increased risk cephalosporin antigens.2 The presence of of allergic reaction among patients these antibodies does not predict allergic, with penicillin allergy (B). IgE cross-sensitivity to a cephalosporin. ■ Cefprozil, cefuroxime, cefpodoxime, Even penicillin skin testing is generally not ceftazidime, and ceftriaxone do not predictive of cephalosporin allergy.3 increase risk of an allergic reaction (B). Reliably predicting cross-reactivity ndoubtedly you have patients who A comprehensive review of the evidence say they are allergic to penicillin shows that the attributable risk of a cross- U but have difficulty recalling details reactive allergic reaction varies and is of the reactions they experienced. To be strongest when the chemical side chain of safe, we often label these patients as peni- the specific cephalosporin is similar to that cillin-allergic without further questioning of penicillin or amoxicillin. and withhold not only penicillins but Administration of cephalothin, cepha- cephalosporins due to concerns about lexin, cefadroxil, and cefazolin in penicillin- potential cross-reactivity and resultant IgE- allergic patients is associated with a mediated, type I reactions.