National Treatment Guidelines for Antimicrobial Use in Infectious Diseases
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Defining Escherichia Coli As a Health-Promoting Microbe Against Intestinal Pseudomonas Aeruginosa
bioRxiv preprint doi: https://doi.org/10.1101/612606; this version posted April 17, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. Defining Escherichia coli as a health-promoting microbe against intestinal Pseudomonas aeruginosa Theodoulakis Christofi1, Stavria Panayidou1, Irini Dieronitou1, Christina Michael1 & Yiorgos Apidianakis1* 1Department of Biological Sciences, University of Cyprus, Nicosia, Cyprus *Corresponding author, email: [email protected] Abstract Gut microbiota acts as a barrier against intestinal pathogens, but species-specific protection of the host from infection remains relatively unexplored. Taking a Koch’s postulates approach in reverse to define health-promoting microbes we find that Escherichia coli naturally colonizes the gut of healthy mice, but it is depleted from the gut of antibiotic-treated mice, which become susceptible to intestinal colonization by Pseudomonas aeruginosa and concomitant mortality. Reintroduction of fecal bacteria and E. coli establishes a high titer of E. coli in the host intestine and increases defence against P. aeruginosa colonization and mortality. Moreover, diet is relevant in this process because high sugars or dietary fat favours E. coli fermentation to lactic acid and P. aeruginosa growth inhibition. To the contrary, low sugars allow P. aeruginosa to produce the oxidative agent pyocyanin that inhibits E. coli growth. Our results provide an explanation as to why P. aeruginosa doesn’t commonly infect the human gut, despite being a formidable microbe in lung and wound infections. -

Medical Review(S) Clinical Review
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 200327 MEDICAL REVIEW(S) CLINICAL REVIEW Application Type NDA Application Number(s) 200327 Priority or Standard Standard Submit Date(s) December 29, 2009 Received Date(s) December 30, 2009 PDUFA Goal Date October 30, 2010 Division / Office Division of Anti-Infective and Ophthalmology Products Office of Antimicrobial Products Reviewer Name(s) Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD Review Completion October 29, 2010 Date Established Name Ceftaroline fosamil for injection (Proposed) Trade Name Teflaro Therapeutic Class Cephalosporin; ß-lactams Applicant Cerexa, Inc. Forest Laboratories, Inc. Formulation(s) 400 mg/vial and 600 mg/vial Intravenous Dosing Regimen 600 mg every 12 hours by IV infusion Indication(s) Acute Bacterial Skin and Skin Structure Infection (ABSSSI); Community-acquired Bacterial Pneumonia (CABP) Intended Population(s) Adults ≥ 18 years of age Template Version: March 6, 2009 Reference ID: 2857265 Clinical Review Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD NDA 200327: Teflaro (ceftaroline fosamil) Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT ......................................... 9 1.1 Recommendation on Regulatory Action ........................................................... 10 1.2 Risk Benefit Assessment.................................................................................. 10 1.3 Recommendations for Postmarketing Risk Evaluation and Mitigation Strategies ........................................................................................................................ -

“Ceftriaxone– Sulbactam–EDTA” and Various Antibiotics Against Gram
ORIGINAL ARTICLE A Comparative In Vitro Sensitivity Study of “Ceftriaxone– Sulbactam–EDTA” and Various Antibiotics against Gram- negative Bacterial Isolates from Intensive Care Unit Sweta Singh1, Chinmoy Sahu2, Sangram Singh Patel3, Abhay Singh4, Nidhi Yaduvanshi5 ABSTRACT Introduction: A rapid increase in multidrug-resistant (MDR) strains is being seen across the globe especially in the Southeast Asian region, including India. Carbapenems and colistin form the mainstay of treatment against gram-negative pathogens, especially extended-spectrum beta-lactamase (ESBL)- and metallo-beta-lactamse (MBL)-producing isolates. However, due to increased resistance to carbapenems and toxicity of colistin, especially in intensive care units (ICUs), carbapenem-sparing antibiotics like ceftriaxone–sulbactam–EDTA (CSE) combination needs to be evaluated. Materials and methods: Bacterial isolates cultured from various clinical samples from all ICUs for a period of 9 months were evaluated. Bacterial identification was performed by matrix assisted laser desorption ionization time of flight mass spectrometry (MALDI-TOF MS) and antibiotic susceptibility testing were performed by disk diffusion and E test method. Antibiogram of various antibiotics was noted. Extended-spectrum beta-lactamase- and MBL-producing bacteria were identified by phenotypic methods. Antibiotic sensitivity results of CSE were compared with the comparator drugs like colistin, carbapenems, and tigecycline in Enterobacteriaceae, Acinetobacter spp., and Pseudomonas spp. along with ESBL and MBL producers. Results: A total of 2,760 samples of blood, cerebrospinal fluid (CSF), respiratory samples, tissue, and pus were collected from ICUs with maximum isolates from pus (37%) followed by respiratory samples (31%) and blood (27%). Escherichia coli and Klebsiella pneumoniae were the predominant gram-negative pathogens accounting for 56% of the isolates followed by Acinetobacter spp. -

Lung Infections Aeruginosa Pseudomonas Hypersusceptibility
TLRs 2 and 4 Are Not Involved in Hypersusceptibility to Acute Pseudomonas aeruginosa Lung Infections This information is current as Reuben Ramphal, Viviane Balloy, Michel Huerre, Mustapha of September 29, 2021. Si-Tahar and Michel Chignard J Immunol 2005; 175:3927-3934; ; doi: 10.4049/jimmunol.175.6.3927 http://www.jimmunol.org/content/175/6/3927 Downloaded from References This article cites 51 articles, 24 of which you can access for free at: http://www.jimmunol.org/content/175/6/3927.full#ref-list-1 http://www.jimmunol.org/ Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication by guest on September 29, 2021 *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2005 by The American Association of Immunologists All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology TLRs 2 and 4 Are Not Involved in Hypersusceptibility to Acute Pseudomonas aeruginosa Lung Infections1 Reuben Ramphal,* Viviane Balloy,† Michel Huerre,‡ Mustapha Si-Tahar,† and Michel Chignard2† TLRs are implicated in defense against microorganisms. Animal models have demonstrated that the susceptibility to a number of Gram-negative pathogens is linked to TLR4, and thus LPS of many Gram-negative bacteria have been implicated as virulence factors. -

Cefoperazone Sodium
Disclaimer While these labels are the most current Drug Control Authority (DCA) approved versions of content, these are not necessarily reflective of final printed labeling currently in distribution LPD Title : Cefoperazone Sodium LPD Date : 07 February 2017 Country : Malaysia Reference : CDS version 5.0 dated 07 January 2017 Reason : Addition of adverse drug reactions (ADRs) Haemorrhage and Coagulopathy to Section 4.8 Undesirable effects, and also, addition of serious hemorrhage and coagulopathy to the warning about vitamin K deficiency in Section 4.4 Special warnings and precautions for use Addition of ADRs Anaphylactic reaction and Anaphylactic shock to Section 4.8 Undesirable effects Addition of ADR Dermatitis exfoliative to Section 4.8 Undesirable Effects and a warning statement on severe cutaneous adverse reactions (SCARs) to Section 4.4, Special warnings and precautions for use 1. NAME OF THE MEDICINAL PRODUCT CEFOBID 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Cefoperazone sodium is a semisynthetic broad-spectrum cephalosporin antibiotic for parenteral use only. Cefoperazone contains 34 mg sodium (1.5 mEq) per gram. Cefoperazone is a white crystalline powder which is freely soluble in water. The pH of a 25% aqueous solution is 5.0 to 6.5 and the solution is colorless to straw-yellow, depending on the concentration. The empirical formula is C25H26N9NaO8S2. 3. PHARMACEUTICAL FORM Cefoperazone sodium is available in vials containing 1 and 2 grams. Not For Distribution PFIZER CONFIDENTIAL Not all product strengths or pack sizes -

Folic Acid Antagonists: Antimicrobial and Immunomodulating Mechanisms and Applications
International Journal of Molecular Sciences Review Folic Acid Antagonists: Antimicrobial and Immunomodulating Mechanisms and Applications Daniel Fernández-Villa 1, Maria Rosa Aguilar 1,2 and Luis Rojo 1,2,* 1 Instituto de Ciencia y Tecnología de Polímeros, Consejo Superior de Investigaciones Científicas, CSIC, 28006 Madrid, Spain; [email protected] (D.F.-V.); [email protected] (M.R.A.) 2 Consorcio Centro de Investigación Biomédica en Red de Bioingeniería, Biomateriales y Nanomedicina, 28029 Madrid, Spain * Correspondence: [email protected]; Tel.: +34-915-622-900 Received: 18 September 2019; Accepted: 7 October 2019; Published: 9 October 2019 Abstract: Bacterial, protozoan and other microbial infections share an accelerated metabolic rate. In order to ensure a proper functioning of cell replication and proteins and nucleic acids synthesis processes, folate metabolism rate is also increased in these cases. For this reason, folic acid antagonists have been used since their discovery to treat different kinds of microbial infections, taking advantage of this metabolic difference when compared with human cells. However, resistances to these compounds have emerged since then and only combined therapies are currently used in clinic. In addition, some of these compounds have been found to have an immunomodulatory behavior that allows clinicians using them as anti-inflammatory or immunosuppressive drugs. Therefore, the aim of this review is to provide an updated state-of-the-art on the use of antifolates as antibacterial and immunomodulating agents in the clinical setting, as well as to present their action mechanisms and currently investigated biomedical applications. Keywords: folic acid antagonists; antifolates; antibiotics; antibacterials; immunomodulation; sulfonamides; antimalarial 1. -

Pyrimethamine
Canterbury District Health Board Neonatal Services PYRIMETHAMINE Trade Name Daraprim (GlaxoSmithKlyne) Class Antiparasitic/antimalarial agent Mechanism of Action Inhibits parasitic dihydrofolate reductase resulting in inhibition of tetrahydrofolic acid synthesis Indications Congenital toxoplasmosis (Used in combination with sulfadiazine) Contraindications Known megaloblastic anaemia Supplied As Pyrimethamine suspension 1mg/mL (prepared by pharmacy) Dilution None required Dosage 1mg/kg/dose If the patient has difficulty tolerating pyrimathamine give each dose with a feed to reduce the incidence of vomiting Interval Once daily for the first 6 months, then Three times a week for the second 6 months Administration Oral Compatible With N/A Incompatible With N/A Interactions Pyrimethamine reduces the anti-epileptic effect of phenytoin Antifolate effects are increased when pyrimethamine is given in combination with sulphonamides, trimethoprim, zidovudine or methotrexate. Folic Acid may reduce the anti-parasitic effect of pyrimethamine and also increase the risk of pyrimethamine induced bone marrow suppression , for these reasons folinic acid is used to counteract the antifolate effect of pyrimethamine. Avoid brands of sunblock that contain PABA as this may reduce the effectiveness of sulfadiazine. Monitoring Full blood count Stability 30 days at 2 – 8 oC Storage In the fridge Adverse Reactions Skin rash (including Stevens Johnson Syndrome), nausea, vomiting, diarrhoea, blood dyscrasias, raised AST, sensory neuropathy Metabolism 15-40% metabolised by liver, approx 60% of dose excreted as Pyrimethamine Printed copies are not controlled and may not be the current version in use Ref.236799 Authorised by: Clinical Director Neonatal Page 1 of 2 March 2016 Canterbury District Health Board Neonatal Services unchanged drug by kidneys. -

Efficacy and Tolerability of Quinacrine Monotherapy and Albendazole Plus Chloroquine Combination Therapy in Nitroimidazole-Refractory Giardiasis: a Tropnet Study
Klinik für Infektiologie & Spitalhygiene Efficacy and tolerability of quinacrine monotherapy and albendazole plus chloroquine combination therapy in nitroimidazole-refractory giardiasis: a TropNet study Andreas Neumayr, Mirjam Schunk, Caroline Theunissen, Marjan Van Esbroeck, Matthieu Mechain, Manuel Jesús Soriano Pérez, Kristine Mørch, Peter Sothmann, Esther Künzli, Camilla Rothe, Emmanuel Bottieau Journal Club 01.03.21 Andreas Neumayr Background on giardia treatment: • 1st-line treatment: 5-nitroimidazoles: metronidazole (1957), tinidazole, ornidazole, secnidazole • cure rate of 5NIs in 1st-line treatment: ~90% • in the last decade, an increase of 5NI-refractory giardia cases has been observed in travel medicine clinics across Europe: Hospital for Tropical Diseases, London: 2008: 15% --> 2013: 40% 70% of 5NI-refractory cases imported from India • 2nd-line treatment: effectiveness of a 2nd round with a 5NI: ~17% alternative drugs: albendazole, mebendazole, nitazoxanide, quinacrine, furazolidone, chloroquine, paromomycin 2012 TropNet member survey: 53 centres use 39 different treatment regimens, consisting of 7 different drugs in mono- or combination-therapy in various dosages and durations JC 01.03.21 Nabarro LE et al. Clin Microbiol Infect. 2015;21:791-6. • by 2013, there were only 13 reports of 2nd-line therapy for giardiasis (8 case series, 5 individual case reports): n=110 Cure rates Albendazole 6/32 18.7% Paromomycin 5/17 29.4% Nitazoxanide 2/5 40.0% Albendazole + 5-NI 42/53 79.2% Quinacrine 19/21 90.5% Quinacrine + 5-NI 14/14 100% Quinacrine + Paromomycin 2/2 100% • 2013: TropNet "GiardiaREF" study kick-off: Study on efficacy and tolerability of two 2nd-line regimens in nitroimidazole-refractory giardiasis: Quinacrine JC 01.03.21 Meltzer E et al. -

Data on Before and After the Traceability System of Veterinary Antimicrobial Prescriptions in Small Animals at the University Veterinary Teaching Hospital of Naples
animals Article Data on before and after the Traceability System of Veterinary Antimicrobial Prescriptions in Small Animals at the University Veterinary Teaching Hospital of Naples Claudia Chirollo , Francesca Paola Nocera , Diego Piantedosi, Gerardo Fatone , Giovanni Della Valle, Luisa De Martino * and Laura Cortese Department of Veterinary Medicine and Animal Productions, University of Naples, “Federico II”, Via Delpino 1, 80137 Naples, Italy; [email protected] (C.C.); [email protected] (F.P.N.); [email protected] (D.P.); [email protected] (G.F.); [email protected] (G.D.V.); [email protected] (L.C.) * Correspondence: [email protected]; Tel.: +39-081-253-6180 Simple Summary: Veterinary electronic prescription (VEP) is mandatory by law, dated 20 November 2017, No. 167 (European Law 2017) Article 3, and has been implemented in Italy since April 2019. In this study, the consumption of antimicrobials before and after the mandatory use of VEP was analyzed at the Italian University Veterinary Teaching Hospital of Naples in order to understand how the traceability of antimicrobials influences veterinary prescriptions. The applicability and utility of VEP may present an effective surveillance strategy able to limit both the improper use of Citation: Chirollo, C.; Nocera, F.P.; antimicrobials and the spread of multidrug-resistant pathogens, which have become a worrying Piantedosi, D.; Fatone, G.; Della Valle, threat both in veterinary and human medicine. G.; De Martino, L.; Cortese, L. Data on before and after the Traceability Abstract: Over recent decades, antimicrobial resistance has been considered one of the most relevant System of Veterinary Antimicrobial issues of public health. -

Jos Journal 2
POST-OPERATIVE AUDIT OF G6PD-DEFICIENT MALE CHILDREN WITH OBSTRUCTIVE ADENOTONSILLAR ENLARGEMENT AT UNIVERSITY COLLEGE HOSPITAL, IBADAN, NIGERIA. John EN1, Totyen EL1, Jacob N2, Nwaorgu OGB1 1 .Department of ENT/Head and Neck Surgery, University College Hospital, Ibadan, Nigeria 2. Department of paediatrics, University College Hospital, Ibadan, Nigeria All correspondences and request for reprint to Dr John EN, Department of ENT/Head and Neck Surgery, University College Hospital, Ibadan, Nigeria Email: [email protected] Telephone: +2348036240109 Abstract Background: G6PD deficiency ranks among the commonest hereditary enzyme deficiency worldwide and notable as a predisposing condition to haemolyticcrises. The fear of possible untoward effects is often expressed by parents of G6PD deficient male children scheduled for surgery after obtaining an informed and understood consent. The parental perception of obstructive adenotonsillar enlargement in this condition was also appraised. Methods: A retrospective chart review of all G6PD deficient male children between ages 1 to 7years who had adenotonsillectomy over a 3year period at University college Hospital, Ibadan, Nigeria. Results: The patients comprised of 22 G6PD deficient male children diagnosed shortly after birth upon development of neonatal jaundice. Fifteen(68.2%) and 6(27.3%) of the patients subsequently developed episodes of drug- induced haemolysis and non-haemolytic drug reactions prior to undergoing adenotonsillectomy by the otolaryngologists. None of the patients was observed to develop haemolytic crises up to 2weeks post-adenotonsillectomy. From the parental perception and responses in the follow-up period,all 22(100%) patient had resolution of noisy breathing, 20(91%) had improvement of snoring and apnoeic spells. Only 15 (68%) were reported to stop mouth-breathing. -

Severe Sepsis and Septic Shock Antibiotic Guide
Stanford Health Issue Date: 05/2017 Stanford Antimicrobial Safety and Sustainability Program Severe Sepsis and Septic Shock Antibiotic Guide Table 1: Antibiotic selection options for healthcare associated and/or immunocompromised patients • Healthcare associated: intravenous therapy, wound care, or intravenous chemotherapy within the prior 30 days, residence in a nursing home or other long-term care facility, hospitalization in an acute care hospital for two or more days within the prior 90 days, attendance at a hospital or hemodialysis clinic within the prior 30 days • Immunocompromised: Receiving chemotherapy, known systemic cancer not in remission, ANC <500, severe cell-mediated immune deficiency Table 2: Antibiotic selection options for community acquired, immunocompetent patients Table 3: Antibiotic selection options for patients with simple sepsis, community acquired, immunocompetent patients requiring hospitalization. Risk Factors for Select Organisms P. aeruginosa MRSA Invasive Candidiasis VRE (and other resistant GNR) Community acquired: • Known colonization with MDROs • Central venous catheter • Liver transplant • Prior IV antibiotics within 90 day • Recent MRSA infection • Broad-spectrum antibiotics • Known colonization • Known colonization with MDROs • Known MRSA colonization • + 1 of the following risk factors: • Prolonged broad antibacterial • Skin & Skin Structure and/or IV access site: ♦ Parenteral nutrition therapy Hospital acquired: ♦ Purulence ♦ Dialysis • Prolonged profound • Prior IV antibiotics within 90 days ♦ Abscess -
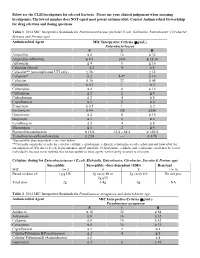
Below Are the CLSI Breakpoints for Selected Bacteria. Please Use Your Clinical Judgement When Assessing Breakpoints
Below are the CLSI breakpoints for selected bacteria. Please use your clinical judgement when assessing breakpoints. The lowest number does NOT equal most potent antimicrobial. Contact Antimicrobial Stewardship for drug selection and dosing questions. Table 1: 2014 MIC Interpretive Standards for Enterobacteriaceae (includes E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia and Proteus spp) Antimicrobial Agent MIC Interpretive Criteria (g/mL) Enterobacteriaceae S I R Ampicillin ≤ 8 16 ≥ 32 Ampicillin-sulbactam ≤ 8/4 16/8 ≥ 32/16 Aztreonam ≤ 4 8 ≥ 16 Cefazolin (blood) ≤ 2 4 ≥ 8 Cefazolin** (uncomplicated UTI only) ≤ 16 ≥ 32 Cefepime* ≤ 2 4-8* ≥ 16 Cefotetan ≤ 16 32 ≥ 64 Ceftaroline ≤ 0.5 1 ≥ 2 Ceftazidime ≤ 4 8 ≥ 16 Ceftriaxone ≤ 1 2 ≥ 4 Cefpodoxime ≤ 2 4 ≥ 8 Ciprofloxacin ≤ 1 2 ≥ 4 Ertapenem ≤ 0.5 1 ≥ 2 Fosfomycin ≤ 64 128 ≥256 Gentamicin ≤ 4 8 ≥ 16 Imipenem ≤ 1 2 ≥ 4 Levofloxacin ≤ 2 4 ≥ 8 Meropenem ≤ 1 2 ≥ 4 Piperacillin-tazobactam ≤ 16/4 32/4 – 64/4 ≥ 128/4 Trimethoprim-sulfamethoxazole ≤ 2/38 --- ≥ 4/76 *Susceptibile dose-dependent – see chart below **Cefazolin can predict results for cefaclor, cefdinir, cefpodoxime, cefprozil, cefuroxime axetil, cephalexin and loracarbef for uncomplicated UTIs due to E.coli, K.pneumoniae, and P.mirabilis. Cefpodoxime, cefinidir, and cefuroxime axetil may be tested individually because some isolated may be susceptible to these agents while testing resistant to cefazolin. Cefepime dosing for Enterobacteriaceae ( E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia & Proteus spp) Susceptible Susceptible –dose-dependent (SDD) Resistant MIC </= 2 4 8 >/= 16 Based on dose of: 1g q12h 1g every 8h or 2g every 8 h Do not give 2g q12 Total dose 2g 3-4g 6g NA Table 2: 2014 MIC Interpretive Standards for Pseudomonas aeruginosa and Acinetobacter spp.