09 Non-Steroidal Anti-Inflammatory Drugs (Nsaids)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Aryl Hydrocarbon Receptor: Structural Analysis and Activation Mechanisms
The Aryl Hydrocarbon Receptor: Structural Analysis and Activation Mechanisms This thesis is submitted in fulfilment of the requirements for the degree of Doctor of Philosophy in the School of Molecular and Biomedical Sciences (Biochemistry), The University of Adelaide, Australia Fiona Whelan, B.Sc. (Hons) 2009 2 Table of Contents THESIS SUMMARY................................................................................. 6 DECLARATION....................................................................................... 7 PUBLICATIONS ARISING FROM THIS THESIS.................................... 8 ACKNOWLEDGEMENTS...................................................................... 10 ABBREVIATIONS ................................................................................. 12 CHAPTER 1: INTRODUCTION ............................................................. 17 1.1 BHLH.PAS PROTEINS ............................................................................................17 1.1.1 General background..................................................................................17 1.1.2 bHLH.PAS Class I Proteins.........................................................................18 1.2 THE ARYL HYDROCARBON RECEPTOR......................................................................19 1.2.1 Domain Structure and Ligand Activation ..............................................19 1.2.2 AhR Expression and Developmental Activity .......................................21 1.2.3 Mouse AhR Knockout Phenotype ...........................................................23 -

Antipyretic Efficacy of an Initial 30-Mg/Kg Loading Dose of Acetaminophen Versus a 15-Mg/Kg Maintenance Dose
Antipyretic Efficacy of an Initial 30-mg/kg Loading Dose of Acetaminophen Versus a 15-mg/kg Maintenance Dose Jean Marc Tre´luyer, MD, PhD*; Sylvie Tonnelier, PharmD*; Philippe d’Athis‡; Beatrice Leclerc§; Isabelle Jolivet-Landreau, MD§; and Ge´rard Pons, MD, PhD* ABSTRACT. Objective. To compare the antipyretic comfort.1 Among the available antipyretic agents, efficacy of an initial 30-mg/kg acetaminophen loading acetaminophen is one of the most widely used be- dose versus a 15-mg/kg maintenance dose. cause it has an antipyretic efficacy equivalent to the Methods. A double-blind, parallel-group, random- other antipyretic drugs and a good safety profile.2–4 ized clinical trial was conducted. A total of 121 febrile Acetaminophen antipyretic efficacy is dose-depen- (rectal temperature between 39°C and 40°C) but other- dent.5,6 The recommended dose is 15 mg/kg every 6 wise healthy outpatients who were 4 months to 9 years of 2 age and weighed 4 to 26 kg were assigned randomly to 1 hours. Following this dosing schedule, it usually -takes several hours after the first acetaminophen ad ؍ and 30 mg/kg (n (62 ؍ of the dose groups: 15 mg/kg (n 59). ministration to obtain the maximal clinical efficacy of Results. In an “intention to treat” analysis, the time to the drug.7 Therefore, Mahar et al8 suggested a phys- obtain a temperature lower than 38.5°C was significantly ical “cold therapy” at the initiation of the chemical shorter in the 30-mg/kg than in the 15-mg/kg group treatment to decrease the time course to antipyretic minutes vs 139 ؎ 113 minutes). -

Open Thesis Master Document V5.0.Pdf
The Pennsylvania State University The Graduate School Department of Veterinary and Biomedical Science IDENTIFICATION OF ENDOGENOUS MODULATORS FOR THE ARYL HYDROCARBON RECEPTOR A Thesis in Genetics by Christopher R. Chiaro © 2007 Christopher R. Chiaro Submitted in Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy December, 2007 The thesis of Christopher R. Chiaro was reviewed and approved* by the following: Gary H. Perdew John T. and Paige S. Smith Professor in Agricultural Sciences Thesis Advisor Chair of Committee C. Channa Reddy Distinguished Professor of Veterinary Science A. Daniel Jones Senior Scientist Department of Chemistry John P. Vanden Heuvel Professor of Veterinary Science Richard Ordway Associate Professor of Biology Chair of Genetics Graduate Program *Signatures are on file in the Graduate School iii ABSTRACT The aryl hydrocarbon receptor (AhR) is a ligand-activated transcription factor capable of being regulated by a structurally diverse array of chemicals ranging from environmental carcinogens to dietary metabolites. A member of the basic helix-loop- helix/ Per-Arnt-Sim (bHLH-PAS) super-family of DNA binding regulatory proteins, the AhR is an important developmental regulator that can be detected in nearly all mammalian tissues. Prior to ligand activation, the AhR resides in the cytosol as part of an inactive oligomeric protein complex comprised of the AhR ligand-binding subunit, a dimer of the 90 kDa heat shock protein, and a single molecule each of the immunophilin like X-associated protein 2 (XAP2) and p23 proteins. Functioning as chemosensor, the AhR responds to both endobiotic and xenobiotic derived chemical ligands by ultimately directing the expression of metabolically important target genes. -

Epigenetic Regulations of Ahr in the Aspect of Immunomodulation
International Journal of Molecular Sciences Review Epigenetic Regulations of AhR in the Aspect of Immunomodulation Anna Wajda 1,* , Joanna Łapczuk-Roma ´nska 2 and Agnieszka Paradowska-Gorycka 1 1 Department of Molecular Biology, National Institute of Geriatrics, Rheumatology and Rehabilitation, 02-637 Warsaw, Poland; [email protected] 2 Department of Experimental and Clinical Pharmacology, Pomeranian Medical University, 70-111 Szczecin, Poland; [email protected] * Correspondence: [email protected] Received: 31 July 2020; Accepted: 28 August 2020; Published: 3 September 2020 Abstract: Environmental factors contribute to autoimmune disease manifestation, and as regarded today, AhR has become an important factor in studies of immunomodulation. Besides immunological aspects, AhR also plays a role in pharmacological, toxicological and many other physiological processes such as adaptive metabolism. In recent years, epigenetic mechanisms have provided new insight into gene regulation and reveal a new contribution to autoimmune disease pathogenesis. DNA methylation, histone modifications, chromatin alterations, microRNA and consequently non-genetic changes in phenotypes connect with environmental factors. Increasing data reveals AhR cross-roads with the most significant in immunology pathways. Although study on epigenetic modulations in autoimmune diseases is still not well understood, therefore future research will help us understand their pathophysiology and help to find new therapeutic strategies. Present literature review -

Clinical Report—Fever and Antipyretic Use in Children Abstract
Guidance for the Clinician in Rendering Pediatric Care Clinical Report—Fever and Antipyretic Use in Children Janice E. Sullivan, MD, Henry C. Farrar, MD, and the abstract SECTION ON CLINICAL PHARMACOLOGY AND THERAPEUTICS, and COMMITTEE ON DRUGS Fever in a child is one of the most common clinical symptoms managed by pediatricians and other health care providers and a frequent cause KEY WORDS fever, antipyretics, children of parental concern. Many parents administer antipyretics even when ABBREVIATIONS there is minimal or no fever, because they are concerned that the child NSAID—nonsteroidal anti-inflammatory drug must maintain a “normal” temperature. Fever, however, is not the The guidance in this report does not indicate an exclusive primary illness but is a physiologic mechanism that has beneficial course of treatment or serve as a standard of medical care. effects in fighting infection. There is no evidence that fever itself wors- Variations, taking into account individual circumstances, may be ens the course of an illness or that it causes long-term neurologic appropriate. complications. Thus, the primary goal of treating the febrile child This document is copyrighted and is property of the American Academy of Pediatrics and its Board of Directors. All authors should be to improve the child’s overall comfort rather than focus on have filed conflict of interest statements with the American the normalization of body temperature. When counseling the parents Academy of Pediatrics. Any conflicts have been resolved through or caregivers of a febrile child, the general well-being of the child, the a process approved by the Board of Directors. -

Role of Arachidonic Acid and Its Metabolites in the Biological and Clinical Manifestations of Idiopathic Nephrotic Syndrome
International Journal of Molecular Sciences Review Role of Arachidonic Acid and Its Metabolites in the Biological and Clinical Manifestations of Idiopathic Nephrotic Syndrome Stefano Turolo 1,* , Alberto Edefonti 1 , Alessandra Mazzocchi 2, Marie Louise Syren 2, William Morello 1, Carlo Agostoni 2,3 and Giovanni Montini 1,2 1 Fondazione IRCCS Ca’ Granda-Ospedale Maggiore Policlinico, Pediatric Nephrology, Dialysis and Transplant Unit, Via della Commenda 9, 20122 Milan, Italy; [email protected] (A.E.); [email protected] (W.M.); [email protected] (G.M.) 2 Department of Clinical Sciences and Community Health, University of Milan, 20122 Milan, Italy; [email protected] (A.M.); [email protected] (M.L.S.); [email protected] (C.A.) 3 Fondazione IRCCS Ca’ Granda Ospedale Maggiore Policlinico, Pediatric Intermediate Care Unit, 20122 Milan, Italy * Correspondence: [email protected] Abstract: Studies concerning the role of arachidonic acid (AA) and its metabolites in kidney disease are scarce, and this applies in particular to idiopathic nephrotic syndrome (INS). INS is one of the most frequent glomerular diseases in childhood; it is characterized by T-lymphocyte dysfunction, alterations of pro- and anti-coagulant factor levels, and increased platelet count and aggregation, leading to thrombophilia. AA and its metabolites are involved in several biological processes. Herein, Citation: Turolo, S.; Edefonti, A.; we describe the main fields where they may play a significant role, particularly as it pertains to their Mazzocchi, A.; Syren, M.L.; effects on the kidney and the mechanisms underlying INS. AA and its metabolites influence cell Morello, W.; Agostoni, C.; Montini, G. -
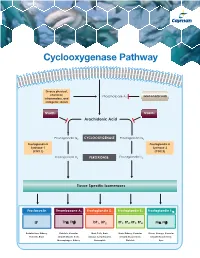
Cyclooxygenase Pathway
Cyclooxygenase Pathway Diverse physical, chemical, Phospholipase A Glucocorticoids inflammatory, and 2 mitogenic stimuli NSAIDs NSAIDs Arachidonic Acid Prostaglandin G2 CYCLOOXYGENASE Prostaglandin G2 Prostaglandin H Prostaglandin H Synthase-1 Synthase-2 (COX 1) (COX 2) Prostaglandin H2 PEROXIDASE Prostaglandin H2 Tissue Specific Isomerases Prostacyclin Thromboxane A2 Prostaglandin D2 Prostaglandin E2 Prostaglandin F2α IP TPα, TPβ DP1, DP2 EP1, EP2, EP3, EP4 FPα, FPβ Endothelium, Kidney, Platelets, Vascular Mast Cells, Brain, Brain, Kidney, Vascular Uterus, Airways, Vascular Platelets, Brain Smooth Muscle Cells, Airways, Lymphocytes, Smooth Muscle Cells, Smooth Muscle Cells, Macrophages, Kidney Eosinophils Platelets Eyes Prostacyclin Item No. Product Features Prostacyclin (Prostaglandin I2; PGI2) is formed from arachidonic acid primarily in the vascular endothelium and renal cortex by sequential 515211 6-keto • Sample Types: Culture Medium | Plasma Prostaglandin • Measure 6-keto PGF levels down to 6 pg/ml activities of COX and prostacyclin synthase. PGI2 is non-enzymatically 1α F ELISA Kit • Incubation : 18 hours | Development: 90-120 minutes | hydrated to 6-keto PGF1α (t½ = 2-3 minutes), and then quickly converted 1α Read: Colorimetric at 405-420 nm to the major metabolite, 2,3-dinor-6-keto PGF1α (t½= 30 minutes). Prostacyclin was once thought to be a circulating hormone that regulated • Assay 24 samples in triplicate or 36 samples in duplicate platelet-vasculature interactions, but the rate of secretion into circulation • NOTE: A portion of urinary 6-keto PGF1α is of renal origin coupled with the short half-life indicate that prostacyclin functions • NOTE : It has been found that normal plasma levels of 6-keto PGF may be low locally. -

The Use of Stems in the Selection of International Nonproprietary Names (INN) for Pharmaceutical Substances
WHO/PSM/QSM/2006.3 The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances 2006 Programme on International Nonproprietary Names (INN) Quality Assurance and Safety: Medicines Medicines Policy and Standards The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances FORMER DOCUMENT NUMBER: WHO/PHARM S/NOM 15 © World Health Organization 2006 All rights reserved. Publications of the World Health Organization can be obtained from WHO Press, World Health Organization, 20 Avenue Appia, 1211 Geneva 27, Switzerland (tel.: +41 22 791 3264; fax: +41 22 791 4857; e-mail: [email protected]). Requests for permission to reproduce or translate WHO publications – whether for sale or for noncommercial distribution – should be addressed to WHO Press, at the above address (fax: +41 22 791 4806; e-mail: [email protected]). The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the World Health Organization concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. Dotted lines on maps represent approximate border lines for which there may not yet be full agreement. The mention of specific companies or of certain manufacturers’ products does not imply that they are endorsed or recommended by the World Health Organization in preference to others of a similar nature that are not mentioned. Errors and omissions excepted, the names of proprietary products are distinguished by initial capital letters. -

Pharmacological Evaluation of Novel Dimers of an Arylpropionic Acid Class of Non-Selective Cyclooxygenase Inhibitors
Halimi et al Tropical Journal of Pharmaceutical Research February 2017; 16 (2): 327-336 ISSN: 1596-5996 (print); 1596-9827 (electronic) © Pharmacotherapy Group, Faculty of Pharmacy, University of Benin, Benin City, 300001 Nigeria. All rights reserved. Available online at http://www.tjpr.org http://dx.doi.org/10.4314/tjpr.v16i2.10 Original Research Article Pharmacological evaluation of novel dimers of an arylpropionic acid class of non-selective cyclooxygenase inhibitors Syed Muhammad Ashhad Halimi1*, Muhammad Saeed1, Safiullah1 and Khalid Muhammed Khan2 1Department of Pharmacy, University of Peshawar, Peshawar 25120, 2H.E.J. International Centre for Chemical and Biological Sciences (ICCBS), University of Karachi, Karachi 75270, Pakistan *For correspondence: Email: [email protected]; Tel: +92 91 9216750; Fax: +92 91 9218318 Received: 4 August 2016 Revised accepted: 5 January 2017 Abstract Purpose: To explore and identify cyclooxygenase (COX) inhibitors with optimal potency and efficacy using an arylpropionic acid class of drugs as lead molecules. Methods: The selected lead molecules were dimerised through chemical processes (reflux condensation) and characterised in terms of structural properties using infrared, proton nuclear magnetic resonance, electron impact mass spectrometry, and elemental analysis techniques. The molecules were evaluated pharmacologically for acute toxicity and anti-inflammatory (carrageenan- induced paw oedema test), analgesic (acetic acid-induced writhing test in mice), and antipyretic (Brewer’s yeast-induced pyrexia test in mice) activities against control (normal saline) and relevant reference standard drugs. Docking analyses were also performed to assess possible protein–ligand interactions. Results: The test compounds were non-toxic at doses of 50, 100 and 150 mg/kg body weight, ip. -

The Protective Effects of Conjugated Linoleic Acid Against Carcinogenesis
The Protective Effects of Conjugated Linoleic Acid Against Carcinogenesis Item Type text; Electronic Dissertation Authors Kemp, Michael Quentin Publisher The University of Arizona. Rights Copyright © is held by the author. Digital access to this material is made possible by the University Libraries, University of Arizona. Further transmission, reproduction or presentation (such as public display or performance) of protected items is prohibited except with permission of the author. Download date 24/09/2021 15:13:15 Link to Item http://hdl.handle.net/10150/193637 THE PROTECTIVE EFFECTS OF CONJUGATED LINOLEIC ACID AGAINST CARCINOGENESIS by Michael Quentin Kemp A Dissertation Submitted to the Faculty of the GRADUATE PROGRAM IN NUTRITIONAL SCIENCES In Partial Fulfillment of the Requirements for the Degree of DOC TORAL OF PHILOSOPHY In the Graduate College THE UNIVERSITY OF ARIZONA 2005 2 THE UNIVERSITY OF ARIZONA GRADUATE COLLEGE As members of the Dissertation Committee, we certify that we have read the dissertation prepared by Michael Q. Kemp entitled The Protective Effec ts of Conjugated Linoleic Acid Against Carcinogenesis and recommend that it be accepted as fulfilling the dissertation requirement for the Deg ree of Doctor of Philosophy _______ ___________________________ __________________ ________ _______ ___ _ Date : 11/18/05 . Donato Romagnolo _______ ___________________________ __________________ ________ ___________ Date: 11/18/05 . Wanda Howell _______ ___________________________ __________________ ________ ___________ Date: 11/18/05 . Linda Houtkooper _______ ___________________________ __________________ ________ ___________ Date: 11/18/05 . Scott Going _______ ___________________________ __________________ ________ ___________ Date: 11/18/05 . Joy Winzerling Final approval and acceptance of this dissertation is contingent upon the candidate’s submission of the final copies of the dissertation to the Graduate College. -

Different Techniques for Analysis of Aspirin, Caffeine, Diclofenac Sodium and Paracetamol: Review Article
ISSN 2692-4374 Pharmaceutical Sciences | Review Article Different techniques for Analysis of Aspirin, Caffeine, Diclofenac Sodium and Paracetamol: Review Article Mahmoud M. Sebaiy1*, Sobhy M. El-Adl1, and Amr A. Mattar1&2 1 Medicinal Chemistry Department, Faculty of Pharmacy, Zagazig University, Zagazig, 44519, Egypt. 2 Pharmaceutical Medicinal Chemistry Department, Faculty of Pharmacy, Egyptian Russian University, Badr City, Cairo 11829, Egypt. *Аuthоrcоrrеspоndеncе: Е-mаil: mmsеbаiу@zu.еdu.еg; sеbаiуm@gmаil.cоm.Tеl: 01062780060. Fаx: 0552303266 Submitted: 27 April 2020 Approved: 11 May 2020 Published: 14 May 2020 How to cite this article: Sebaiy MM, El-Adl SM, Mattar AA. Different techniques for Analysis of Aspirin, Caffeine, Diclofenac Sodium and Paracetamol: Review Article. G Med Sci. 2020; 1(1): 013-031. https://www.doi.org/10.46766/thegms.pharma.20042701 Copyright: © 2020 Mahmoud MS. This is an open access article distributed under the Creative Commons Attribution License, which permits unre- stricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTracT Early treatment of pain is of a great importance as unrelieved pain can have profound psychological effects on the patient, and acute pain that is poorly managed initially can degenerate into chronic pain, which may prove to be much more difficult to treat. It is important to assess and treat the article,mental weand will emotional shed the aspects light on of different the pain waysas well of assome its physicalanalgesic aspects. drugs monitoring Although drug and therapyanalysis isusing a mainstay different of techniques pain treatment, in addition physical to methodsthe most such as physiotherapy (including massage and the application of heat and cold), surgery, and drug monitoring are also very valuable. -

Formulations Comprising Nanoparticulate Meloxicam
(19) TZZ¥ZZ¥__T (11) EP 3 090 731 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: (51) Int Cl.: 09.11.2016 Bulletin 2016/45 A61K 9/14 (2006.01) A61K 31/5415 (2006.01) (21) Application number: 16161176.9 (22) Date of filing: 27.02.2004 (84) Designated Contracting States: • Ryde, Tuula AT BE BG CH CY CZ DE DK EE ES FI FR GB GR Malvern, PA Pennsylvania 19355 (US) HU IE IT LI LU MC NL PT RO SE SI SK TR • Pruitt, John, D. Suwanee, GA Georgia 30024 (US) (30) Priority: 03.03.2003 US 450705 P • Kline, Laura Harleysville, PA Pennsylvania 19438 (US) (62) Document number(s) of the earlier application(s) in accordance with Art. 76 EPC: (74) Representative: Wichmann, Hendrik 08006465.2 / 1 938 803 Wuesthoff & Wuesthoff 04785761.0 / 1 617 816 Patentanwälte PartG mbB Schweigerstraße 2 (71) Applicant: DV Technology LLC 81541 München (DE) Wilmington, Delaware 19801 (US) Remarks: (72) Inventors: This application was filed on 18-03-2016 as a • Cooper, Eugene, R. divisional application to the application mentioned Berwyn, PA Pennsylvania 19312 (US) under INID code 62. (54) FORMULATIONS COMPRISING NANOPARTICULATE MELOXICAM (57) The present invention is directed to composi- than about 2 microns; wherein said effective average par- tions comprising meloxicam. The stable meloxicam com- ticle size is different than the particle size of the nano- positions comprise (a) nanoparticulate particles of mel- particulate particles of meloxicam (a); and (c) at least one oxicam having an effective average particle size of less surface stabilizer. The invention relates also to uses of than about 2000 nm, (b) particles of meloxicam having the composition.