Histologic Variations in the Structure of Human Eccrine Sweat Glands* John B
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Distribution of Sweat Glands Over the Human Body Has So Far Been In
NOTES ON THE VERTICAL DISTRIBUTION OF THE HUMAN SWEAT GLANDS SHUNZO TAKAGI AND KO TOBARU* Institute of Physiology, School of Medicine, University of Nagoya•õ The distribution of sweat glands over the human body has so far been in- vestigated in the dimension of area, and we have no general idea how deep they are distributed in the skin. In 1943, Kuno and his collaborators (3) expressed the opinion that chloride would be accumulated in the skin during the activity of sweat glands. The truth of this assumption has been confirmed with more certainty by Yoshimura and Chihaya (unpublished), who measured the chloride content in the skin tissue by means of Ag-AgCl electrodes. The chloride may presumably be accumulated in the immediate neighbourhood of the glomeruli of sweat glands, or more diffusely in the layers of skin tissues where the glomeruli are situated. For consideration of the amount of the accumulated chloride, the total volume of these skin layers, which can be estimated by the vertical distribution of the sweat-gland glomeruli, seems to be useful. The fol- lowing investigation was therefore performed. MATERIALS AND METHOD Skin samples of 33 regions of the body, as specified in table 1, were taken from the corpse of a Japanese male of 30 years old, who died an accidental death. The samples were fixed in 10 per cent formalin, embedded in celloidin and cut into sections 15 micra thick. The sections were stained with Delafield's hematoxylin and eosin. Observations of sweat glands were made with 2-3 pieces of the skin about 100 sq. -

Periodic Acid-Schiff Positive Material Accumulating Within the Lumen of Eccrine Sweat Glands*
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector PERIODIC ACID-SCHIFF POSITIVE MATERIAL ACCUMULATING WITHIN THE LUMEN OF ECCRINE SWEAT GLANDS* GEORGE W. HAMBRICK, JR., M.D. The purpose of this paper is to record findings with regard to diastase-resistant, periodic acid-Schiff positive material in the lumen of eccrine ducts and glands of two individuals. This material has two possible sources in the normal eccrine sweat gland, namely, the cuticle lining of the eccrine duct and the cells of the secretory tubule. Holyoke and Lobits (3) studying 35 normal skin biopsies re- B pp HI D FIG. 1. A. Vertical section through skin of the control area showing dilatation of an eccrine sweat duct with eosinophilic material in the lumen. H. and E., X178. B. Same find- ings as in A above from an area treated daily with 3 per cent hexachloronaphthalene in acetone for one week, Xl7S. C. Section through dermal parts of eccrine duct containing a cast. H. and E., X355. D. Section through dermal eccrine duct containing periodic acid- Schiff positive material in the lumen, X660. *Fromthe Department of Dermatology (Donald M. Pillsbury, M.D., Director), School of Medicine, University of Pennsylvania, Philadelphia 4, Pennsylvania. This study was supported by U. S. Army grant DA-49-007-MD-154. Received for publication March 29, 1957. 213 214 THF JOURNAL OF INVESTIGATIVE DERMATOLOGY ported the presence of material in the lumen of the eccrine duct and gland; they classified the material as amorphous, cast, cellular or bacterial. -

Sweat Gland Myoepithelial Cell Differentiation
Journal of Cell Science 112, 1925-1936 (1999) 1925 Printed in Great Britain © The Company of Biologists Limited 1999 JCS4638 Human sweat gland myoepithelial cells express a unique set of cytokeratins and reveal the potential for alternative epithelial and mesenchymal differentiation states in culture Margarete Schön1,*, Jennifer Benwood1, Therese O’Connell-Willstaedt2 and James G. Rheinwald1,2,‡ 1Division of Dermatology/Department of Medicine, Brigham and Women’s Hospital, and 2Division of Cell Growth and Regulation, Dana-Farber Cancer Institute, Harvard Medical School, Boston, MA 02115, USA *Present address: Department of Dermatology, Heinrich-Heine University, Moorenstrasse 5, 40225 Düsseldorf, Germany ‡Author for correspondence (e-mail: [email protected]) Accepted 9 April; published on WWW 26 May 1999 SUMMARY We have characterized precisely the cytokeratin expression myoepithelial cells, a constituent of secretory glands. pattern of sweat gland myoepithelial cells and have Immunostaining of skin sections revealed that only sweat identified conditions for propagating this cell type and gland myoepithelial cells expressed the same pattern of modulating its differentiation in culture. Rare, unstratified keratins and α-sma and lack of E-cadherin as the cell type epithelioid colonies were identified in cultures initiated we had cultured. Interestingly, our immunocytochemical from several specimens of full-thickness human skin. These analysis of ndk, a skin-derived cell line of uncertain cells divided rapidly in medium containing serum, identity, suggests that this line is of myoepithelial origin. epidermal growth factor (EGF), and hydrocortisone, and Earlier immunohistochemical studies by others had found maintained a closely packed, epithelioid morphology when myoepithelial cells to be K7-negative. -

Nomina Histologica Veterinaria, First Edition
NOMINA HISTOLOGICA VETERINARIA Submitted by the International Committee on Veterinary Histological Nomenclature (ICVHN) to the World Association of Veterinary Anatomists Published on the website of the World Association of Veterinary Anatomists www.wava-amav.org 2017 CONTENTS Introduction i Principles of term construction in N.H.V. iii Cytologia – Cytology 1 Textus epithelialis – Epithelial tissue 10 Textus connectivus – Connective tissue 13 Sanguis et Lympha – Blood and Lymph 17 Textus muscularis – Muscle tissue 19 Textus nervosus – Nerve tissue 20 Splanchnologia – Viscera 23 Systema digestorium – Digestive system 24 Systema respiratorium – Respiratory system 32 Systema urinarium – Urinary system 35 Organa genitalia masculina – Male genital system 38 Organa genitalia feminina – Female genital system 42 Systema endocrinum – Endocrine system 45 Systema cardiovasculare et lymphaticum [Angiologia] – Cardiovascular and lymphatic system 47 Systema nervosum – Nervous system 52 Receptores sensorii et Organa sensuum – Sensory receptors and Sense organs 58 Integumentum – Integument 64 INTRODUCTION The preparations leading to the publication of the present first edition of the Nomina Histologica Veterinaria has a long history spanning more than 50 years. Under the auspices of the World Association of Veterinary Anatomists (W.A.V.A.), the International Committee on Veterinary Anatomical Nomenclature (I.C.V.A.N.) appointed in Giessen, 1965, a Subcommittee on Histology and Embryology which started a working relation with the Subcommittee on Histology of the former International Anatomical Nomenclature Committee. In Mexico City, 1971, this Subcommittee presented a document entitled Nomina Histologica Veterinaria: A Working Draft as a basis for the continued work of the newly-appointed Subcommittee on Histological Nomenclature. This resulted in the editing of the Nomina Histologica Veterinaria: A Working Draft II (Toulouse, 1974), followed by preparations for publication of a Nomina Histologica Veterinaria. -

Sweat Glands • Oil Glands • Mammary Glands
Chapter 4 The Integumentary System Lecture Presentation by Steven Bassett Southeast Community College © 2015 Pearson Education, Inc. Introduction • The integumentary system is composed of: • Skin • Hair • Nails • Sweat glands • Oil glands • Mammary glands © 2015 Pearson Education, Inc. Introduction • The skin is the most visible organ of the body • Clinicians can tell a lot about the overall health of the body by examining the skin • Skin helps protect from the environment • Skin helps to regulate body temperature © 2015 Pearson Education, Inc. Integumentary Structure and Function • Cutaneous Membrane • Epidermis • Dermis • Accessory Structures • Hair follicles • Exocrine glands • Nails © 2015 Pearson Education, Inc. Figure 4.1 Functional Organization of the Integumentary System Integumentary System FUNCTIONS • Physical protection from • Synthesis and storage • Coordination of immune • Sensory information • Excretion environmental hazards of lipid reserves response to pathogens • Synthesis of vitamin D3 • Thermoregulation and cancers in skin Cutaneous Membrane Accessory Structures Epidermis Dermis Hair Follicles Exocrine Glands Nails • Protects dermis from Papillary Layer Reticular Layer • Produce hairs that • Assist in • Protect and trauma, chemicals protect skull thermoregulation support tips • Nourishes and • Restricts spread of • Controls skin permeability, • Produce hairs that • Excrete wastes of fingers and supports pathogens prevents water loss provide delicate • Lubricate toes epidermis penetrating epidermis • Prevents entry of -

HYPERHIDROSIS 101: Understanding Excessive Sweat
HYPERHIDROSIS 101: Understanding Excessive Sweat Hyperhidrosis is a medical condition in which excessive sweating occurs beyond what is needed to maintain normal body temperature.1,2 Excessive sweating can occur in the hands, feet, underarms or face, and it often interferes with everyday activities.1 Understanding Sweat Sweat glands are small tubular structures in the skin that secrete sweat onto the skin via a duct.3 Eccrine sweat glands are distributed throughout almost the entire human body and they secrete directly onto the surface of the skin.3 Apocrine sweat glands are ten times larger than eccrine sweat glands.3 They are localized in the axilla (underarms) and perianal areas.3 Rather than directly opening onto the skin surface, these glands secrete sweat into the pilary canal of the hair follicle.3 What is Hyperhidrosis? We all sweat. It’s the body’s way of cooling itself and preventing 1 ourselves from overheating. People living with hyperhidrosis, What Causes Us to Sweat? however, sweat when the body doesn’t necessarily need cooling.1 Their sweat is excessive, often visible to others and usually occurs The main reason we sweat is to control without physical exertion or extreme heat. our body temperature.2 There are two different types of hyperhidrosis - primary (also known as focal Here’s how it works: Sensors in our skin can detect changes in temperature hyperhidrosis) and secondary hyperhidrosis.6 Primary hyperhidrosis often begins and relay signals to our brain when we in childhood or adolescence and affects localized areas of the body, including the exercise or when it is hot outside.4 In hands, feet, underarms or face.6 The condition may be inherited and many members turn, our brain signals the sweat glands 7 of the same family may suffer from hyperhidrosis. -
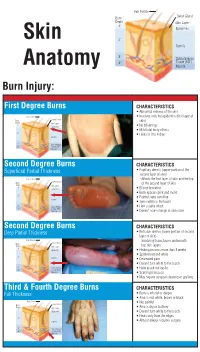
Identifying the Degree of a Burn Poster
Hair Folicle Sweat Gland Burn Depth Skin Layer 1° Epidermis Skin 2° Dermis 3° Subcutaneous 4° Tissue (FAT) Anatomy Muscle Burn Injury: First Degree Burns CHARACTERISTICS • Abnormal redness of the skin Hair Folicle • Involves only the epidermis (first layer of Sweat Gland Burn skin) Depth Skin Layer 1° Epidermis • No blistering • Mild total body effects 2° • Heals in 3 to 4 days Dermis 3° Subcutaneous 4° Tissue (FAT) Muscle Second Degree Burns CHARACTERISTICS Superficial Partial Thickness • Papillary dermis (upper portion of the second layer of skin) Hair Folicle – Affects the first layer of skin and the top Sweat Gland Burn of the second layer of skin Depth Skin Layer 1° Epidermis • Blister formation • Burn appears pink and moist 2° • Painful, very sensitive Dermis • Turns white to the touch 3° Subcutaneous • Hair usually intact 4° Tissue (FAT) Muscle • Doesn’t scar–change in skin color Second Degree Burns CHARACTERISTICS Deep Partial Thickness • Reticular dermis (lower portion of second layer of skin) Hair Folicle – Involving tissue layers underneath Sweat Gland Burn Depth Skin Layer top skin layers 1° Epidermis • Healing process more than 3 weeks • Spotted red and white 2° Dermis • Decreased pain • Doesn’t turn white to the touch 3° Subcutaneous • Hairs pluck out easily 4° Tissue (FAT) Muscle • Scarring increases • May require surgical cleaning or grafting Third & Fourth Degree Burns CHARACTERISTICS Full Thickness • Burn is into fat or deeper • Area is red, white, brown or black Hair Folicle • Not painful Sweat Gland Burn Depth Skin Layer • Area is dry or leathery 1° Epidermis • Doesn’t turn white to the touch • Heals only from the edges 2° Dermis • Almost always requires surgery 3° Subcutaneous 4° Tissue (FAT) Muscle. -

26 April 2010 TE Prepublication Page 1 Nomina Generalia General Terms
26 April 2010 TE PrePublication Page 1 Nomina generalia General terms E1.0.0.0.0.0.1 Modus reproductionis Reproductive mode E1.0.0.0.0.0.2 Reproductio sexualis Sexual reproduction E1.0.0.0.0.0.3 Viviparitas Viviparity E1.0.0.0.0.0.4 Heterogamia Heterogamy E1.0.0.0.0.0.5 Endogamia Endogamy E1.0.0.0.0.0.6 Sequentia reproductionis Reproductive sequence E1.0.0.0.0.0.7 Ovulatio Ovulation E1.0.0.0.0.0.8 Erectio Erection E1.0.0.0.0.0.9 Coitus Coitus; Sexual intercourse E1.0.0.0.0.0.10 Ejaculatio1 Ejaculation E1.0.0.0.0.0.11 Emissio Emission E1.0.0.0.0.0.12 Ejaculatio vera Ejaculation proper E1.0.0.0.0.0.13 Semen Semen; Ejaculate E1.0.0.0.0.0.14 Inseminatio Insemination E1.0.0.0.0.0.15 Fertilisatio Fertilization E1.0.0.0.0.0.16 Fecundatio Fecundation; Impregnation E1.0.0.0.0.0.17 Superfecundatio Superfecundation E1.0.0.0.0.0.18 Superimpregnatio Superimpregnation E1.0.0.0.0.0.19 Superfetatio Superfetation E1.0.0.0.0.0.20 Ontogenesis Ontogeny E1.0.0.0.0.0.21 Ontogenesis praenatalis Prenatal ontogeny E1.0.0.0.0.0.22 Tempus praenatale; Tempus gestationis Prenatal period; Gestation period E1.0.0.0.0.0.23 Vita praenatalis Prenatal life E1.0.0.0.0.0.24 Vita intrauterina Intra-uterine life E1.0.0.0.0.0.25 Embryogenesis2 Embryogenesis; Embryogeny E1.0.0.0.0.0.26 Fetogenesis3 Fetogenesis E1.0.0.0.0.0.27 Tempus natale Birth period E1.0.0.0.0.0.28 Ontogenesis postnatalis Postnatal ontogeny E1.0.0.0.0.0.29 Vita postnatalis Postnatal life E1.0.1.0.0.0.1 Mensurae embryonicae et fetales4 Embryonic and fetal measurements E1.0.1.0.0.0.2 Aetas a fecundatione5 Fertilization -

A Concise Review on Tissue Engineered Artificial Skin
cells Review A Concise Review on Tissue Engineered Artificial Skin Grafts for Chronic Wound Treatment: Can We Reconstruct Functional Skin Tissue In Vitro? Agata Przekora Department of Biochemistry and Biotechnology, Medical University of Lublin, Chodzki 1 Street, 20-093 Lublin, Poland; [email protected]; Tel.: +48-81-448-7026 Received: 26 May 2020; Accepted: 21 June 2020; Published: 6 July 2020 Abstract: Chronic wounds occur as a consequence of a prolonged inflammatory phase during the healing process, which precludes skin regeneration. Typical treatment for chronic wounds includes application of autografts, allografts collected from cadaver, and topical delivery of antioxidant, anti-inflammatory, and antibacterial agents. Nevertheless, the mentioned therapies are not sufficient for extensive or deep wounds. Moreover, application of allogeneic skin grafts carries high risk of rejection and treatment failure. Advanced therapies for chronic wounds involve application of bioengineered artificial skin substitutes to overcome graft rejection as well as topical delivery of mesenchymal stem cells to reduce inflammation and accelerate the healing process. This review focuses on the concept of skin tissue engineering, which is a modern approach to chronic wound treatment. The aim of the article is to summarize common therapies for chronic wounds and recent achievements in the development of bioengineered artificial skin constructs, including analysis of biomaterials and cells widely used for skin graft production. This review also presents attempts to reconstruct nerves, pigmentation, and skin appendages (hair follicles, sweat glands) using artificial skin grafts as well as recent trends in the engineering of biomaterials, aiming to produce nanocomposite skin substitutes (nanofilled polymer composites) with controlled antibacterial activity. -

Physiology of Sweat Gland Function: the Roles of Sweating and Sweat Composition in Human Health Lindsay B
COMPREHENSIVE REVIEW Physiology of sweat gland function: The roles of sweating and sweat composition in human health Lindsay B. Baker Gatorade Sports Science Institute, PepsiCo R&D Physiology and Life Sciences, Barrington, IL, USA ABSTRACT ARTICLE HISTORY The purpose of this comprehensive review is to: 1) review the physiology of sweat gland function Received 30 April 2019 and mechanisms determining the amount and composition of sweat excreted onto the skin Revised 6 June 2019 surface; 2) provide an overview of the well-established thermoregulatory functions and adaptive Accepted 8 June 2019 responses of the sweat gland; and 3) discuss the state of evidence for potential non-thermo- KEYWORDS regulatory roles of sweat in the maintenance and/or perturbation of human health. The role of Chloride; potassium; sauna; sweating to eliminate waste products and toxicants seems to be minor compared with other sodium; sweat biomarkers; avenues of excretion via the kidneys and gastrointestinal tract; as eccrine glands do not adapt to thermoregulation increase excretion rates either via concentrating sweat or increasing overall sweating rate. Studies suggesting a larger role of sweat glands in clearing waste products or toxicants from the body may be an artifact of methodological issues rather than evidence for selective transport. Furthermore, unlike the renal system, it seems that sweat glands do not conserve water loss or concentrate sweat fluid through vasopressin-mediated water reabsorption. Individuals with high NaCl concentrations in sweat (e.g. cystic fibrosis) have an increased risk of NaCl imbalances during prolonged periods of heavy sweating; however, sweat-induced deficiencies appear to be of minimal risk for trace minerals and vitamins. -

Human Sweat Gland Myoepithelial Cells Express a Unique Set Of
Journal of Cell Science 112, 1925-1936 (1999) 1925 Printed in Great Britain © The Company of Biologists Limited 1999 JCS4638 Human sweat gland myoepithelial cells express a unique set of cytokeratins and reveal the potential for alternative epithelial and mesenchymal differentiation states in culture Margarete Schön1,*, Jennifer Benwood1, Therese O’Connell-Willstaedt2 and James G. Rheinwald1,2,‡ 1Division of Dermatology/Department of Medicine, Brigham and Women’s Hospital, and 2Division of Cell Growth and Regulation, Dana-Farber Cancer Institute, Harvard Medical School, Boston, MA 02115, USA *Present address: Department of Dermatology, Heinrich-Heine University, Moorenstrasse 5, 40225 Düsseldorf, Germany ‡Author for correspondence (e-mail: [email protected]) Accepted 9 April; published on WWW 26 May 1999 SUMMARY We have characterized precisely the cytokeratin expression myoepithelial cells, a constituent of secretory glands. pattern of sweat gland myoepithelial cells and have Immunostaining of skin sections revealed that only sweat identified conditions for propagating this cell type and gland myoepithelial cells expressed the same pattern of modulating its differentiation in culture. Rare, unstratified keratins and α-sma and lack of E-cadherin as the cell type epithelioid colonies were identified in cultures initiated we had cultured. Interestingly, our immunocytochemical from several specimens of full-thickness human skin. These analysis of ndk, a skin-derived cell line of uncertain cells divided rapidly in medium containing serum, identity, suggests that this line is of myoepithelial origin. epidermal growth factor (EGF), and hydrocortisone, and Earlier immunohistochemical studies by others had found maintained a closely packed, epithelioid morphology when myoepithelial cells to be K7-negative. -

Structure and Function of the Skin
2 Structure and Function of the Skin Skin disease illustrates structure and function. Loss of or Chapter Contents defects in skin structure impair skin function. Skin dis ease is discussed in more detail in the other chapters. ● Epidermis ● Structure ● Other Cellular Components EPIDERMIS ● Dermal–Epidermal Junction – The Basement Membrane Zone ● Dermis ● Skin Appendages Key Points ● Subcutaneous Fat 1. Keratinocytes are the principal cell of the epidermis 2. Layers in ascending order: basal cell, stratum spinosum, stratum granulosum, stratum corneum 3. Basal cells are undifferentiated, proliferating cells Key Points 4. Stratum spinosum contains keratinocytes connected by desmosomes 1. The major function of the skin is as a barrier to maintain 5. Keratohyalin granules are seen in the stratum granulosum internal homeostasis 6. Stratum corneum is the major physical barrier 2. The epidermis is the major barrier of the skin 7. The number and size of melanosomes, not melanocytes, determine skin color 8. Langerhans cells are derived from bone marrow and are the skin’s first line of immunologic defense ABSTRACT 9. The basement membrane zone is the substrate for attach- ment of the epidermis to the dermis The skin is a large organ, weighing an average of 4 kg and 10. The four major ultrastructural regions of the basement covering an area of 2 m2. Its major function is to act as membrane zone include the hemidesmosomal plaque of a barrier against an inhospitable environment – to pro the basal keratinocyte, lamina lucida, lamina densa, and tect the body from the influences of the outside world. anchoring fibrils located in the sublamina densa region of The importance of the skin is well illustrated by the high the papillary dermis mortality rate associated with extensive loss of skin from burns.