Vitalant | Red Cell Contamination in Platelets and Apheresis Granulocytes
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
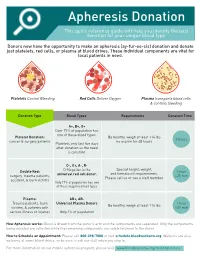
Apheresis Donation This Quick Reference Guide Will Help You Identify the Best Donation for Your Unique Blood Type
Apheresis Donation This quick reference guide will help you identify the best donation for your unique blood type. Donors now have the opportunity to make an apheresis (ay-fur-ee-sis) donation and donate just platelets, red cells, or plasma at blood drives. These individual components are vital for local patients in need. Platelets Control Bleeding Red Cells Deliver Oxygen Plasma transports blood cells & controls bleeding Donation Type Blood Types Requirements Donation Time A+, B+, O+ Over 75% of population has one of these blood types. Platelet Donation: Be healthy, weigh at least 114 lbs 2 hours cancer & surgery patients no aspirin for 48 hours Platelets only last five days after donation so the need is constant. O-, O+, A-, B- Special height, weight, Double Red: O-Negative is the 1 hour and hematocrit requirements. surgery, trauma patients, universal red cell donor. +25 min Please call us or see a staff member accident, & burn victims Only 17% of population has one of these negative blood types Plasma: AB+, AB- Trauma patients, burn Universal Plasma Donors 1 hour Be healthy, weigh at least 114 lbs victims, & patients with +30 min serious illness or injuries Only 4% of population How Apheresis works: Blood is drawn from the donor’s arm and the components are separated. Only the components being donated are collected while the remaining components are safely returned to the donor How to Schedule an Appointment: Please call 800-398-7888 or visit schedule.bloodworksnw.org. Walk-ins are also welcome at some blood drives, so be sure to ask our staff when you stop in. -

Blood Product Modifications: Leukofiltration, Irradiation and Washing
Blood Product Modifications: Leukofiltration, Irradiation and Washing 1. Leukocyte Reduction Definitions and Standards: o Process also known as leukoreduction, or leukofiltration o Applicable AABB Standards, 25th ed. Leukocyte-reduced RBCs At least 85% of original RBCs < 5 x 106 WBCs in 95% of units tested . Leukocyte-reduced Platelet Concentrates: At least 5.5 x 1010 platelets in 75% of units tested < 8.3 x 105 WBCs in 95% of units tested pH≥6.2 in at least 90% of units tested . Leukocyte-reduced Apheresis Platelets: At least 3.0 x 1011 platelets in 90% of units tested < 5.0 x 106 WBCs 95% of units tested pH≥6.2 in at least 90% of units tested Methods o Filter: “Fourth-generation” filters remove 99.99% WBCs o Apheresis methods: most apheresis machines have built-in leukoreduction mechanisms o Less efficient methods of reducing WBC content . Washing, deglycerolizing after thawing a frozen unit, centrifugation . These methods do not meet requirement of < 5.0 x 106 WBCs per unit of RBCs/apheresis platelets. Types of leukofiltration/leukoreduction o “Pre-storage” . Done within 24 hours of collection . May use inline filters at time of collection (apheresis) or post collection o “Pre-transfusion” leukoreduction/bedside leukoreduction . Done prior to transfusion . “Bedside” leukoreduction uses gravity-based filters at time of transfusion. Least desirable given variability in practice and absence of proficiency . Alternatively performed by transfusion service prior to issuing Benefits of leukoreduction o Prevention of alloimmunization to donor HLA antigens . Anti-HLA can mediate graft rejection and immune mediated destruction of platelets o Leukoreduced products are indicated for transplant recipients or patients who are likely platelet transfusion dependent o Prevention of febrile non-hemolytic transfusion reactions (FNHTR) . -

Cord Blood Stem Cell Transplantation
LEUKEMIA LYMPHOMA MYELOMA FACTS Cord Blood Stem Cell Transplantation No. 2 in a series providing the latest information on blood cancers Highlights • Umbilical cord blood, like bone marrow and peripheral blood, is a rich source of stem cells for transplantation. There may be advantages for certain patients to have cord blood stem cell transplants instead of transplants with marrow or peripheral blood stem cells (PBSCs). • Stem cell transplants (peripheral blood, marrow or cord blood) may use the patient’s own stem cells (called “autologous transplants”) or use donor stem cells. Donor cells may come from either a related or unrelated matched donor (called an “allogeneic transplant”). Most transplant physicians would not want to use a baby’s own cord blood (“autologous transplant”) to treat his or her leukemia. This is because donor stem cells might better fight the leukemia than the child’s own stem cells. • Cord blood for transplantation is collected from the umbilical cord and placenta after a baby is delivered. Donated cord blood that meets requirements is frozen and stored at a cord blood bank for future use. • The American Academy of Pediatrics’s (AAP) policy statement (Pediatrics; 2007;119:165-170.) addresses public and private banking options available to parents. Among several recommendations, the report encourages parents to donate to public cord blood banks and discourages parents from using private cord blood banks for personal or family cord blood storage unless they have an older child with a condition that could benefit from transplantation. • The Stem Cell Therapeutic and Research Act of 2005 put several programs in place, including creation of the National Cord Blood Inventory (NCBI) for patients in need of transplantation. -

Therapeutic Apheresis, J Clin Apheresis 2007, 22, 104-105
Apheresis: Basic Principles, Practical Considerations and Clinical Applications Joseph Schwartz, MD Anand Padmanabhan, MD PhD Director, Transfusion Medicine Assoc Med Director/Asst Prof Columbia Univ. Medical Center BloodCenter of Wisconsin New York Presbyterian Hospital Medical College of Wisconsin Review Session, ASFA Annual meeting, Scottsdale, Arizona, June 2011 Objectives (Part 1) • Mechanism of Action • Definitions • Technology (ies) • Use • Practical Considerations • Math • Clinical applications – HPC Collection Objectives (Part 2) • Clinical applications: System/ Disease Specific Indications • ASFA Fact Sheet Apheresis •Derives from Greek, “to carry away” •A technique in which whole blood is taken and separated extracorporealy, separating the portion desired from the remaining blood. •This allows the desired portion (e.g., plasma) to be removed and the reminder returned. Apheresis- Mechanism of Action •Large-bore intravenous catheter connected to a spinning centrifuge bowl •Whole blood is drawn from donor/patient into the centrifuge bowl •The more dense elements, namely the RBC, settle to the bottom with less dense elements such as WBC and platelets overlying the RBC layer and finally, plasma at the very top. Apheresis: Principles of Separation Platelets (1040) Lymphocytes Torloni MD (1050-1061) Monocytes (1065 - 1069) Granulocyte (1087 - 1092) RBC Torloni MD Torloni MD Separate blood components is based on density with removal of the desired component Graphics owned by and courtesy of Gambro BCT Principals of Apheresis WBC Plasma Torlo RBC ni MD Torloni MD RBC WBC Plasma G Cobe Spectra Apheresis- Mechanism of Action Definitions • Plasmapheresis: plasma is separated, removed (i.e. less than 15% of total plasma volume) without the use of replacement solution • Plasma exchange (TPE): plasma is separated, removed and replaced with a replacement solution such as colloid (e.g. -

Recommendations for Collecting Red Blood Cells by Automated Apheresis Methods
Guidance for Industry Recommendations for Collecting Red Blood Cells by Automated Apheresis Methods Additional copies of this guidance document are available from: Office of Communication, Training and Manufacturers Assistance (HFM-40) 1401 Rockville Pike, Rockville, MD 20852-1448 (Tel) 1-800-835-4709 or 301-827-1800 (Internet) http://www.fda.gov/cber/guidelines.htm U.S. Department of Health and Human Services Food and Drug Administration Center for Biologics Evaluation and Research (CBER) January 2001 Technical Correction February 2001 TABLE OF CONTENTS Note: Page numbering may vary for documents distributed electronically. I. INTRODUCTION ............................................................................................................. 1 II. BACKGROUND................................................................................................................ 1 III. CHANGES FROM THE DRAFT GUIDANCE .............................................................. 2 IV. RECOMMENDED DONOR SELECTION CRITERIA FOR THE AUTOMATED RED BLOOD CELL COLLECTION PROTOCOLS ..................................................... 3 V. RECOMMENDED RED BLOOD CELL PRODUCT QUALITY CONTROL............ 5 VI. REGISTRATION AND LICENSING PROCEDURES FOR THE MANUFACTURE OF RED BLOOD CELLS COLLECTED BY AUTOMATED METHODS.................. 7 VII. ADDITIONAL REQUIREMENTS.................................................................................. 9 i GUIDANCE FOR INDUSTRY Recommendations for Collecting Red Blood Cells by Automated Apheresis Methods This -

Principles of Blood Separation and Apheresis Instrumentation
Principles of Blood Separation and Apheresis Instrumentation Dobri Kiprov, M.D., H.P. Chief, Division of Immunotherapy, California Pacific Medical Center, San Francisco, CA Medical Director, Apheresis Care Group Apheresis History Apheresis History Apheresis History Apheresis From the Greek - “to take away” Blood separation Donor apheresis Therapeutic apheresis Principles of Blood Separation Filtration Centrifugation Combined centrifugation and filtration Membrane Separation Blood is pumped through a membrane with pores allowing plasma to pass through whilst retaining blood cells. Available as a hollow fiber membrane (older devices used parallel-plate membranes) Pore diameter for plasma separation: 0.2 to 0.6μm. A number of parameters need to be closely controlled Detail of Membrane Separation Courtesy of CaridianBCT Membrane Blood Separation Trans Membrane Pressure (TMP) Too High = Hemolysis TMP Too Low = No Separation Optimal TMP = Good Separation Membrane Apheresis in the US - PrismaFlex (Gambro – Baxter) - NxStage - BBraun Filtration vs. Centrifugation Apheresis Filtration Centrifugation Minimal availability The standard in the in the USA USA • Poor industry support • Very good industry support Limited to plasma Multiple procedures (cytapheresis) exchange • Opportunity to provide • Low efficiency cellular therapies Centrifugation vs. Filtration Apheresis Centrifugation Apheresis Filtration Apheresis Blood Flow 10 – 100 ml/min 150 ml/min Efficiency of Plasma 60 – 65% 30% Removal Apheresis in Clinical Practice and Blood Banking Sickle Cell Disease Falciparum Malaria Thrombocytosis RBC WBC PLT Plasma Leukemias TTP-HUS Cell Therapies Guillain Barre Syndrome Myasthenia Gravis CIDP Autoimmune Renal Disease Hyperviscosity Syndromes Centrifugal Separation Based on the different specific gravity of the blood components. In some instruments, also based on the cellular size (Elutriation). -

Erythrocytapheresis in Sickle Cell Disease)
Apheresis in Hematology (Erythrocytapheresis in Sickle Cell Disease) Haewon C. Kim, M.D. The Children’s Hospital of Philadelphia Perelman School of Medicine University of Pennsylvania Philadelphia, PA, USA Apheresis in Hematology (Erythrocytapheresis in Sickle Cell Disease) Objectives Application of Apheresis in Hematology Overview of Erythrocytapheresis Erythrocytapheresis for Management of Acute and Chronic Complications of Sickle Cell Disease Technical Challenges with Apheresis in Children Indication Categories for TPE in Hematologic Disorders (ASFA 2013 ) Disease TA Category Modality Aplastic anemia; Pure red cell aplasia TPE III Autoimmune hemolytic anemia: - Severe WAHA TPE III - Severe cold agglutinin disease TPE II Catastrophic antiphospholipid syndrome TPE II Coagulation factor inhibitors: Alloantibody TPE IV IA III Coagulation factor inhibitors: Autoantibody TPE III IA III Cryoglobulinemia: Symptomatic/Severe TPE I IA II Heparin-induced thrombocytopenia: - Pre-CPB TPE III - Thrombosis Henoch-Schonlein purpura: - Crescentric TPE III - Severe extrarenal disease Indication Categories for TPE in Hematologic Disorders (ASFA 2013 ) Disease TA Category Modality Hyperviscosity in monoclonal gammopathies: TPE I - Symptomatic, - Prophylaxis for rituximab Immune thrombocytopenia (ITP): Refractory TPE IV IA III Post-transfusion purpura TPE III Red cell alloimmunization in pregnancy TPE III SLE: - Severe TPE II - Nephritis IV TMA, Drug associated: - Ticlopidine TPE I - Clopidogrel, Cyclosporine, Tacrolimus TPE III TMA, HSCT associated, -

Transplantation
Kvalheim_EUHaemotology_EU Haematology 09/03/2010 09:12 Page 24 Transplantation The Use of Plerixafor for Peripheral Blood Stem Cell Mobilisation Reduces the Frequency of Mobilisation Failure in Patients Planned to Undergo Autologous Transplantation Dag Josefsen,1 Catherine Rechnitzer,2 Katriina Parto3 and Gunnar Kvalheim1 1. Department of Oncology, Norwegian Radium Hospital, Oslo University Hospital; 2. Department of Paediatrics, Rigshospitalet, Copenhagen University Hospital; 3. Paediatric Haematology, Tampere University Hospital DOI: 10.17925/EOH.2010.04.0.24 Abstract High-dose chemotherapy with or without radiation followed by autologous haematopoietic stem cell transplantation (auto-HSCT) is now the standard of care for patients with chemosensitive relapsed aggressive non-Hodgkin’s lymphoma (NHL), chemosensitive relapsed Hodgkin’s disease (HD) and multiple myeloma (MM). Autologous haematopoietic stem cells also provide haematopoietic support after the administration of high-dose chemotherapy in relapsed NHL and MM. However, certain patients fail to mobilise a sufficient number of haematopoietic stem cells using standard cytokine-assisted mobilisation strategies. Recently, plerixafor, a novel bicyclam capable of specifically and reversibly binding to the CXCR4 receptor on haematopoietic stem cells, has been granted European approval, in combination with granulocyte colony-stimulating factor, for the enhancement of haematopoietic stem cell mobilisation to the peripheral blood for collection and subsequent autotransplantation in poorly mobilising lymphoma and MM patients. In this article the authors present their initial experience with plerixafor in a case series at their own institutions in Scandinavia. Keywords Autologous, stem cell, mobilisation, granulocyte colony-stimulating factor (G-CSF), autologous stem cell transplantation (auto-SCT), peripheral blood stem cells (PBSCs), chemotherapy, plerixafor Disclosure: The authors have no conflicts of interest to declare. -
Circular of Information for the Use of Human Blood and Blood Components
CIRCULAR OF INFORMATION FOR THE USE OF HUMAN BLOOD Y AND BLOOD COMPONENTS This Circular was prepared jointly by AABB, the AmericanP Red Cross, America’s Blood Centers, and the Armed Ser- vices Blood Program. The Food and Drug Administration recognizes this Circular of Information as an acceptable extension of container labels. CO OT N O Federal Law prohibits dispensing the blood and blood compo- nents describedD in this circular without a prescription. THIS DOCUMENT IS POSTED AT THE REQUEST OF FDA TO PROVIDE A PUBLIC RECORD OF THE CONTENT IN THE OCTOBER 2017 CIRCULAR OF INFORMATION. THIS DOCUMENT IS INTENDED AS A REFERENCE AND PROVIDES: Y • GENERAL INFORMATION ON WHOLE BLOOD AND BLOOD COMPONENTS • INSTRUCTIONS FOR USE • SIDE EFFECTS AND HAZARDS P THIS DOCUMENT DOES NOT SERVE AS AN EXTENSION OF LABELING REQUIRED BY FDA REGUALTIONS AT 21 CFR 606.122. REFER TO THE CIRCULAR OF INFORMATIONO WEB- PAGE AND THE DECEMBER 2O17 FDA GUIDANCE FOR IMPORTANT INFORMATION ON THE CIRCULAR. C T O N O D Table of Contents Notice to All Users . 1 General Information for Whole Blood and All Blood Components . 1 Donors . 1 Y Testing of Donor Blood . 2 Blood and Component Labeling . 3 Instructions for Use . 4 Side Effects and Hazards for Whole Blood and P All Blood Components . 5 Immunologic Complications, Immediate. 5 Immunologic Complications, Delayed. 7 Nonimmunologic Complications . 8 Fatal Transfusion Reactions. O. 11 Red Blood Cell Components . 11 Overview . 11 Components Available . 19 Plasma Components . 23 Overview . 23 Fresh Frozen Plasma . .C . 23 Plasma Frozen Within 24 Hours After Phlebotomy . 28 Components Available . -

Form: Parental Consent for Blood Donation
American Red Cross Biomedical Services Washington, DC 20006 Form: Parental Consent for Blood Donation Information This form must be completed by a parent or legal guardian. Parental permission is required for All donations by 16-year-olds All donations by any age student at high school blood drives in Utah Donations by 17-year-olds as required by state law or blood drive sponsor Before giving consent, please read the information on the back of this form and “A Student’s Guide to Blood Donation.” You should also read “Possible Use of Donor Information and Blood Samples in Medical Research” and the research study sheets for your state, which can be found at https://www.redcrossblood.org/donate-blood/how-to-donate/info-for- student-donors.html. If you do not have internet access, please call the Donor and Client Support Center at 1-866-236-3276 for relevant information regarding research studies. Before donating blood, your child will read “What You Must Know Before Giving Blood,” which describes the blood donation process. It explains the importance of accurate and honest answers to health history questions, what happens when a person gives blood, and tips for having a positive donation experience. It also explains why the Red Cross asks questions about sexual contact and identifies profiles of persons who should not donate (because of physical conditions, travel to certain countries, or high-risk behavior). “What You Must Know Before Giving Blood” contains explicit language defining “sexual contact.” A copy of this document is on file and available for viewing at your child’s school. -

Case Reports
CASE REPORTS units that were group A, D+, E−, K−, and Fy(a−). Severe delayed hemolytic transfusion reaction Although anti-Le a was microscopically positive at poly - due to anti-Fy3 in a patient with sickle cell disease ethylene glycol (PEG) IgG, anti-Lea is not routinely undergoing red cell exchange prior to matched for at the study institution nor had been hematopoietic progenitor cell collection for gene matched for with transfusions at his primary medical therapy facility. After RCE, his Hb rose from 9.3 to 11.2 g/dL, and his HbS% decreased from 78% to 22% (HbA% of 72%). To our knowledge, most patients with sickle cell di- The following day, the patient was given a single 240 sease (SCD) who have undergone hematopoietic progen - µg/kg dose of plerixafor for HPC mobilization and started itor cell (HPC) collection for gene therapy have been con - leukapheresis about 3 hours afterward. He had no ditioned with a series of monthly simple or exchange adverse events and was discharged home after his leuka - transfusions. The rationale for this practice is to improve pheresis. safety and decrease bone marrow inflammation, poten - Five days after RCE, the patient developed throbbing + tially leading to higher CD34 mobilization and low back pain, subjective fevers, dark urine, and scleral decreased risk of adverse events during HPC collection. icterus, symptoms differing from his typical vaso-occlu - + Whether transfusion actually improves CD34 mobiliza - sive crises usually manifesting as right-sided flank/upper tion is not clear however, and we previously showed that back discomfort and priapism. -

Side Effects of Blood Donation by Apheresis Adverse Events Donor
Adverse events Side effects of blood donation by apheresis Adverse events of “blood collections” • Whole blood collections Hans Vrielink, MD, PhD • Apheresis collections Department of Transfusion Medicine • Donors • Adverse events c an be local and / or systemic or both • Events must be registered 2 Donor side effects after WB-donation Donor complications after WB-donation 1000 random donors interview ed 3 w eeks post donation (WB; 500 mL) Adverse Events Incidence % • 36% donors had one or more A E • Female : men = 2:1 (48 vs 23%) Local / Systemic • FD > RD (47 vs 36%) Bruis e / Hematoma 25 • Race affects bruising • Spontaneous reported < solic ited rates Arm pain 10 • Rates differ w ith interview er and used questions Burning Numbness Tingling 1 Fatigue 8 Vasovagal sy mptoms 5 Nausea vomiting 1 3 4 Newman B, et al. Transfusion 2003 Newman B, et al. Transfusion 2003 Effect Adverse Events on Return Rates (RR) Effect Adverse Events on Return Rates (RR) 1000 interview ed random WB-donors number return v isits (follow -up 9-21 Adverse Events % decrease months) Hematoma (inc idence 15%) 0 (n.s.) Estimated overall effect various A E on s ubsequent donation in general Pain in arm (7%) 2 (n.s.) blood donor population = 6% reduction Fatigue (5%) 20 •Greatest impact Vas ovagal sy mptoms Donor reaction (4%) 34 •Combinations: synergistic reducing effect (pain + fatigue: 65 instead 22) Donor reaction + pain arm 35 Pain arm + fatigue 65 Donor reaction + fatigue 66 Donor reaction + fatigue + pain arm 85 Estimated RR without AE: 1.32 visits / yr 5 6 Newman B, et al.