Overnight Storage of Autologous Stem Cell Apheresis Products Before
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
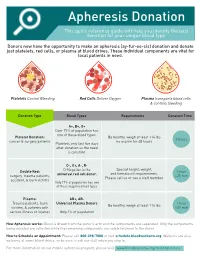
Apheresis Donation This Quick Reference Guide Will Help You Identify the Best Donation for Your Unique Blood Type
Apheresis Donation This quick reference guide will help you identify the best donation for your unique blood type. Donors now have the opportunity to make an apheresis (ay-fur-ee-sis) donation and donate just platelets, red cells, or plasma at blood drives. These individual components are vital for local patients in need. Platelets Control Bleeding Red Cells Deliver Oxygen Plasma transports blood cells & controls bleeding Donation Type Blood Types Requirements Donation Time A+, B+, O+ Over 75% of population has one of these blood types. Platelet Donation: Be healthy, weigh at least 114 lbs 2 hours cancer & surgery patients no aspirin for 48 hours Platelets only last five days after donation so the need is constant. O-, O+, A-, B- Special height, weight, Double Red: O-Negative is the 1 hour and hematocrit requirements. surgery, trauma patients, universal red cell donor. +25 min Please call us or see a staff member accident, & burn victims Only 17% of population has one of these negative blood types Plasma: AB+, AB- Trauma patients, burn Universal Plasma Donors 1 hour Be healthy, weigh at least 114 lbs victims, & patients with +30 min serious illness or injuries Only 4% of population How Apheresis works: Blood is drawn from the donor’s arm and the components are separated. Only the components being donated are collected while the remaining components are safely returned to the donor How to Schedule an Appointment: Please call 800-398-7888 or visit schedule.bloodworksnw.org. Walk-ins are also welcome at some blood drives, so be sure to ask our staff when you stop in. -

Blood Product Modifications: Leukofiltration, Irradiation and Washing
Blood Product Modifications: Leukofiltration, Irradiation and Washing 1. Leukocyte Reduction Definitions and Standards: o Process also known as leukoreduction, or leukofiltration o Applicable AABB Standards, 25th ed. Leukocyte-reduced RBCs At least 85% of original RBCs < 5 x 106 WBCs in 95% of units tested . Leukocyte-reduced Platelet Concentrates: At least 5.5 x 1010 platelets in 75% of units tested < 8.3 x 105 WBCs in 95% of units tested pH≥6.2 in at least 90% of units tested . Leukocyte-reduced Apheresis Platelets: At least 3.0 x 1011 platelets in 90% of units tested < 5.0 x 106 WBCs 95% of units tested pH≥6.2 in at least 90% of units tested Methods o Filter: “Fourth-generation” filters remove 99.99% WBCs o Apheresis methods: most apheresis machines have built-in leukoreduction mechanisms o Less efficient methods of reducing WBC content . Washing, deglycerolizing after thawing a frozen unit, centrifugation . These methods do not meet requirement of < 5.0 x 106 WBCs per unit of RBCs/apheresis platelets. Types of leukofiltration/leukoreduction o “Pre-storage” . Done within 24 hours of collection . May use inline filters at time of collection (apheresis) or post collection o “Pre-transfusion” leukoreduction/bedside leukoreduction . Done prior to transfusion . “Bedside” leukoreduction uses gravity-based filters at time of transfusion. Least desirable given variability in practice and absence of proficiency . Alternatively performed by transfusion service prior to issuing Benefits of leukoreduction o Prevention of alloimmunization to donor HLA antigens . Anti-HLA can mediate graft rejection and immune mediated destruction of platelets o Leukoreduced products are indicated for transplant recipients or patients who are likely platelet transfusion dependent o Prevention of febrile non-hemolytic transfusion reactions (FNHTR) . -

Cord Blood Stem Cell Transplantation
LEUKEMIA LYMPHOMA MYELOMA FACTS Cord Blood Stem Cell Transplantation No. 2 in a series providing the latest information on blood cancers Highlights • Umbilical cord blood, like bone marrow and peripheral blood, is a rich source of stem cells for transplantation. There may be advantages for certain patients to have cord blood stem cell transplants instead of transplants with marrow or peripheral blood stem cells (PBSCs). • Stem cell transplants (peripheral blood, marrow or cord blood) may use the patient’s own stem cells (called “autologous transplants”) or use donor stem cells. Donor cells may come from either a related or unrelated matched donor (called an “allogeneic transplant”). Most transplant physicians would not want to use a baby’s own cord blood (“autologous transplant”) to treat his or her leukemia. This is because donor stem cells might better fight the leukemia than the child’s own stem cells. • Cord blood for transplantation is collected from the umbilical cord and placenta after a baby is delivered. Donated cord blood that meets requirements is frozen and stored at a cord blood bank for future use. • The American Academy of Pediatrics’s (AAP) policy statement (Pediatrics; 2007;119:165-170.) addresses public and private banking options available to parents. Among several recommendations, the report encourages parents to donate to public cord blood banks and discourages parents from using private cord blood banks for personal or family cord blood storage unless they have an older child with a condition that could benefit from transplantation. • The Stem Cell Therapeutic and Research Act of 2005 put several programs in place, including creation of the National Cord Blood Inventory (NCBI) for patients in need of transplantation. -

Therapeutic Apheresis, J Clin Apheresis 2007, 22, 104-105
Apheresis: Basic Principles, Practical Considerations and Clinical Applications Joseph Schwartz, MD Anand Padmanabhan, MD PhD Director, Transfusion Medicine Assoc Med Director/Asst Prof Columbia Univ. Medical Center BloodCenter of Wisconsin New York Presbyterian Hospital Medical College of Wisconsin Review Session, ASFA Annual meeting, Scottsdale, Arizona, June 2011 Objectives (Part 1) • Mechanism of Action • Definitions • Technology (ies) • Use • Practical Considerations • Math • Clinical applications – HPC Collection Objectives (Part 2) • Clinical applications: System/ Disease Specific Indications • ASFA Fact Sheet Apheresis •Derives from Greek, “to carry away” •A technique in which whole blood is taken and separated extracorporealy, separating the portion desired from the remaining blood. •This allows the desired portion (e.g., plasma) to be removed and the reminder returned. Apheresis- Mechanism of Action •Large-bore intravenous catheter connected to a spinning centrifuge bowl •Whole blood is drawn from donor/patient into the centrifuge bowl •The more dense elements, namely the RBC, settle to the bottom with less dense elements such as WBC and platelets overlying the RBC layer and finally, plasma at the very top. Apheresis: Principles of Separation Platelets (1040) Lymphocytes Torloni MD (1050-1061) Monocytes (1065 - 1069) Granulocyte (1087 - 1092) RBC Torloni MD Torloni MD Separate blood components is based on density with removal of the desired component Graphics owned by and courtesy of Gambro BCT Principals of Apheresis WBC Plasma Torlo RBC ni MD Torloni MD RBC WBC Plasma G Cobe Spectra Apheresis- Mechanism of Action Definitions • Plasmapheresis: plasma is separated, removed (i.e. less than 15% of total plasma volume) without the use of replacement solution • Plasma exchange (TPE): plasma is separated, removed and replaced with a replacement solution such as colloid (e.g. -

Time Course of Immune Recovery and Viral Reactivation Following Hematopoietic Stem Cell Transplantation
CLINICAL ARTICLES Cellular Therapy and Transplantation (CTT). Vol.5, No.4 (17), 2016 doi: 10.18620/ctt-1866-8836-2016-5-4-32-43 Submitted:02 November 2016, accepted: 09 December 2016 Time course of immune recovery and viral reactivation following hematopoietic stem cell transplantation 1Olga S. Pankratova, 2Alexei B. Chukhlovin 1Tampere University Hospital, Tampere, Finland 2R. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov Medical University CD8+ cells specific for cytomegalovirus (CMV), or Ep- Summary stein-Barr virus (EBV) rapidly expand in cases of CMV or EBV activation. Total depletion of innate and adaptive immune cell pop- ulations occurs after intensive chemotherapy and he- Despite recovery of absolute B-cell counts by day 30 matopoietic stem cell transplantation (HSCT) then fol- post-HSCT, their functions, i.e., antigen-specific anti- lowed by gradual recovery of immune populations, due body production, are reduced for months and years after to progenitors derived from donor hematopoietic cells HSCT, due to slow restoration of mature immune cell which differentiate to myeloid and lymphoid lineages. populations, thus resemling normal evolution of B cell Time dynamics of immune reconstitution and differen- hierarchy in human organism. tial maturation of distinct immune populations is only partially evaluated, especially, at early terms post-trans- Reactivation of herpesviruses (mostly, CMV, EBV and plant. E.g., innate immunity is restored within 1st month Herpes Simplex) is a known feature of immune de- after HSCT, due to rapid reconstitution of granulocytes, ficiency. Timing of maximal herpesvirus incidence monocytes, and natural killer (NK) cells. -

Recommendations for Collecting Red Blood Cells by Automated Apheresis Methods
Guidance for Industry Recommendations for Collecting Red Blood Cells by Automated Apheresis Methods Additional copies of this guidance document are available from: Office of Communication, Training and Manufacturers Assistance (HFM-40) 1401 Rockville Pike, Rockville, MD 20852-1448 (Tel) 1-800-835-4709 or 301-827-1800 (Internet) http://www.fda.gov/cber/guidelines.htm U.S. Department of Health and Human Services Food and Drug Administration Center for Biologics Evaluation and Research (CBER) January 2001 Technical Correction February 2001 TABLE OF CONTENTS Note: Page numbering may vary for documents distributed electronically. I. INTRODUCTION ............................................................................................................. 1 II. BACKGROUND................................................................................................................ 1 III. CHANGES FROM THE DRAFT GUIDANCE .............................................................. 2 IV. RECOMMENDED DONOR SELECTION CRITERIA FOR THE AUTOMATED RED BLOOD CELL COLLECTION PROTOCOLS ..................................................... 3 V. RECOMMENDED RED BLOOD CELL PRODUCT QUALITY CONTROL............ 5 VI. REGISTRATION AND LICENSING PROCEDURES FOR THE MANUFACTURE OF RED BLOOD CELLS COLLECTED BY AUTOMATED METHODS.................. 7 VII. ADDITIONAL REQUIREMENTS.................................................................................. 9 i GUIDANCE FOR INDUSTRY Recommendations for Collecting Red Blood Cells by Automated Apheresis Methods This -

Chapter 118: Transplantation-Related Malignancies
CHAPTER 118 — REFERENCES 1. Bhatia S, Ramsay NK, Steinbuch M, et al. Malignant neoplasms following 30. Ellis NA, Huo D, Yildiz O, et al. MDM2 SNP309 and TP53 Arg72Pro in- bone marrow transplantation. Blood 1996;87:3633–3639. teract to alter therapy-related acute myeloid leukemia susceptibility. Blood 2. Witherspoon RP, Fisher LD, Schoch G, et al. Secondary cancers after bone 2008;112:741–749. marrow transplantation for leukemia or aplastic anemia. N Engl J Med 31. Casorelli I, Offman J, Mele L, et al. Drug treatment in the development 1989;321:784–789. of mismatch repair defective acute leukemia and myelodysplastic syndrome. 3. Curtis RE, Rowlings PA, Deeg HJ, et al. Solid cancers after bone marrow DNA Repair (Amst) 2003;2:547–559. transplantation. N Engl J Med 1997;336:897–904. 32. Seedhouse CH, Das-Gupta EP, Russell NH. Methylation of the hMLH1 4. Krishnan A, Bhatia S, Slovak ML, et al. Predictors of therapy-related leuke- promoter and its association with microsatellite instability in acute myeloid mia and myelodysplasia following autologous transplantation for lymphoma: leukemia. Leukemia 2003;17:83–88. an assessment of risk factors. Blood 2000;95:1588–1593. 33. Seedhouse C, Faulkner R, Ashraf N, et al. Polymorphisms in genes involved 5. Rowlings PA, Curtis RE, Passweg JR, et al. Increased incidence of Hodg- in homologous recombination repair interact to increase the risk of develop- kin’s disease after allogeneic bone marrow transplantation. J Clin Oncol ing acute myeloid leukemia. Clin Cancer Res 2004;10:2675–2680. 1999;17:3122–3127. 34. Matullo G, Palli D, Peluso M, et al. -

Autotransplantation of the Third Molar: a Therapeutic Alternative to the Rehabilitation of a Missing Tooth: a Scoping Review
bioengineering Review Autotransplantation of the Third Molar: A Therapeutic Alternative to the Rehabilitation of a Missing Tooth: A Scoping Review Mario Dioguardi 1,* , Cristian Quarta 1 , Diego Sovereto 1, Giuseppe Troiano 1 , Michele Melillo 1, Michele Di Cosola 1, Angela Pia Cazzolla 1 , Luigi Laino 2 and Lorenzo Lo Muzio 1 1 Department of Clinical and Experimental Medicine, University of Foggia, Via Rovelli 50, 71122 Foggia, Italy; [email protected] (C.Q.); [email protected] (D.S.); [email protected] (G.T.); [email protected] (M.M.); [email protected] (M.D.C.); [email protected] (A.P.C.); [email protected] (L.L.M.) 2 Multidisciplinary Department of Medical-Surgical and Odontostomatological Specialties, University of Campania “Luigi Vanvitelli”, 80121 Naples, Italy; [email protected] * Correspondence: [email protected] Abstract: Introduction: Tooth autotransplantation is the repositioning of an erupted, partially erupted, or non-erupted autologous tooth from one site to another within the same individual. Several factors influence the success rate of the autotransplant, such as the stage of root development, the morphology of the tooth, the surgical procedure selected, the extraoral time, the shape of the recipient socket, the vascularity of the recipient bed, and the vitality of the cells of the periodontal ligament. The aim of this scoping review was to provide the most up-to-date information and data on the clinical principles of the third-molar autograft and thus provide clinical considerations for its success. Citation: Dioguardi, M.; Quarta, C.; Materials and methods: This review was conducted based on PRISMA-ScR (Preferred Reporting Sovereto, D.; Troiano, G.; Melillo, M.; Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews). -

Principles of Blood Separation and Apheresis Instrumentation
Principles of Blood Separation and Apheresis Instrumentation Dobri Kiprov, M.D., H.P. Chief, Division of Immunotherapy, California Pacific Medical Center, San Francisco, CA Medical Director, Apheresis Care Group Apheresis History Apheresis History Apheresis History Apheresis From the Greek - “to take away” Blood separation Donor apheresis Therapeutic apheresis Principles of Blood Separation Filtration Centrifugation Combined centrifugation and filtration Membrane Separation Blood is pumped through a membrane with pores allowing plasma to pass through whilst retaining blood cells. Available as a hollow fiber membrane (older devices used parallel-plate membranes) Pore diameter for plasma separation: 0.2 to 0.6μm. A number of parameters need to be closely controlled Detail of Membrane Separation Courtesy of CaridianBCT Membrane Blood Separation Trans Membrane Pressure (TMP) Too High = Hemolysis TMP Too Low = No Separation Optimal TMP = Good Separation Membrane Apheresis in the US - PrismaFlex (Gambro – Baxter) - NxStage - BBraun Filtration vs. Centrifugation Apheresis Filtration Centrifugation Minimal availability The standard in the in the USA USA • Poor industry support • Very good industry support Limited to plasma Multiple procedures (cytapheresis) exchange • Opportunity to provide • Low efficiency cellular therapies Centrifugation vs. Filtration Apheresis Centrifugation Apheresis Filtration Apheresis Blood Flow 10 – 100 ml/min 150 ml/min Efficiency of Plasma 60 – 65% 30% Removal Apheresis in Clinical Practice and Blood Banking Sickle Cell Disease Falciparum Malaria Thrombocytosis RBC WBC PLT Plasma Leukemias TTP-HUS Cell Therapies Guillain Barre Syndrome Myasthenia Gravis CIDP Autoimmune Renal Disease Hyperviscosity Syndromes Centrifugal Separation Based on the different specific gravity of the blood components. In some instruments, also based on the cellular size (Elutriation). -

A Nationwide Analysis of Kidney Autotransplantation
A Nationwide Analysis of Kidney Autotransplantation ZHOBIN MOGHADAMYEGHANEH, M.D., MARK H. HANNA, M.D., REZA FAZLALIZADEH, M.D., YOSHITSUGU OBI, M.D., PH.D., CLARENCE E. FOSTER, M.D., MICHAEL J. STAMOS, M.D., HIROHITO ICHII, M.D., PH.D. From the Department of Surgery, University of California, Irvine, School of Medicine, Orange, California There are limited data regarding outcomes of patients underwent kidney autotransplantation. This study aims to investigate outcomes of such patients. The nationwide inpatient sample database was used to identify patients underwent kidney autotransplantation during 2002 to 2012. Multivariate analyses using logistic regression were performed to investigate morbidity predictors. A total of 817 patients underwent kidney autotransplantation from 2002 to 2012. The most common indication of surgery was renal artery pathology (22.7%) followed by ureter pathology (17%). Overall, 97.7 per cent of operations were performed in urban teaching hospitals. The number of procedures from 2008 to 2012 were significantly higher compared with the number of them from 2002 to 2007 (473 vs 345, P < 0.01). The overall mortality and morbidity of patients were 1.3 and 46.2 per cent, respectively. The most common postoperative complications were transplanted kidney failure (10.7%) followed by hemorrhagic complications (9.7%). Obesity [adjusted odds ratio (AOR): 9.62, P < 0.01], fluid and electrolyte disorders (AOR: 3.67, P < 0.01), and preoperative chronic kidney disease (AOR: 1.80, P 5 0.03) were predictors of morbidity in patients. In conclusion, Kidney autotransplantation is associated with low mortality but a high morbidity rate. The most common indications of kidney autotransplantation are renal artery and ureter pathologies, respectively. -

Outpatient Immunosuppressive Drugs Under Medicare
Outpatient Immunosuppressive Drugs Under Medicare July 1991 OTA-H-452 NTIS order #PB92-117720 Recommended Citation: U.S. Congress, Office of Technology Assessment, Outpatient Immunosuppressive Drugs Under Medicare, OTA-H-452 (Washington, DC: U.S. Government Printing Office, Septem- ber 1991). For sale by the U.S. Government Printing Office Superintendent of Documents, Mail Stop: SSOP, Washington, DC 20402-9328” ISBN 0-16 -035315-7 Foreword Of all the astonishing achievements of modern medicine, the ability to successfully transplant a living organ from one human being to another is perhaps one of the most awesome. Immunosuppressive drugs are one of the spectrum of technological advances that have made organ transplants an everyday phenomenon. At the same time, however, transplant recipients’ needs for these drugs have presented Medicare with a continuing policy dilemma, because Medicare does not usually pay for outpatient prescription drugs. In 1984, the year after cyclosporine made its debut onto the health care market, OTA reported to Congress on the likely benefits of the drug for Medicare kidney transplant recipients. The present report, requested by the Senate Committee on Finance in the wake of the repeal of the Medicare Catastrophic Coverage Act, examines Medicare’s current immunosuppressive drug coverage dilemma and the policy tradeoffs it entails for the 1990s. OTA reports would not be possible without the assistance and input of a wide variety of individuals from both the public and the private sectors. OTA staff and contractors gratefully acknowledge the contributions of the many people who provided data, clarified facts, presented views, and reviewed the drafts of this report. -

Xenotransplantation of Ovarian Tissue Into Male
XENOTRANSPLANTATION OF OVARIAN TISSUE INTO MALE IMMUNODEFICIENT MICE by HUGO JOSE HERNANDEZ FONSECA (Under the direction of BENJAMÍN G. BRACKETT) ABSTRACT A male immunodeficient mouse model for transplantation of ovarian tissue was investigated. Bovine and human ovarian tissues were surgically placed either under the kidney capsule or in the subcutaneous spaces of male non obese diabetic (NOD) severe combined immunodeficient (SCID) mice. Time intervals required for development of growing follicles were determined for neonatal and adult bovine ovarian tissue grafts. This interval was much shorter (P <0.01) in adult tissue than in one-week-old calf tissue, i.e. 55 vs 124 days. The increase in the proportion of growing follicles was coincidental with a decrease in the proportion of resting follicles. This increment in the growing follicle populations took place abruptly and was significant by 55 days and by 124 days after transplantation in the adult cow and calf ovarian grafts, respectively. Recovery of oocytes from bovine ovarian grafts was successful. Several immature oocytes were recovered and evidence of maturation in one oocyte was obtained after 24 hours of in vitro maturation. Treatment of host mice with an FSH:LH preparation increased follicular development but did not enhance oocyte recovery rates. Human ovarian tissue grafted under the kidney capsule of intact male NOD SCID mice showed a greater proportion of growing follicles than similar grafts transplanted to the kidney of castrated hosts and to the subcutaneous space of intact hosts. However, no differences in follicular growth and development were detected between the intact/ kidney capsule and the castrated / subcutaneous groups.