Therapeutic Apheresis, J Clin Apheresis 2007, 22, 104-105
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Association Between ABO and Duffy Blood Types and Circulating Chemokines and Cytokines
Genes & Immunity (2021) 22:161–171 https://doi.org/10.1038/s41435-021-00137-5 ARTICLE Association between ABO and Duffy blood types and circulating chemokines and cytokines 1 2 3 4 5 6 Sarah C. Van Alsten ● John G. Aversa ● Loredana Santo ● M. Constanza Camargo ● Troy Kemp ● Jia Liu ● 4 7 8 Wen-Yi Huang ● Joshua Sampson ● Charles S. Rabkin Received: 11 February 2021 / Revised: 30 April 2021 / Accepted: 17 May 2021 / Published online: 8 June 2021 This is a U.S. government work and not under copyright protection in the U.S.; foreign copyright protection may apply 2021, corrected publication 2021 Abstract Blood group antigens are inherited traits that may play a role in immune and inflammatory processes. We investigated associations between blood groups and circulating inflammation-related molecules in 3537 non-Hispanic white participants selected from the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial. Whole-genome scans were used to infer blood types for 12 common antigen systems based on well-characterized single-nucleotide polymorphisms. Serum levels of 96 biomarkers were measured on multiplex fluorescent bead-based panels. We estimated marker associations with blood type using weighted linear or logistic regression models adjusted for age, sex, smoking status, and principal components of p 1234567890();,: 1234567890();,: population substructure. Bonferroni correction was used to control for multiple comparisons, with two-sided values < 0.05 considered statistically significant. Among the 1152 associations tested, 10 were statistically significant. Duffy blood type was associated with levels of CXCL6/GCP2, CXCL5/ENA78, CCL11/EOTAXIN, CXCL1/GRO, CCL2/MCP1, CCL13/ MCP4, and CCL17/TARC, whereas ABO blood type was associated with levels of sVEGFR2, sVEGFR3, and sGP130. -
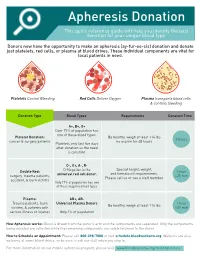
Apheresis Donation This Quick Reference Guide Will Help You Identify the Best Donation for Your Unique Blood Type
Apheresis Donation This quick reference guide will help you identify the best donation for your unique blood type. Donors now have the opportunity to make an apheresis (ay-fur-ee-sis) donation and donate just platelets, red cells, or plasma at blood drives. These individual components are vital for local patients in need. Platelets Control Bleeding Red Cells Deliver Oxygen Plasma transports blood cells & controls bleeding Donation Type Blood Types Requirements Donation Time A+, B+, O+ Over 75% of population has one of these blood types. Platelet Donation: Be healthy, weigh at least 114 lbs 2 hours cancer & surgery patients no aspirin for 48 hours Platelets only last five days after donation so the need is constant. O-, O+, A-, B- Special height, weight, Double Red: O-Negative is the 1 hour and hematocrit requirements. surgery, trauma patients, universal red cell donor. +25 min Please call us or see a staff member accident, & burn victims Only 17% of population has one of these negative blood types Plasma: AB+, AB- Trauma patients, burn Universal Plasma Donors 1 hour Be healthy, weigh at least 114 lbs victims, & patients with +30 min serious illness or injuries Only 4% of population How Apheresis works: Blood is drawn from the donor’s arm and the components are separated. Only the components being donated are collected while the remaining components are safely returned to the donor How to Schedule an Appointment: Please call 800-398-7888 or visit schedule.bloodworksnw.org. Walk-ins are also welcome at some blood drives, so be sure to ask our staff when you stop in. -

Guidelines for Transfusion and Patient Blood Management, and Discuss Relevant Transfusion Related Topics
Guidelines for Transfusion and Community Transfusion Committee Patient Blood Management Community Transfusion Committee CHAIR: Aina Silenieks, M.D., [email protected] MEMBERS: A.Owusu-Ansah, M.D. S. Dunder, M.D. M. Furasek, M.D. D. Lester, M.D. D. Voigt, M.D. B. J. Wilson, M.D. COMMUNITY Juliana Cordero, Blood Bank Coordinator, CHI Health Nebraska Heart REPRESENTATIVES: Becky Croner, Laboratory Services Manager, CHI Health St. Elizabeth Mackenzie Gasper, Trauma Performance Improvement, Bryan Medical Center Kelly Gillaspie, Account Executive, Nebraska Community Blood Bank Mel Hanlon, Laboratory Specialist - Transfusion Medicine, Bryan Medical Center Kyle Kapple, Laboratory Quality Manager, Bryan Medical Center Lauren Kroeker, Nurse Manager, Bryan Medical Center Christina Nickel, Clinical Laboratory Director, Bryan Medical Center Rachael Saniuk, Anesthesia and Perfusion Manager, Bryan Medical Center Julie Smith, Perioperative & Anesthesia Services Director, Bryan Medical Center Elaine Thiel, Clinical Quality Improvement/Trans. Safety Officer, Bryan Med Center Kelley Thiemann, Blood Bank Lead Technologist, CHI Health St. Elizabeth Cheryl Warholoski, Director, Nebraska Operations, Nebraska Community Blood Bank Jackie Wright, Trauma Program Manager, Bryan Medical Center CONSULTANTS: Jed Gorlin, M.D., Innovative Blood Resources [email protected] Michael Kafka, M.D., LifeServe Blood Center [email protected] Alex Smith, D.O., LifeServe Blood Center [email protected] Nancy Van Buren, M.D., Innovative -

Blood Product Modifications: Leukofiltration, Irradiation and Washing
Blood Product Modifications: Leukofiltration, Irradiation and Washing 1. Leukocyte Reduction Definitions and Standards: o Process also known as leukoreduction, or leukofiltration o Applicable AABB Standards, 25th ed. Leukocyte-reduced RBCs At least 85% of original RBCs < 5 x 106 WBCs in 95% of units tested . Leukocyte-reduced Platelet Concentrates: At least 5.5 x 1010 platelets in 75% of units tested < 8.3 x 105 WBCs in 95% of units tested pH≥6.2 in at least 90% of units tested . Leukocyte-reduced Apheresis Platelets: At least 3.0 x 1011 platelets in 90% of units tested < 5.0 x 106 WBCs 95% of units tested pH≥6.2 in at least 90% of units tested Methods o Filter: “Fourth-generation” filters remove 99.99% WBCs o Apheresis methods: most apheresis machines have built-in leukoreduction mechanisms o Less efficient methods of reducing WBC content . Washing, deglycerolizing after thawing a frozen unit, centrifugation . These methods do not meet requirement of < 5.0 x 106 WBCs per unit of RBCs/apheresis platelets. Types of leukofiltration/leukoreduction o “Pre-storage” . Done within 24 hours of collection . May use inline filters at time of collection (apheresis) or post collection o “Pre-transfusion” leukoreduction/bedside leukoreduction . Done prior to transfusion . “Bedside” leukoreduction uses gravity-based filters at time of transfusion. Least desirable given variability in practice and absence of proficiency . Alternatively performed by transfusion service prior to issuing Benefits of leukoreduction o Prevention of alloimmunization to donor HLA antigens . Anti-HLA can mediate graft rejection and immune mediated destruction of platelets o Leukoreduced products are indicated for transplant recipients or patients who are likely platelet transfusion dependent o Prevention of febrile non-hemolytic transfusion reactions (FNHTR) . -

Cord Blood Stem Cell Transplantation
LEUKEMIA LYMPHOMA MYELOMA FACTS Cord Blood Stem Cell Transplantation No. 2 in a series providing the latest information on blood cancers Highlights • Umbilical cord blood, like bone marrow and peripheral blood, is a rich source of stem cells for transplantation. There may be advantages for certain patients to have cord blood stem cell transplants instead of transplants with marrow or peripheral blood stem cells (PBSCs). • Stem cell transplants (peripheral blood, marrow or cord blood) may use the patient’s own stem cells (called “autologous transplants”) or use donor stem cells. Donor cells may come from either a related or unrelated matched donor (called an “allogeneic transplant”). Most transplant physicians would not want to use a baby’s own cord blood (“autologous transplant”) to treat his or her leukemia. This is because donor stem cells might better fight the leukemia than the child’s own stem cells. • Cord blood for transplantation is collected from the umbilical cord and placenta after a baby is delivered. Donated cord blood that meets requirements is frozen and stored at a cord blood bank for future use. • The American Academy of Pediatrics’s (AAP) policy statement (Pediatrics; 2007;119:165-170.) addresses public and private banking options available to parents. Among several recommendations, the report encourages parents to donate to public cord blood banks and discourages parents from using private cord blood banks for personal or family cord blood storage unless they have an older child with a condition that could benefit from transplantation. • The Stem Cell Therapeutic and Research Act of 2005 put several programs in place, including creation of the National Cord Blood Inventory (NCBI) for patients in need of transplantation. -

Recommendations for Collecting Red Blood Cells by Automated Apheresis Methods
Guidance for Industry Recommendations for Collecting Red Blood Cells by Automated Apheresis Methods Additional copies of this guidance document are available from: Office of Communication, Training and Manufacturers Assistance (HFM-40) 1401 Rockville Pike, Rockville, MD 20852-1448 (Tel) 1-800-835-4709 or 301-827-1800 (Internet) http://www.fda.gov/cber/guidelines.htm U.S. Department of Health and Human Services Food and Drug Administration Center for Biologics Evaluation and Research (CBER) January 2001 Technical Correction February 2001 TABLE OF CONTENTS Note: Page numbering may vary for documents distributed electronically. I. INTRODUCTION ............................................................................................................. 1 II. BACKGROUND................................................................................................................ 1 III. CHANGES FROM THE DRAFT GUIDANCE .............................................................. 2 IV. RECOMMENDED DONOR SELECTION CRITERIA FOR THE AUTOMATED RED BLOOD CELL COLLECTION PROTOCOLS ..................................................... 3 V. RECOMMENDED RED BLOOD CELL PRODUCT QUALITY CONTROL............ 5 VI. REGISTRATION AND LICENSING PROCEDURES FOR THE MANUFACTURE OF RED BLOOD CELLS COLLECTED BY AUTOMATED METHODS.................. 7 VII. ADDITIONAL REQUIREMENTS.................................................................................. 9 i GUIDANCE FOR INDUSTRY Recommendations for Collecting Red Blood Cells by Automated Apheresis Methods This -

Principles of Blood Separation and Apheresis Instrumentation
Principles of Blood Separation and Apheresis Instrumentation Dobri Kiprov, M.D., H.P. Chief, Division of Immunotherapy, California Pacific Medical Center, San Francisco, CA Medical Director, Apheresis Care Group Apheresis History Apheresis History Apheresis History Apheresis From the Greek - “to take away” Blood separation Donor apheresis Therapeutic apheresis Principles of Blood Separation Filtration Centrifugation Combined centrifugation and filtration Membrane Separation Blood is pumped through a membrane with pores allowing plasma to pass through whilst retaining blood cells. Available as a hollow fiber membrane (older devices used parallel-plate membranes) Pore diameter for plasma separation: 0.2 to 0.6μm. A number of parameters need to be closely controlled Detail of Membrane Separation Courtesy of CaridianBCT Membrane Blood Separation Trans Membrane Pressure (TMP) Too High = Hemolysis TMP Too Low = No Separation Optimal TMP = Good Separation Membrane Apheresis in the US - PrismaFlex (Gambro – Baxter) - NxStage - BBraun Filtration vs. Centrifugation Apheresis Filtration Centrifugation Minimal availability The standard in the in the USA USA • Poor industry support • Very good industry support Limited to plasma Multiple procedures (cytapheresis) exchange • Opportunity to provide • Low efficiency cellular therapies Centrifugation vs. Filtration Apheresis Centrifugation Apheresis Filtration Apheresis Blood Flow 10 – 100 ml/min 150 ml/min Efficiency of Plasma 60 – 65% 30% Removal Apheresis in Clinical Practice and Blood Banking Sickle Cell Disease Falciparum Malaria Thrombocytosis RBC WBC PLT Plasma Leukemias TTP-HUS Cell Therapies Guillain Barre Syndrome Myasthenia Gravis CIDP Autoimmune Renal Disease Hyperviscosity Syndromes Centrifugal Separation Based on the different specific gravity of the blood components. In some instruments, also based on the cellular size (Elutriation). -

Guide to Blood Plasma Products and Their Applications – Part One
Vet Times The website for the veterinary profession https://www.vettimes.co.uk Guide to blood plasma products and their applications – part one Author : Amanda Boag, Jenny Walton Categories : Vets Date : September 26, 2011 Amanda Boag and Jenny Walton discuss indications for the use of plasma products, their production, and storage within the practice in the first of a three-part series FOLLOWING changes in legislation in 2005, veterinary blood banking and component therapy has developed rapidly within the UK. Practitioners now have easy access to canine blood and its products – notably, packed red cells and various plasma products. This series of articles will focus on indications for plasma product use, their production and storage, the administration and monitoring of plasma transfusions and will include a series of five case studies that have used plasma products. Indications for plasma component therapy Although well-defined indications exist for the use of plasma products in single or multiple coagulation deficiencies, indications for many of its other uses are empiric. Haemostatic disorders Plasma components are most commonly used to treat haemostatic disorders and are generally effective in treating both inherited and acquired conditions. Specific diagnosis enables selection of the optimum replacement product. It is important to remember that any samples drawn for diagnostic purposes (for example, measurement of clotting factor levels) must be taken either 1 / 5 before or at least 36 hours after transfusion to allow the measurement of endogenous levels. Inherited bleeding disorders Plasma components are administered to control active haemorrhage or as preoperative prophylaxis. Inherited bleeding disorders are associated with deficiency of a specific factor and the optimal product for treatment depends on which factor is lacking. -

Blood Collection and Handling Tube Additives, Tube Type Most Laboratory Tests Are Performed on Plasma, Serum, Or Whole Blood
Blood Collection and Handling Tube Additives, Tube Type Most laboratory tests are performed on plasma, serum, or whole blood. To preserve the specimen in the form required by the test, collection tubes contain additives that either prevent coagulation (for plasma and whole blood recovery), or activate coagulation (for serum recovery). Please refer to individual test requirements. Drawing Order When multiple tubes are drawn, it is important to prioritize the drawing order to prevent a Table 1. Vacutainer Order Of Draw tube additive from contaminating the next tube and altering the chemical composition of the 1. Navy Blue (metals testing) following specimen. Coagulation tests are highly susceptible to interference from 2. Blood culture bottles / SPS tubes contamination from tissue fluid and tube additives; therefore these tests are usually collected 3. Coagulation tests: first when a series of tubes are collected. Prior to collecting tests for coagulation (i.e. Blue top a. Clear top “waste” tube tube) a plain Clear top tube containing no additive must be partially filled and discarded. b. Light Blue top This “waste” tube prevents tissue thromboplastins from contaminating the Blue top tube. Blue top tubes must be allowed to fill to the line indicated on the tube, exhausting the 4. Gold top vacuum. See Table 1, “Vacutainer Order of Draw” for proper collection order of vacutainer 5. Plain Red top tubes. 6. Dark Green top (heparin) 7. Light Green top Certain blood collection techniques have been identified as possible sources of error in 8. Lavender or Pink top laboratory testing. Avoid the following sources of test error when collecting blood: 9. -

A Fatal Case of Severe Hemolytic Disease of Newborn Associated with Anti-Jkb
J Korean Med Sci 2006; 21: 151-4 Copyright � The Korean Academy ISSN 1011-8934 of Medical Sciences A Fatal Case of Severe Hemolytic Disease of Newborn Associated with Anti-Jkb The Kidd blood group is clinically significant since the Jk antibodies can cause acute Won Duck Kim, Young Hwan Lee* and delayed transfusion reactions as well as hemolytic disease of newborn (HDN). In general, HDN due to anti-Jkb incompatibility is rare and it usually displays mild Department of Pediatrics, Dongguk University, College of Medicine, Gyeongju; Department of Pediatrics*, clinical symptoms with a favorable prognosis. Yet, we apparently experienced the Yeungnam University, College of Medicine, Daegu, second case of HDN due to anti-Jkb with severe clinical symptoms and a fatal out- Korea come. A female patient having the AB, Rh(D)-positive boodtype was admitted for jaundice on the fourth day after birth. At the time of admission, the patient was lethar- gic and exhibited high pitched crying. The laboratory data indicated a hemoglobin value of 11.4 mg/dL, a reticulocyte count of 14.9% and a total bilirubin of 46.1 mg/dL, Received : 18 October 2004 a direct bilirubin of 1.1 mg/dL and a strong positive result (+++) on the direct Coomb’s Accepted : 11 February 2005 test. As a result of the identification of irregular antibody from the maternal serum, anti-Jkb was detected, which was also found in the eluate made from infant’s blood. Despite the aggressive treatment with exchange transfusion and intensive photother- apy, the patient died of intractable seizure and acute renal failure on the fourth day Address for correspondence of admission. -

05-2019 Human Plasma Fraction.Pdf
ANNEX 4 WHO RECOMMENDATIONS FOR THE PRODUCTION, CONTROL AND REGULATION OF HUMAN PLASMA FOR FRACTIONATION Adopted by the 56th meeting of the WHO Expert Committee on Biological Standardization, 24-28 October 2005. A definitive version of this document, which will differ from this version in editorial but not scientific detail, will be published in the WHO Technical Report Series. Page 2 TABLE OF CONTENTS Page TABLE OF CONTENTS................................................................................................................. INTRODUCTION............................................................................................................................ 1 INTERNATIONAL BIOLOGICAL REFERENCE PREPARATIONS ............................ 2 LIST OF ABBREVIATIONS AND DEFINITION USED ................................................... 3 GENERAL CONSIDERATIONS........................................................................................... 3.1 Range of products made from human blood and plasma ....................................................... 3.2 Composition of human plasma............................................................................................... 3.3 Pathogens present in blood and plasma.................................................................................. 4 MEASURES TO EXCLUDE INFECTIOUS DONATIONS ............................................... 4.1 Appropriate selection of blood/plasma donors....................................................................... 4.2 Screening of -

There Are Four Basic Components That Comprise Human Blood: Plasma, Red Blood Cells, White Blood Cells and Platelets
There are four basic components that comprise human blood: plasma, red blood cells, white blood cells and platelets. Red Blood Cells Red blood cells represent 40%-45% of your blood volume. They are generated from your bone marrow at a rate of four to five billion per hour. They have a lifecycle of about 120 days in the body. Platelets Platelets are an amazing part of your blood. Platelets are the smallest of our blood cells and literally look like small plates in their non-active form. Platelets control bleeding. Wherever a wound occurs, the blood vessel will send out a signal. Platelets receive that signal and travel to the area and transform into their “active” formation, growing long tentacles to make contact with the vessel and form clusters to plug the wound until it heals. Plasma Plasma is the liquid portion of your blood. Plasma is yellowish in color and is made up mostly of water, but it also contains proteins, sugars, hormones and salts. It transports water and nutrients to your body’s tissues. It is made up of: • Hormones. • Antibodies. • Enzymes. • Glucose. • Fat particles. • Salts. White Blood Cells Although white blood cells (leukocytes) only account for about 1% of your blood, they are very important. White blood cells are essential for good health and protection against illness and disease. Like red blood cells, they are constantly being generated from your bone marrow. They flow through the bloodstream and attack foreign bodies, like viruses and bacteria. They can even leave the bloodstream to extend the fight into tissue.