Principles of Blood Separation and Apheresis Instrumentation
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

27. Clinical Indications for Cryoprecipitate And
27. CLINICAL INDICATIONS FOR CRYOPRECIPITATE AND FIBRINOGEN CONCENTRATE Cryoprecipitate is indicated in the treatment of fibrinogen deficiency or dysfibrinogenaemia.1 Fibrinogen concentrate is licenced for the treatment of acute bleeding episodes in patients with congenital fibrinogen deficiency, including afibrinogenaemia and hypofibrinogenaemia,2 and is currently funded under the National Blood Agreement. Key messages y Fibrinogen is an essential component of the coagulation system, due to its role in initial platelet aggregation and formation of a stable fibrin clot.3 y The decision to transfuse cryoprecipitate or fibrinogen concentrate to an individual patient should take into account the relative risks and benefits.3 y The routine use of cryoprecipitate or fibrinogen concentrate is not advised in medical or critically ill patients.2,4 y Cryoprecipitate or fibrinogen concentrate may be indicated in critical bleeding if fibrinogen levels are not maintained using FFP. In the setting of major obstetric haemorrhage, early administration of cryoprecipitate or fibrinogen concentrate may be necessary.3 Clinical implications y The routine use of cryoprecipitate or fibrinogen concentrate in medical or critically ill patients with coagulopathy is not advised. The underlying causes of coagulopathy should be identified; where transfusion is considered necessary, the risks and benefits should be considered for each patient. Specialist opinion is advised for the management of disseminated intravascular coagulopathy (MED-PP18, CC-PP7).2,4 y Cryoprecipitate or fibrinogen concentrate may be indicated in critical bleeding if fibrinogen levels are not maintained using FFP. In patients with critical bleeding requiring massive transfusion, suggested doses of blood components is 3-4g (CBMT-PP10)3 in adults or as per the local Massive Transfusion Protocol. -
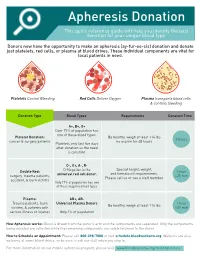
Apheresis Donation This Quick Reference Guide Will Help You Identify the Best Donation for Your Unique Blood Type
Apheresis Donation This quick reference guide will help you identify the best donation for your unique blood type. Donors now have the opportunity to make an apheresis (ay-fur-ee-sis) donation and donate just platelets, red cells, or plasma at blood drives. These individual components are vital for local patients in need. Platelets Control Bleeding Red Cells Deliver Oxygen Plasma transports blood cells & controls bleeding Donation Type Blood Types Requirements Donation Time A+, B+, O+ Over 75% of population has one of these blood types. Platelet Donation: Be healthy, weigh at least 114 lbs 2 hours cancer & surgery patients no aspirin for 48 hours Platelets only last five days after donation so the need is constant. O-, O+, A-, B- Special height, weight, Double Red: O-Negative is the 1 hour and hematocrit requirements. surgery, trauma patients, universal red cell donor. +25 min Please call us or see a staff member accident, & burn victims Only 17% of population has one of these negative blood types Plasma: AB+, AB- Trauma patients, burn Universal Plasma Donors 1 hour Be healthy, weigh at least 114 lbs victims, & patients with +30 min serious illness or injuries Only 4% of population How Apheresis works: Blood is drawn from the donor’s arm and the components are separated. Only the components being donated are collected while the remaining components are safely returned to the donor How to Schedule an Appointment: Please call 800-398-7888 or visit schedule.bloodworksnw.org. Walk-ins are also welcome at some blood drives, so be sure to ask our staff when you stop in. -

Blood Product Modifications: Leukofiltration, Irradiation and Washing
Blood Product Modifications: Leukofiltration, Irradiation and Washing 1. Leukocyte Reduction Definitions and Standards: o Process also known as leukoreduction, or leukofiltration o Applicable AABB Standards, 25th ed. Leukocyte-reduced RBCs At least 85% of original RBCs < 5 x 106 WBCs in 95% of units tested . Leukocyte-reduced Platelet Concentrates: At least 5.5 x 1010 platelets in 75% of units tested < 8.3 x 105 WBCs in 95% of units tested pH≥6.2 in at least 90% of units tested . Leukocyte-reduced Apheresis Platelets: At least 3.0 x 1011 platelets in 90% of units tested < 5.0 x 106 WBCs 95% of units tested pH≥6.2 in at least 90% of units tested Methods o Filter: “Fourth-generation” filters remove 99.99% WBCs o Apheresis methods: most apheresis machines have built-in leukoreduction mechanisms o Less efficient methods of reducing WBC content . Washing, deglycerolizing after thawing a frozen unit, centrifugation . These methods do not meet requirement of < 5.0 x 106 WBCs per unit of RBCs/apheresis platelets. Types of leukofiltration/leukoreduction o “Pre-storage” . Done within 24 hours of collection . May use inline filters at time of collection (apheresis) or post collection o “Pre-transfusion” leukoreduction/bedside leukoreduction . Done prior to transfusion . “Bedside” leukoreduction uses gravity-based filters at time of transfusion. Least desirable given variability in practice and absence of proficiency . Alternatively performed by transfusion service prior to issuing Benefits of leukoreduction o Prevention of alloimmunization to donor HLA antigens . Anti-HLA can mediate graft rejection and immune mediated destruction of platelets o Leukoreduced products are indicated for transplant recipients or patients who are likely platelet transfusion dependent o Prevention of febrile non-hemolytic transfusion reactions (FNHTR) . -

Characterization of Buffy Coat-Derived Granulocytes for Clinical
UvA-DARE (Digital Academic Repository) Neutrophil defects and deficiencies van de Geer, A. Publication date 2020 Document Version Other version License Other Link to publication Citation for published version (APA): van de Geer, A. (2020). Neutrophil defects and deficiencies. General rights It is not permitted to download or to forward/distribute the text or part of it without the consent of the author(s) and/or copyright holder(s), other than for strictly personal, individual use, unless the work is under an open content license (like Creative Commons). Disclaimer/Complaints regulations If you believe that digital publication of certain material infringes any of your rights or (privacy) interests, please let the Library know, stating your reasons. In case of a legitimate complaint, the Library will make the material inaccessible and/or remove it from the website. Please Ask the Library: https://uba.uva.nl/en/contact, or a letter to: Library of the University of Amsterdam, Secretariat, Singel 425, 1012 WP Amsterdam, The Netherlands. You will be contacted as soon as possible. UvA-DARE is a service provided by the library of the University of Amsterdam (https://dare.uva.nl) Download date:01 Oct 2021 Chapter 6 1 Characterization of buffy-coat-derived granulocytes for clinical use: a 2 comparison with granulocyte-colony stimulating factor/dexamethasone- pretreated donor-derived products. 3 Annemarie van de Geer1, Roel P. Gazendam1, Anton T.J. Tool1, John L. van Hamme1, Dirk de Korte1,2, Timo K. van den Berg1, Sacha S. Zeerleder3,4 and Taco W. Kuijpers1,5. 4 1. Dept. of Blood Cell Research, Sanquin Research, Amsterdam, The Netherlands 2. -

Patient Information Leaflet – Plasma Exchange Procedure
Therapeutic Apheresis Services Patient Information Leaflet – Plasma Exchange Procedure Introduction Antibodies, which normally help to protect you from infection, can begin to attack your own This leaflet has been written to give patients healthy cells, or an over production of proteins can information about plasma exchange (sometimes cause your blood to become thicker and slow down called plasmapheresis). If you would like any more the blood flow throughout your body. A plasma information or have any questions, please ask the exchange can help improve your symptoms, doctors and nurses involved in your treatment at although this may not happen immediately. the NHS Blood and Transplant (NHSBT) Therapeutic Apheresis Services Unit. Although plasma exchange may help with symptoms, it will not normally cure your condition When you have considered the information given as it does not switch off the production of the in this leaflet, and after we have discussed the harmful antibodies or proteins. It is likely that procedure and its possible risks with you, we will this procedure will form only one part of your ask you to sign a consent form to indicate that you treatment. are happy for the procedure to go ahead. Before any further procedures we will again check that you are happy to proceed. How do we perform Plasma Exchange? What is a plasma exchange? Plasma exchange is performed using a machine Blood is made up of red cells, white cells and called a Blood Cell Separator which can separate platelets which are carried around in fluid called blood into its various parts. The machine separates plasma. -

Your Blood Cells
Page 1 of 2 Original Date The Johns Hopkins Hospital Patient Information 12/00 Oncology ReviseD/ RevieweD 6/14 Your Blood Cells Where are Blood cells are made in the bone marrow. The bone marrow blood cells is a liquid that looks like blood. It is found in several places of made? the body, such as your hips, chest bone and the middle part of your arm and leg bones. What types of • The three main types of blood cells are the red blood cells, blood cells do the white blood cells and the platelets. I have? • Red blood cells carry oxygen to all parts of the body. The normal hematocrit (or percentage of red blood cells in the blood) is 41-53%. Anemia means low red blood cells. • White blood cells fight infection. The normal white blood cell count is 4.5-11 (K/cu mm). The most important white blood cell to fight infection is the neutrophil. Forty to seventy percent (40-70%) of your white blood cells should be neutrophils. Neutropenia means your neutrophils are low, or less than 40%. • Platelets help your blood to clot and stop bleeding. The normal platelet count is 150-350 (K/cu mm). Thrombocytopenia means low platelets. How do you Your blood cells are measured by a test called the Complete measure my Blood Count (CBC) or Heme 8/Diff. You may want to keep track blood cells? of your blood counts on the back of this sheet. What When your blood counts are low, you may become anemic, get happens infections and bleed or bruise easier. -

Important Information for Female Platelet Donors
Important Information for Female Platelet Donors We are grateful for the support you provide our community blood program and especially appreciate your willingness to help save lives as a volunteer platelet donor. We also take our responsibility to provide a safe and adequate blood supply very seriously and need to share the following information regarding a change to our donor eligibility criteria for female platelet donors. We recently began performing a Human Leukocyte Antigen (HLA) antibody test on each of our current female platelet donors who have ever been pregnant. In addition, we modified our Medical History Questionnaire to ask donors whether they have been pregnant since their last donation. Platelet donors who respond yes to that question will be screened for HLA antibodies. Platelet donors will also be retested after every subsequent pregnancy. These adjustments are being made as part of our effort to reduce occurrences of Transfusion-Related Acute Lung Injury (TRALI). TRALI is a rare but serious complication of blood transfusions most commonly thought to be caused by a reaction to HLA antibodies present in the donor’s plasma. When transfused, these antibodies can sometimes cause plasma to leak into the patient’s lungs, creating fluid accumulation — a condition referred to as acute pulmonary edema. Female donors who have been pregnant are more likely than others to have these HLA antibodies in their plasma. Once the antibodies develop, they are present forever. The antibodies could be harmful if transfused into certain patients. The antibodies are present in plasma — and platelet donations contain a high volume of plasma, so our current efforts are directed at screening blood samples from female platelet donors to test for the HLA antibody. -

FACTS ABOUT DONATING Blood WHY SHOULD I DONATE BLOOD? HOW MUCH BLOOD DO I HAVE? Blood Donors Save Lives
FACTS ABOUT DONATING BLOOD WHY SHOULD I DONATE BLOOD? HOW MUCH BLOOD DO I HAVE? Blood donors save lives. Volunteer blood donors provide An adult has about 10–11 pints. 100 percent of our community’s blood supply. Almost all of us will need blood products. HOW MUCH BLOOD WILL I donate? Whole blood donors give 500 milliliters, about one pint. WHO CAN DONATE BLOOD? Generally, you are eligible to donate if you are 16 years of WHAT HAPPENS TO BLOOD AFTER I donate? age or older, weigh at least 110 pounds and are in good Your blood is tested, separated into components, then health. Donors under 18 must have written permission distributed to local hospitals and trauma centers for from a parent or guardian prior to donation. patient transfusions. DOES IT HURT TO GIVE BLOOD? WHAT ARE THE MAIN BLOOD COMPONENTS? The sensation you feel is similar to a slight pinch on the Frequently transfused components include red blood arm. The process of drawing blood should take less than cells, which replace blood loss in patients during surgery ten minutes. or trauma; platelets, which are often used to control or prevent bleeding in surgery and trauma patients; and HOW DO I PREPARE FOR A BLOOD DONATION? plasma, which helps stop bleeding and can be used to We recommend that donors be well rested, eat a healthy treat severe burns. Both red blood cells and platelets meal, drink plenty of fluids and avoid caffeine and are often used to support patients undergoing cancer alcohol prior to donating. treatments. WHAT CAN I EXPECT WHEN I DONATE? WHEN CAN I DONATE AGAIN? At every donation, you will fill out a short health history You can donate whole blood every eight weeks, platelets questionaire. -

FDA Regulation of Blood and Blood Components in the United States
FDA Regulation of Blood and Blood Components in the United States SLIDE 1 This presentation will review the FDA Regulation of Blood and Blood Components in the U.S. SLIDE 2 The FDA Center for Biologics Evaluation and Research, or CBER, Office of Blood Research and Review, called OBRR, reviews several different types of regulatory applications with respect to blood and blood components. This includes biologics license applications, called BLAs, which represent the regulatory pathway for blood components. The BLA regulatory process also applies to biological drugs such as fractionated plasma products. OBRR also regulates in-vitro diagnostic devices used for screening of collected blood, and does so also using the BLA regulatory pathway. Review of these devices as biologic licenses allows CBER to apply a higher level of manufacturing oversight, including lot release testing and pre-licensure inspection. This regulatory pathway applies to infectious disease tests for blood screening, as well as blood grouping and phenotyping reagents. SLIDE 3 The regulatory pathway for New Drug Applications, or NDAs, is most commonly used within the Center for Drug Evaluation and Research, or CDER. Within the Office of Blood, several NDA applications are reviewed each year, mostly involving solutions used for the collection of blood, such as anticoagulants and red cell nutritive solutions. Interestingly, a blood bag that does not have a solution inside is regulated as a device. If the bag has a solution, then it is a drug-device combination product, but is regulated as a drug. SLIDE 4 OBRR reviews both Class Two and Class Three devices. Class Three devices in OBRR include diagnostic tests for HIV regulated as pre-market approvals, called PMAs. -

Human ICAM-4 Antibody
Human ICAM-4 Antibody Monoclonal Mouse IgG2B Clone # 729632 Catalog Number: MAB8397 DESCRIPTION Species Reactivity Human Specificity Detects human ICAM4 in direct ELISAs. Source Monoclonal Mouse IgG2B Clone # 729632 Purification Protein A or G purified from hybridoma culture supernatant Immunogen Chinese hamster ovary cell line CHOderived recombinant human ICAM4 Ala23Ala240 Accession # Q14773 Formulation Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details. *Small pack size (SP) is supplied either lyophilized or as a 0.2 μm filtered solution in PBS. APPLICATIONS Please Note: Optimal dilutions should be determined by each laboratory for each application. General Protocols are available in the Technical Information section on our website. Recommended Sample Concentration Flow Cytometry 0.25 µg/106 cells See Below CyTOFready Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation. DATA Flow Cytometry Detection of ICAM4 in Human Red Blood Cells by Flow Cytometry. Human red blood cells were stained with Mouse Anti Human ICAM4 Monoclonal Antibody (Catalog # MAB8397, filled histogram) or isotype control antibody (Catalog # MAB0041, open histogram), followed by Allophycocyaninconjugated AntiMouse IgG Secondary Antibody (Catalog # F0101B). PREPARATION AND STORAGE Reconstitution Reconstitute at 0.5 mg/mL in sterile PBS. Shipping The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. *Small pack size (SP) is shipped with polar packs. Upon receipt, store it immediately at 20 to 70 °C Stability & Storage Use a manual defrost freezer and avoid repeated freezethaw cycles. -

(2019). Manufacturing Variables and Hemostatic Function of Cold Stored Platelets: a Systematic Review of the Literature
Scorer, T., Williams, A., Reddoch-Cardenas, K., & Mumford, A. (2019). Manufacturing Variables and Hemostatic Function of Cold Stored Platelets: A Systematic review of the Literature. Transfusion, 59(8), 2722 - 2732. https://doi.org/10.1111/trf.15396 Peer reviewed version Link to published version (if available): 10.1111/trf.15396 Link to publication record in Explore Bristol Research PDF-document This is the author accepted manuscript (AAM). The final published version (version of record) is available online via Wiley at 10.1111/trf.15396 . Please refer to any applicable terms of use of the publisher. University of Bristol - Explore Bristol Research General rights This document is made available in accordance with publisher policies. Please cite only the published version using the reference above. Full terms of use are available: http://www.bristol.ac.uk/red/research-policy/pure/user-guides/ebr-terms/ Manufacturing Variables and Hemostatic Function of Cold Stored Platelets: A Systematic review of the Literature Thomas Scorer1,2,3 Ashleigh Williams4 Kristin Reddoch-Cardenas3 Andrew Mumford1 1. School of Cellular and Molecular Medicine, University of Bristol, Bristol, UK 2. Centre of Defence Pathology, RCDM, Birmingham, UK 3. Coagulation and Blood Research, U.S. Army Institute of Surgical Research, JBSA Ft Sam Houston, Texas, USA. 4. Department of Anaesthesia, Derriford Hospital, Plymouth Word count: 4352 Abstract word count - 250 Number of figures and tables: 3 figures and 5 tables References: 54 Corresponding author: Dr Tom Scorer, Research Floor Level 7, University of Bristol, Bristol Royal Infirmary, Bristol, BS2 8HW United Kingdom. Email: [email protected] The authors have no relevant conflicts of interest to declare. -

Cord Blood Stem Cell Transplantation
LEUKEMIA LYMPHOMA MYELOMA FACTS Cord Blood Stem Cell Transplantation No. 2 in a series providing the latest information on blood cancers Highlights • Umbilical cord blood, like bone marrow and peripheral blood, is a rich source of stem cells for transplantation. There may be advantages for certain patients to have cord blood stem cell transplants instead of transplants with marrow or peripheral blood stem cells (PBSCs). • Stem cell transplants (peripheral blood, marrow or cord blood) may use the patient’s own stem cells (called “autologous transplants”) or use donor stem cells. Donor cells may come from either a related or unrelated matched donor (called an “allogeneic transplant”). Most transplant physicians would not want to use a baby’s own cord blood (“autologous transplant”) to treat his or her leukemia. This is because donor stem cells might better fight the leukemia than the child’s own stem cells. • Cord blood for transplantation is collected from the umbilical cord and placenta after a baby is delivered. Donated cord blood that meets requirements is frozen and stored at a cord blood bank for future use. • The American Academy of Pediatrics’s (AAP) policy statement (Pediatrics; 2007;119:165-170.) addresses public and private banking options available to parents. Among several recommendations, the report encourages parents to donate to public cord blood banks and discourages parents from using private cord blood banks for personal or family cord blood storage unless they have an older child with a condition that could benefit from transplantation. • The Stem Cell Therapeutic and Research Act of 2005 put several programs in place, including creation of the National Cord Blood Inventory (NCBI) for patients in need of transplantation.