Outpatient Blood Platelet Orders
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Guidelines for Transfusions
Guidelines for Transfu- sion Prepared by: Community Transfusion Committee Lincoln, Nebraska CHAIR: Aina Silenieks, MD MEMBERS: L. Bausch, M.D. R. Burton, M.D. S. Dunder, M.D. D. Voigt, M.D. B. J. Wilson, M.D. COMMUNITY Becky Croner REPRESENTATIVES: Ellen DiSalvo Christa Engel Phyllis Ericson Kelly Gillaspie Pat Gilles Vic Grdina Jessica Henrichs Kelly Jensen Laurel McReynolds Christina Nickel Angela Novotny Kelley Thiemann Janet Wachter Jodi Wikoff Guidelines For Transfusion Community Transfusion Committee INTRODUCTION The Community Transfusion Committee is a multidisciplinary group that meets to monitor blood utilization practices, establish guidelines for transfusion and discuss relevant transfusion related topics. It is comprised of physicians from local hospitals, invited guests, and community representatives from the hospitals’ transfusion services, nursing services, perfusion services, health information management, and the Nebraska Community Blood Bank. These Guidelines for Transfusion are reviewed and revised biannually by the Community Trans- fusion Committee to ensure that the industry’s most current practices are promoted. The Guidelines are the standard by which utilization practices are evaluated. They are also de- signed to provide helpful information to assist physicians to provide appropriate blood compo- nent therapy to patients. Appendices have been added for informational purposes and are not to be used as guidance for clinical decision making. ADULT RED CELLS A. Indications 1. One of the following a. Hypovolemia and hypoxia (signs/symptoms: syncope, dyspnea, postural hypoten- sion, tachycardia, angina, or TIA) secondary to surgery, trauma, GI tract bleeding, or intravascular hemolysis, OR b. Evidence of acute loss of 15% of total blood volume or >750 mL blood loss, OR c. -

Platelet Transfusion
Lab Dept: Transfusion Services Test Name: PLATELET TRANSFUSION General Information Lab Order Codes: TPLT Special charges: HLA Matched-MHLA, AHLA PLA1 Negative – PLAT, APLA1 Platelet Crossmatching PLTX – CPLT Synonyms: Leukocyte Reduced Platelets; Random Platelets; Platelet Rich Plasma; Platelet concentrate; Leukocyte Reduced Platelet Pheresis; Single- Donor Platelets; SDP Platelet pheresis; Apheresis Platelets; LRPH CPT Codes: P9035 – Platelet pheresis, Leukocyte reduced 86806 – Lymphocytotoxicity assay, visual crossmatch, without titration 86813 – HLA Matched 86945 – Irradiation 86985 – Volume Reduction 86965 – Pooling 86903 – Platelet Antigen typing 86999 – Washing 86022 – Platelet Crossmatch Test Includes: Leukocyte Reduced Platelet Pheresis consists of platelets suspended in 200-300 mL of plasma collected by cytapheresis. Each unit contains at least 3 x 1011 platelets, and ≤5.0 x 106 leukocytes. One pheresis unit equals 5-6 random unit platelet concentrate. Logistics Test Indications: Refer to Guidelines for the Transfusion of Blood Components. Platelet Crossmatching or HLA-Matched Platelets may be useful for patients receiving repeated platelet transfusions who have become refractile, and for patients who repeatedly develop febrile non-hemolytic transfusion reactions after platelet transfusions. Volume Reduced Platelets are indicated in the event that ABO - incompatible platelets must be transfused due to availability or in patients with severe fluid restrictions. Refer to Blood Component Compatibility Chart Lab Testing Sections: Transfusion Services Phone Numbers: MIN Lab: 612-813-6824 STP Lab: 651-220-6558 Test Availability: Daily, 24 hours Turnaround Time: 1 - 2 hours Standard Dose/Volume: <10 kg: 10 – 15 mL/kg up to one unit or 50 mL maximum 10 – 15 kg: 1/3 pheresis unit 15 – 25 kg: 1/2 pheresis unit >25 kg: 1 pheresis unit Rate of Infusion: 10 minutes/unit, <4 hours Administration: Must be administered through a blood component administration filter. -
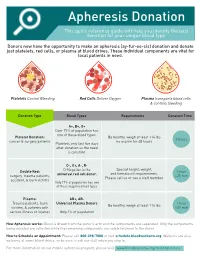
Apheresis Donation This Quick Reference Guide Will Help You Identify the Best Donation for Your Unique Blood Type
Apheresis Donation This quick reference guide will help you identify the best donation for your unique blood type. Donors now have the opportunity to make an apheresis (ay-fur-ee-sis) donation and donate just platelets, red cells, or plasma at blood drives. These individual components are vital for local patients in need. Platelets Control Bleeding Red Cells Deliver Oxygen Plasma transports blood cells & controls bleeding Donation Type Blood Types Requirements Donation Time A+, B+, O+ Over 75% of population has one of these blood types. Platelet Donation: Be healthy, weigh at least 114 lbs 2 hours cancer & surgery patients no aspirin for 48 hours Platelets only last five days after donation so the need is constant. O-, O+, A-, B- Special height, weight, Double Red: O-Negative is the 1 hour and hematocrit requirements. surgery, trauma patients, universal red cell donor. +25 min Please call us or see a staff member accident, & burn victims Only 17% of population has one of these negative blood types Plasma: AB+, AB- Trauma patients, burn Universal Plasma Donors 1 hour Be healthy, weigh at least 114 lbs victims, & patients with +30 min serious illness or injuries Only 4% of population How Apheresis works: Blood is drawn from the donor’s arm and the components are separated. Only the components being donated are collected while the remaining components are safely returned to the donor How to Schedule an Appointment: Please call 800-398-7888 or visit schedule.bloodworksnw.org. Walk-ins are also welcome at some blood drives, so be sure to ask our staff when you stop in. -

Blood Product Modifications: Leukofiltration, Irradiation and Washing
Blood Product Modifications: Leukofiltration, Irradiation and Washing 1. Leukocyte Reduction Definitions and Standards: o Process also known as leukoreduction, or leukofiltration o Applicable AABB Standards, 25th ed. Leukocyte-reduced RBCs At least 85% of original RBCs < 5 x 106 WBCs in 95% of units tested . Leukocyte-reduced Platelet Concentrates: At least 5.5 x 1010 platelets in 75% of units tested < 8.3 x 105 WBCs in 95% of units tested pH≥6.2 in at least 90% of units tested . Leukocyte-reduced Apheresis Platelets: At least 3.0 x 1011 platelets in 90% of units tested < 5.0 x 106 WBCs 95% of units tested pH≥6.2 in at least 90% of units tested Methods o Filter: “Fourth-generation” filters remove 99.99% WBCs o Apheresis methods: most apheresis machines have built-in leukoreduction mechanisms o Less efficient methods of reducing WBC content . Washing, deglycerolizing after thawing a frozen unit, centrifugation . These methods do not meet requirement of < 5.0 x 106 WBCs per unit of RBCs/apheresis platelets. Types of leukofiltration/leukoreduction o “Pre-storage” . Done within 24 hours of collection . May use inline filters at time of collection (apheresis) or post collection o “Pre-transfusion” leukoreduction/bedside leukoreduction . Done prior to transfusion . “Bedside” leukoreduction uses gravity-based filters at time of transfusion. Least desirable given variability in practice and absence of proficiency . Alternatively performed by transfusion service prior to issuing Benefits of leukoreduction o Prevention of alloimmunization to donor HLA antigens . Anti-HLA can mediate graft rejection and immune mediated destruction of platelets o Leukoreduced products are indicated for transplant recipients or patients who are likely platelet transfusion dependent o Prevention of febrile non-hemolytic transfusion reactions (FNHTR) . -

Patient Information Leaflet – Plasma Exchange Procedure
Therapeutic Apheresis Services Patient Information Leaflet – Plasma Exchange Procedure Introduction Antibodies, which normally help to protect you from infection, can begin to attack your own This leaflet has been written to give patients healthy cells, or an over production of proteins can information about plasma exchange (sometimes cause your blood to become thicker and slow down called plasmapheresis). If you would like any more the blood flow throughout your body. A plasma information or have any questions, please ask the exchange can help improve your symptoms, doctors and nurses involved in your treatment at although this may not happen immediately. the NHS Blood and Transplant (NHSBT) Therapeutic Apheresis Services Unit. Although plasma exchange may help with symptoms, it will not normally cure your condition When you have considered the information given as it does not switch off the production of the in this leaflet, and after we have discussed the harmful antibodies or proteins. It is likely that procedure and its possible risks with you, we will this procedure will form only one part of your ask you to sign a consent form to indicate that you treatment. are happy for the procedure to go ahead. Before any further procedures we will again check that you are happy to proceed. How do we perform Plasma Exchange? What is a plasma exchange? Plasma exchange is performed using a machine Blood is made up of red cells, white cells and called a Blood Cell Separator which can separate platelets which are carried around in fluid called blood into its various parts. The machine separates plasma. -

Your Blood Cells
Page 1 of 2 Original Date The Johns Hopkins Hospital Patient Information 12/00 Oncology ReviseD/ RevieweD 6/14 Your Blood Cells Where are Blood cells are made in the bone marrow. The bone marrow blood cells is a liquid that looks like blood. It is found in several places of made? the body, such as your hips, chest bone and the middle part of your arm and leg bones. What types of • The three main types of blood cells are the red blood cells, blood cells do the white blood cells and the platelets. I have? • Red blood cells carry oxygen to all parts of the body. The normal hematocrit (or percentage of red blood cells in the blood) is 41-53%. Anemia means low red blood cells. • White blood cells fight infection. The normal white blood cell count is 4.5-11 (K/cu mm). The most important white blood cell to fight infection is the neutrophil. Forty to seventy percent (40-70%) of your white blood cells should be neutrophils. Neutropenia means your neutrophils are low, or less than 40%. • Platelets help your blood to clot and stop bleeding. The normal platelet count is 150-350 (K/cu mm). Thrombocytopenia means low platelets. How do you Your blood cells are measured by a test called the Complete measure my Blood Count (CBC) or Heme 8/Diff. You may want to keep track blood cells? of your blood counts on the back of this sheet. What When your blood counts are low, you may become anemic, get happens infections and bleed or bruise easier. -

Important Information for Female Platelet Donors
Important Information for Female Platelet Donors We are grateful for the support you provide our community blood program and especially appreciate your willingness to help save lives as a volunteer platelet donor. We also take our responsibility to provide a safe and adequate blood supply very seriously and need to share the following information regarding a change to our donor eligibility criteria for female platelet donors. We recently began performing a Human Leukocyte Antigen (HLA) antibody test on each of our current female platelet donors who have ever been pregnant. In addition, we modified our Medical History Questionnaire to ask donors whether they have been pregnant since their last donation. Platelet donors who respond yes to that question will be screened for HLA antibodies. Platelet donors will also be retested after every subsequent pregnancy. These adjustments are being made as part of our effort to reduce occurrences of Transfusion-Related Acute Lung Injury (TRALI). TRALI is a rare but serious complication of blood transfusions most commonly thought to be caused by a reaction to HLA antibodies present in the donor’s plasma. When transfused, these antibodies can sometimes cause plasma to leak into the patient’s lungs, creating fluid accumulation — a condition referred to as acute pulmonary edema. Female donors who have been pregnant are more likely than others to have these HLA antibodies in their plasma. Once the antibodies develop, they are present forever. The antibodies could be harmful if transfused into certain patients. The antibodies are present in plasma — and platelet donations contain a high volume of plasma, so our current efforts are directed at screening blood samples from female platelet donors to test for the HLA antibody. -

FACTS ABOUT DONATING Blood WHY SHOULD I DONATE BLOOD? HOW MUCH BLOOD DO I HAVE? Blood Donors Save Lives
FACTS ABOUT DONATING BLOOD WHY SHOULD I DONATE BLOOD? HOW MUCH BLOOD DO I HAVE? Blood donors save lives. Volunteer blood donors provide An adult has about 10–11 pints. 100 percent of our community’s blood supply. Almost all of us will need blood products. HOW MUCH BLOOD WILL I donate? Whole blood donors give 500 milliliters, about one pint. WHO CAN DONATE BLOOD? Generally, you are eligible to donate if you are 16 years of WHAT HAPPENS TO BLOOD AFTER I donate? age or older, weigh at least 110 pounds and are in good Your blood is tested, separated into components, then health. Donors under 18 must have written permission distributed to local hospitals and trauma centers for from a parent or guardian prior to donation. patient transfusions. DOES IT HURT TO GIVE BLOOD? WHAT ARE THE MAIN BLOOD COMPONENTS? The sensation you feel is similar to a slight pinch on the Frequently transfused components include red blood arm. The process of drawing blood should take less than cells, which replace blood loss in patients during surgery ten minutes. or trauma; platelets, which are often used to control or prevent bleeding in surgery and trauma patients; and HOW DO I PREPARE FOR A BLOOD DONATION? plasma, which helps stop bleeding and can be used to We recommend that donors be well rested, eat a healthy treat severe burns. Both red blood cells and platelets meal, drink plenty of fluids and avoid caffeine and are often used to support patients undergoing cancer alcohol prior to donating. treatments. WHAT CAN I EXPECT WHEN I DONATE? WHEN CAN I DONATE AGAIN? At every donation, you will fill out a short health history You can donate whole blood every eight weeks, platelets questionaire. -

Transfusion Triggers for Platelets and Other Blood Products Sunil Karanth Indian Journal of Critical Care Medicine (2019): 10.5005/Jp-Journals-10071-23250
INVITED ARTICLE Transfusion Triggers for Platelets and Other Blood Products Sunil Karanth Indian Journal of Critical Care Medicine (2019): 10.5005/jp-journals-10071-23250 INTRODUCTION Department of Intensive Care Unit, Manipal Hospital, Bengaluru, Transfusion of whole blood is not associated with any significant Karnataka, India benefit, rather can be harmful. Significant advances have been Corresponding Author: Sunil Karanth, Department of Intensive made in transfusion medicine, facilitating the use of blood product Care Unit, Manipal Hospital, Bengaluru, Karnataka, India, e-mail: or component therapy than use of whole blood (Fig. 1). In 2009, a [email protected] report on Serious Hazards of Transfusion in the UK, estimated that How to cite this article: Karanth S. Transfusion Triggers for Platelets a total of 3 million units of blood components were released. The and Other Blood Products. Indian J Crit Care Med 2019;23(Suppl requirement of the same in South-East Asia is much higher to the 3):S189–S190. tune of 15 million units annually. The current recommendation Source of support: Nil is to use blood only in life-threatening situations, rather than to Conflict of interest: None normalize abnormal numbers. PLATELETS Platelets are derived from the buffy coat of whole blood donations. A pooled platelet concentrate includes pooled buffy-coat derived platelets from four whole-blood donations suspended in platelet additive solution and plasma of one of the four donors. It contains 240000 platelets pooled from 4–6 donors. A Single donor platelet is derived from a single-donor by a process of apheresis. In view of the lesser number of donors and the theoretical advantage of involving a single-donor platelet (SDP) may be preferred over the use of platelets from multiple donors. -

Blood-Platelet-Orders-Outpatient-V3
Blood / Platelet Orders Outpt v3 USE THIS ORDER SET FOR OUTPATIENT NON URGENT BLOOD OR PLATELET TRANSFUSION 24 Hours advanced notice required for infusion center NO MORE THAN 2 UNITS OF RED BLOOD CELLS CAN BE INFUSED IN THE INFUSION CENTER PER DAY BMH Outpatient Infusion Center: Fax completed order set to 843-522-7313/Phone 843-522-7680 Hospital Outpatient: Fax Completed order set to Registration at 843-522-5741 and notify Nursing Supervisor at 843-522-7653 Service Designation THIS IS NOT AN ADMISSION SET Patient Name ___________________________________ Patient DOB ____________ Service Designation Blood Platelet Outpatient Attending ___________________________________ Date Requested ___________________________________ Status Outpatient Check Appropriate Diagnosis Below: [ Anemia of chronic renal disease Anemia related to chemotherapy Aplastic anemia Anemia related to cancer Anemia related to blood loss Sickle cell disease Anemia unspecified Thombocytopenia (platelets) Other _________________________ ] Allergies Update Allergies in the Summary Panel in MEDITECH Special Requirement: (REQUIRES SPECIAL ORDER ONE DAY IN ADVANCE) Special Requirement RBC or Platelets REQUIRES SPECIAL ORDER ONE DAY IN ADVANCE [ Irradiated CMV Negative HgBS Negative ] Medications diphenhydrAMINE HCl (Benadryl) 25 mg orally single dose premedicate prior to transfusion diphenhydrAMINE HCl (Benadryl) 50 mg orally single dose premedicate prior to transfusion diphenhydrAMINE HCl (Benadryl) 25 mg intravenously single dose premedicate prior to transfusion diphenhydrAMINE -

FDA Regulation of Blood and Blood Components in the United States
FDA Regulation of Blood and Blood Components in the United States SLIDE 1 This presentation will review the FDA Regulation of Blood and Blood Components in the U.S. SLIDE 2 The FDA Center for Biologics Evaluation and Research, or CBER, Office of Blood Research and Review, called OBRR, reviews several different types of regulatory applications with respect to blood and blood components. This includes biologics license applications, called BLAs, which represent the regulatory pathway for blood components. The BLA regulatory process also applies to biological drugs such as fractionated plasma products. OBRR also regulates in-vitro diagnostic devices used for screening of collected blood, and does so also using the BLA regulatory pathway. Review of these devices as biologic licenses allows CBER to apply a higher level of manufacturing oversight, including lot release testing and pre-licensure inspection. This regulatory pathway applies to infectious disease tests for blood screening, as well as blood grouping and phenotyping reagents. SLIDE 3 The regulatory pathway for New Drug Applications, or NDAs, is most commonly used within the Center for Drug Evaluation and Research, or CDER. Within the Office of Blood, several NDA applications are reviewed each year, mostly involving solutions used for the collection of blood, such as anticoagulants and red cell nutritive solutions. Interestingly, a blood bag that does not have a solution inside is regulated as a device. If the bag has a solution, then it is a drug-device combination product, but is regulated as a drug. SLIDE 4 OBRR reviews both Class Two and Class Three devices. Class Three devices in OBRR include diagnostic tests for HIV regulated as pre-market approvals, called PMAs. -

Human ICAM-4 Antibody
Human ICAM-4 Antibody Monoclonal Mouse IgG2B Clone # 729632 Catalog Number: MAB8397 DESCRIPTION Species Reactivity Human Specificity Detects human ICAM4 in direct ELISAs. Source Monoclonal Mouse IgG2B Clone # 729632 Purification Protein A or G purified from hybridoma culture supernatant Immunogen Chinese hamster ovary cell line CHOderived recombinant human ICAM4 Ala23Ala240 Accession # Q14773 Formulation Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details. *Small pack size (SP) is supplied either lyophilized or as a 0.2 μm filtered solution in PBS. APPLICATIONS Please Note: Optimal dilutions should be determined by each laboratory for each application. General Protocols are available in the Technical Information section on our website. Recommended Sample Concentration Flow Cytometry 0.25 µg/106 cells See Below CyTOFready Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation. DATA Flow Cytometry Detection of ICAM4 in Human Red Blood Cells by Flow Cytometry. Human red blood cells were stained with Mouse Anti Human ICAM4 Monoclonal Antibody (Catalog # MAB8397, filled histogram) or isotype control antibody (Catalog # MAB0041, open histogram), followed by Allophycocyaninconjugated AntiMouse IgG Secondary Antibody (Catalog # F0101B). PREPARATION AND STORAGE Reconstitution Reconstitute at 0.5 mg/mL in sterile PBS. Shipping The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. *Small pack size (SP) is shipped with polar packs. Upon receipt, store it immediately at 20 to 70 °C Stability & Storage Use a manual defrost freezer and avoid repeated freezethaw cycles.