Cord Blood Stem Cell Transplantation
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Umbilical Cord Blood Banking. a Guide for the Parents
UMBILICAL CORD BLOOD BANKING A guide for parents 2nd Edition 2016 This guide has been elaborated by the Council of Europe European Committee on Organ Transplantation (CD-P-TO). For more information, please visit https://go.edqm.eu/transplantation. All rights conferred by virtue of the International Copyright Convention are specifically reserved to the Council of Europe and any reproduction or translation requires the written consent of the Publisher. Director of the Publication: Dr S. Keitel Page layout and cover: EDQM Photo: © millaf – Fotolia.com Illustrations: © aeroking – Fotolia.com European Directorate for the Quality of Medicines & HealthCare (EDQM) Council of Europe 7, allée Kastner CS 30026 F-67081 STRASBOURG FRANCE Internet: www.edqm.eu © Council of Europe, 2015, 2016 First published 2015 Second Edition 2016 2 INTRODUCTION The collection and storage of umbilical cord blood when a baby is born is becoming increasingly common. The reason is that the cells contained in the The cells contained in the umbilical cord blood have therapeu- umbilical cord blood have tic value for the treatment of malignant therapeutic value for and non-malignant blood disorders and the treatment of blood immune diseases. Cord blood has been disorders and immune used in transplant medicine since the diseases. first allogeneic cord blood transplant was performed in 1988 and, over the last 25 years, this activity has grown rapidly. Allogeneic cord blood transplantation in children has similar survival rates compared to transplantation of haema- topoietic stem cells from other sources (e.g. bone marrow), and results for adults continue to improve. In recent years, the number of cord blood banks offering families to store the cord blood of their babies for possible future private uses against up front and yearly fees has grown. -

Comparison Between Human Cord Blood Serum and Platelet-Rich Plasma Supplementation for Human Wharton's Jelly Stem Cells and Dermal Fibroblasts Culture
Available online at www.ijmrhs.com International Journal of Medical Research & ISSN No: 2319-5886 Health Sciences, 2016, 5, 8:191-196 Comparison between human cord blood serum and platelet-rich plasma supplementation for Human Wharton's Jelly Stem Cells and dermal fibroblasts culture Hashemi S. S1* and Rafati A.R 2 1 Burn and Wound Healing Research Centre, Shiraz University of Medical Sciences, Shiraz, Iran 2Division of Pharmacology & Pharmaceutical Chemistry, Sarvestan Branch, Islamic Azad University, Sarvestan, Iran *Corresponding E-mail: [email protected] _____________________________________________________________________________________________ ABSTRACT We carried out a side-by-side comparison of the effects of Human cord blood serum (HcbS) versus embryonic PRP on Human Wharton's Jelly Stem Cells(hWMSC)and dermal fibroblasts proliferation. Human umbilical cord blood was collected to prepare activated serum (HCS) and platelet-rich plasma (CPRP).Wharton's Jelly Stem Cells and dermal fibroblasts were cultured in complete medium with10% CPRP, 10%HCSor 10% fetal bovine serumand control (serum-free media).The efficiency of the protocols was evaluated in terms of the number of adherent cells and their expansion and Cell proliferation. We showed that proliferation of fibroblasts and mesenchymal stem cells in the presence of cord blood serum and platelet-rich plasma significantly more than the control group (p ≤0/05). As an alternative to FBS, cord blood serum has been proved as an effective component in cell tissue culture applications and embraced a vast future in clinical applications of regenerative medicine. However, there is still a need to explore the potential of HCS and its safe applications in humanized cell therapy or tissue engineering. -
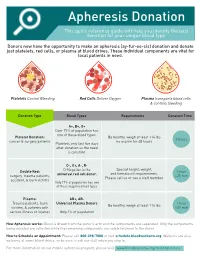
Apheresis Donation This Quick Reference Guide Will Help You Identify the Best Donation for Your Unique Blood Type
Apheresis Donation This quick reference guide will help you identify the best donation for your unique blood type. Donors now have the opportunity to make an apheresis (ay-fur-ee-sis) donation and donate just platelets, red cells, or plasma at blood drives. These individual components are vital for local patients in need. Platelets Control Bleeding Red Cells Deliver Oxygen Plasma transports blood cells & controls bleeding Donation Type Blood Types Requirements Donation Time A+, B+, O+ Over 75% of population has one of these blood types. Platelet Donation: Be healthy, weigh at least 114 lbs 2 hours cancer & surgery patients no aspirin for 48 hours Platelets only last five days after donation so the need is constant. O-, O+, A-, B- Special height, weight, Double Red: O-Negative is the 1 hour and hematocrit requirements. surgery, trauma patients, universal red cell donor. +25 min Please call us or see a staff member accident, & burn victims Only 17% of population has one of these negative blood types Plasma: AB+, AB- Trauma patients, burn Universal Plasma Donors 1 hour Be healthy, weigh at least 114 lbs victims, & patients with +30 min serious illness or injuries Only 4% of population How Apheresis works: Blood is drawn from the donor’s arm and the components are separated. Only the components being donated are collected while the remaining components are safely returned to the donor How to Schedule an Appointment: Please call 800-398-7888 or visit schedule.bloodworksnw.org. Walk-ins are also welcome at some blood drives, so be sure to ask our staff when you stop in. -

Blood Product Modifications: Leukofiltration, Irradiation and Washing
Blood Product Modifications: Leukofiltration, Irradiation and Washing 1. Leukocyte Reduction Definitions and Standards: o Process also known as leukoreduction, or leukofiltration o Applicable AABB Standards, 25th ed. Leukocyte-reduced RBCs At least 85% of original RBCs < 5 x 106 WBCs in 95% of units tested . Leukocyte-reduced Platelet Concentrates: At least 5.5 x 1010 platelets in 75% of units tested < 8.3 x 105 WBCs in 95% of units tested pH≥6.2 in at least 90% of units tested . Leukocyte-reduced Apheresis Platelets: At least 3.0 x 1011 platelets in 90% of units tested < 5.0 x 106 WBCs 95% of units tested pH≥6.2 in at least 90% of units tested Methods o Filter: “Fourth-generation” filters remove 99.99% WBCs o Apheresis methods: most apheresis machines have built-in leukoreduction mechanisms o Less efficient methods of reducing WBC content . Washing, deglycerolizing after thawing a frozen unit, centrifugation . These methods do not meet requirement of < 5.0 x 106 WBCs per unit of RBCs/apheresis platelets. Types of leukofiltration/leukoreduction o “Pre-storage” . Done within 24 hours of collection . May use inline filters at time of collection (apheresis) or post collection o “Pre-transfusion” leukoreduction/bedside leukoreduction . Done prior to transfusion . “Bedside” leukoreduction uses gravity-based filters at time of transfusion. Least desirable given variability in practice and absence of proficiency . Alternatively performed by transfusion service prior to issuing Benefits of leukoreduction o Prevention of alloimmunization to donor HLA antigens . Anti-HLA can mediate graft rejection and immune mediated destruction of platelets o Leukoreduced products are indicated for transplant recipients or patients who are likely platelet transfusion dependent o Prevention of febrile non-hemolytic transfusion reactions (FNHTR) . -

FACTS ABOUT DONATING Blood WHY SHOULD I DONATE BLOOD? HOW MUCH BLOOD DO I HAVE? Blood Donors Save Lives
FACTS ABOUT DONATING BLOOD WHY SHOULD I DONATE BLOOD? HOW MUCH BLOOD DO I HAVE? Blood donors save lives. Volunteer blood donors provide An adult has about 10–11 pints. 100 percent of our community’s blood supply. Almost all of us will need blood products. HOW MUCH BLOOD WILL I donate? Whole blood donors give 500 milliliters, about one pint. WHO CAN DONATE BLOOD? Generally, you are eligible to donate if you are 16 years of WHAT HAPPENS TO BLOOD AFTER I donate? age or older, weigh at least 110 pounds and are in good Your blood is tested, separated into components, then health. Donors under 18 must have written permission distributed to local hospitals and trauma centers for from a parent or guardian prior to donation. patient transfusions. DOES IT HURT TO GIVE BLOOD? WHAT ARE THE MAIN BLOOD COMPONENTS? The sensation you feel is similar to a slight pinch on the Frequently transfused components include red blood arm. The process of drawing blood should take less than cells, which replace blood loss in patients during surgery ten minutes. or trauma; platelets, which are often used to control or prevent bleeding in surgery and trauma patients; and HOW DO I PREPARE FOR A BLOOD DONATION? plasma, which helps stop bleeding and can be used to We recommend that donors be well rested, eat a healthy treat severe burns. Both red blood cells and platelets meal, drink plenty of fluids and avoid caffeine and are often used to support patients undergoing cancer alcohol prior to donating. treatments. WHAT CAN I EXPECT WHEN I DONATE? WHEN CAN I DONATE AGAIN? At every donation, you will fill out a short health history You can donate whole blood every eight weeks, platelets questionaire. -
The Amazing Stem Cell What Are They? Where Do They Come From? How Are They Changing Medicine? Stem Cells Are “Master Cells”
The Amazing Stem Cell What are they? Where do they come from? How are they changing medicine? Stem cells are “master cells” Stem cells can be “guided” to become many other cell types. Stem Cell Bone cell Self-renewed stem cell Brain cell Heart muscle Blood cell cell There are several types of stem cells, each from a unique source Embryonic stem cells* • Removed from embryos created for in vitro fertilization after donation consent is given. (Not sourced from aborted fetuses.) • Embryos are 3-5 days old (blastocyst) and have about 150 cells. • Can become any type of cell in the body, also called pluripotent cells. • Can regenerate or repair diseased tissue and organs. • Current use limited to eye-related disorders. * Not used by Mayo Clinic. Adult stem cells • Found in most adult organs and tissues, including bone marrow. • Often taken from bone marrow in the hip. • Blood stem cells can be collected through apheresis (separated from blood). • Can regenerate and repair diseased or damaged tissues (regenerative medicine). • Can be used as specialized “drugs” to potentially treat degenerative conditions. • Currently tested in people with neurological and heart disease. Umbilical cord blood stem cells • Found in blood in placenta and umbilical cord after childbirth. • Have the ability to change into specialized cells (like blood cells), also called progenitor cells. • Parents choose to donate umbilical cord blood for use in research, or have it stored for private or public banks. • Can be used in place of bone marrow stem cell transplants in some clinical applications. Bioengineered stem cells • Regular adult cells (e.g., blood, skin) reprogrammed to act like embryonic stem cells (induced pluripotent stem cells). -

Cord Blood Stem Cells Umbilical Cord Blood Transplant for Adult Patients
Bone Marrow Transplantation (2004) 33, 33–38 & 2004 Nature Publishing Group All rights reserved 0268-3369/04 $25.00 www.nature.com/bmt Cord blood stem cells Umbilical cord blood transplant for adult patients with severe aplastic anemia using anti-lymphocyte globulin and cyclophosphamide as conditioning therapy P Mao, S Wang, S Wang, Z Zhu, Q Liv, Y Xuv, W Mo and Y Ying Department of Haematology, First Municipal People’s Hospital, Guangzhou, China Summary: therapy with immunosuppressive agents.1 For patients without a sibling donor having no response to one or Allo-CBSCT (cord blood stem cell transplant) has more courses of immunosuppressive therapy a fully been applied in sixadult patients with severe aplastic matched unrelated donor BMT should be considered anemia (SAA). Anti-lymphocyte globulin (ALG) asalternative salvagetherapy. Finding related or unrelated 40 mg kgÀ1 dÀ1 Â 3 days combined with cyclophosphamide individualswho are HLA-identical to some (CTX) 20 mg kgÀ1 dÀ1 Â 3 days constituted a lower patients, however, is difficult and time consuming because intensive conditioning regimen. The prophylaxis of of the extreme polymorphism of most HLA GVHD consisted of standard CsA and MTX. Patients loci. Umbilical cord blood isanother alternative sources are all male having a mean age of 26.5 years (range 22– of stem cells that improves donor availability for trans- 38), and a median weight of 55.6 kg (range 52–60 kg). plantation because frozen and stored UCB can be Cord blood searches were all conducted at Guangzhou made available on demand. Infectiousagents,particularly Cord Blood Bank. Three of sixpatients in our study cytomegalovirus(CMV), are rarely seenin the new received one unit of cord blood in a procedure, whereas for born than in adults. -

Summary of Blood Donor Deferral Following COVID-19 Vaccine And
Updated 04 14 2021 Updated Information from FDA on Donation of CCP, Blood Components and HCT/Ps, Including Information on COVID-19 Vaccines, Treatment with CCP or Monoclonals 1) HCT/P DONOR ELIGIBILITY: For the agency’s current thinking on HCT/P donation during the pandemic, including donor eligibility following vaccination to prevent COVID-19, refer to FDA’s January 4th Safety and Availability communication, Updated Information for Human Cell, Tissue, or Cellular or Tissue-based Product (HCT/P) Establishments Regarding the COVID-19 Pandemic. 2) CCP and ROUTINE BLOOD DONOR ELIGIBILITY: 2-1. Blood donor eligibility following COVID-19 vaccine Blood donor deferral following COVID-19 vaccines is not required. Consider the following information on FDA’s web page Updated Information for Blood Establishments Regarding the COVID-19 Pandemic and Blood Donation last updated 01/19/21” – see the full document on page 2 below. The blood establishment’s responsible physician must evaluate prospective donors and determine eligibility (21 CFR 630.5). The donor must be in good health and meet all donor eligibility criteria on the day of donation (21 CFR 630.10). The responsible physician may wish to consider the following: o individuals who received a nonreplicating, inactivated, or mRNA-based COVID-19 vaccine can donate blood without a waiting period, o individuals who received a live-attenuated viral COVID-19 vaccine, refrain from donating blood for a short waiting period (e.g., 14 days) after receipt of the vaccine, o individuals who are uncertain about which COVID-19 vaccine was administered, refrain from donating for a short waiting period (e.g., 14 days) if it is possible that the individual received a live-attenuated viral vaccine. -

Comparative Analysis of Human Mesenchymal Stem Cells from Bone Marrow, Adipose Tissue, and Umbilical Cord Blood As Sources of Cell Therapy
Int. J. Mol. Sci. 2013, 14, 17986-18001; doi:10.3390/ijms140917986 OPEN ACCESS International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms Article Comparative Analysis of Human Mesenchymal Stem Cells from Bone Marrow, Adipose Tissue, and Umbilical Cord Blood as Sources of Cell Therapy Hye Jin Jin 1,2,†, Yun Kyung Bae 1,†, Miyeon Kim 1, Soon-Jae Kwon 1, Hong Bae Jeon 1, Soo Jin Choi 1, Seong Who Kim 2, Yoon Sun Yang 1, Wonil Oh 1 and Jong Wook Chang 1,* 1 Biomedical Research Institute, MEDIPOST Co., Ltd., Seoul 137-874, Korea; E-Mails: [email protected] (H.J.J.); [email protected] (Y.K.B.); [email protected] (M.K.); [email protected] (S.-J.K.); [email protected] (H.B.J.); [email protected] (S.J.C.); [email protected] (Y.S.Y.); [email protected] (W.O.) 2 Molecular Biology, University of Ulsan College of Medicine, Seoul 138-736, Korea; E-Mail: [email protected] † These authors contributed equally to this work. * Author to whom correspondence should be addressed; E-Mail: [email protected]; Tel.: +82-2-3465-6771; Fax: +82-2-475-1991. Received: 27 May 2013; in revised form: 18 July 2013 / Accepted: 22 August 2013 / Published: 3 September 2013 Abstract: Various source-derived mesenchymal stem cells (MSCs) have been considered for cell therapeutics in incurable diseases. To characterize MSCs from different sources, we compared human bone marrow (BM), adipose tissue (AT), and umbilical cord blood-derived MSCs (UCB-MSCs) for surface antigen expression, differentiation ability, proliferation capacity, clonality, tolerance for aging, and paracrine activity. -

Therapeutic Apheresis, J Clin Apheresis 2007, 22, 104-105
Apheresis: Basic Principles, Practical Considerations and Clinical Applications Joseph Schwartz, MD Anand Padmanabhan, MD PhD Director, Transfusion Medicine Assoc Med Director/Asst Prof Columbia Univ. Medical Center BloodCenter of Wisconsin New York Presbyterian Hospital Medical College of Wisconsin Review Session, ASFA Annual meeting, Scottsdale, Arizona, June 2011 Objectives (Part 1) • Mechanism of Action • Definitions • Technology (ies) • Use • Practical Considerations • Math • Clinical applications – HPC Collection Objectives (Part 2) • Clinical applications: System/ Disease Specific Indications • ASFA Fact Sheet Apheresis •Derives from Greek, “to carry away” •A technique in which whole blood is taken and separated extracorporealy, separating the portion desired from the remaining blood. •This allows the desired portion (e.g., plasma) to be removed and the reminder returned. Apheresis- Mechanism of Action •Large-bore intravenous catheter connected to a spinning centrifuge bowl •Whole blood is drawn from donor/patient into the centrifuge bowl •The more dense elements, namely the RBC, settle to the bottom with less dense elements such as WBC and platelets overlying the RBC layer and finally, plasma at the very top. Apheresis: Principles of Separation Platelets (1040) Lymphocytes Torloni MD (1050-1061) Monocytes (1065 - 1069) Granulocyte (1087 - 1092) RBC Torloni MD Torloni MD Separate blood components is based on density with removal of the desired component Graphics owned by and courtesy of Gambro BCT Principals of Apheresis WBC Plasma Torlo RBC ni MD Torloni MD RBC WBC Plasma G Cobe Spectra Apheresis- Mechanism of Action Definitions • Plasmapheresis: plasma is separated, removed (i.e. less than 15% of total plasma volume) without the use of replacement solution • Plasma exchange (TPE): plasma is separated, removed and replaced with a replacement solution such as colloid (e.g. -

When Is Blood Donation Safe? T Lise Sofie H
Transfusion and Apheresis Science 58 (2019) 113–116 Contents lists available at ScienceDirect Transfusion and Apheresis Science journal homepage: www.elsevier.com/locate/transci Review ☆ Donor health assessment – When is blood donation safe? T Lise Sofie H. Nissen-Meyera, Jerard Seghatchianb a Oslo Blood Centre, Department of Immunology and Transfusion Medicine, Oslo University Hospital, Oslo, Norway b International Consultancy in Blood Components Quality/Safety Improvement and DDR Strategies, London, United Kingdom ARTICLE INFO ABSTRACT Keywords: Blood donation is a highly regulated practice in the world, ensuring the safety and efficacy of collected blood and its Blood donation components whether used as irreplaceable parts of modern transfusion medicine, as a therapeutic modality or addi- Donor health assessment tional support to other clinical therapies. In Norway blood donation is regulated by governmental regulations Health and disease (“Blodforskriften”) and further instructed by national guidelines, “Veileder for transfusjonstjenesten” [1], providing an Transfusion aid for assessment of donor health. This concise review touches upon: definitions of donor health and disease; some Blood components important pitfalls; and the handling of some common and less common pathophysiological conditions; with an example from the Blood center of Oslo University Hospital, Norway’s largest blood center. I also comment on some medications used by a number of blood donors, although wounds, ulcers and surgery are not included. Considering the panorama of conditions blood donors can suffer from, blood donation can never be completely safe for everybody, as zero riskdoes not exist, but it is our task through donor evaluation to identify and reduce risk as much as possible. 1. -

Recommendations for Collecting Red Blood Cells by Automated Apheresis Methods
Guidance for Industry Recommendations for Collecting Red Blood Cells by Automated Apheresis Methods Additional copies of this guidance document are available from: Office of Communication, Training and Manufacturers Assistance (HFM-40) 1401 Rockville Pike, Rockville, MD 20852-1448 (Tel) 1-800-835-4709 or 301-827-1800 (Internet) http://www.fda.gov/cber/guidelines.htm U.S. Department of Health and Human Services Food and Drug Administration Center for Biologics Evaluation and Research (CBER) January 2001 Technical Correction February 2001 TABLE OF CONTENTS Note: Page numbering may vary for documents distributed electronically. I. INTRODUCTION ............................................................................................................. 1 II. BACKGROUND................................................................................................................ 1 III. CHANGES FROM THE DRAFT GUIDANCE .............................................................. 2 IV. RECOMMENDED DONOR SELECTION CRITERIA FOR THE AUTOMATED RED BLOOD CELL COLLECTION PROTOCOLS ..................................................... 3 V. RECOMMENDED RED BLOOD CELL PRODUCT QUALITY CONTROL............ 5 VI. REGISTRATION AND LICENSING PROCEDURES FOR THE MANUFACTURE OF RED BLOOD CELLS COLLECTED BY AUTOMATED METHODS.................. 7 VII. ADDITIONAL REQUIREMENTS.................................................................................. 9 i GUIDANCE FOR INDUSTRY Recommendations for Collecting Red Blood Cells by Automated Apheresis Methods This