Guidelines on the Use of Therapeutic Apheresis In
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Pathologic Rupture of the Spleen in a Patient with Acute Myelogenous Leukemia and Leukostasis
rev bras hematol hemoter. 2014;36(4):290–292 Revista Brasileira de Hematologia e Hemoterapia Brazilian Journal of Hematology and Hemotherapy www.rbhh.org Case report Pathologic rupture of the spleen in a patient with acute myelogenous leukemia and leukostasis Gil Cunha De Santis ∗, Luciana Correa Oliveira, Aline Fernanda Ramos, Nataly Dantas Fortes da Silva, Roberto Passetto Falcão Universidade de São Paulo (USP), Ribeirão Preto, SP, Brazil article info abstract Article history: Rupture of the spleen can be classified as spontaneous, traumatic, or pathologic. Patho- Received 14 October 2013 logic rupture has been reported in infectious diseases such as infectious mononucleosis, Accepted 14 January 2014 and hematologic malignancies such as acute and chronic leukemias. Splenomegaly is con- Available online 28 May 2014 sidered the most relevant factor that predisposes to splenic rupture. A 66-year-old man with acute myeloid leukemia evolved from an unclassified myeloproliferative neoplasm, Keywords: complaining of fatigue and mild upper left abdominal pain. He was pale and presented Splenomegaly fever and tachypnea. Laboratory analyses showed hemoglobin 8.3 g/dL, white blood cell 9 9 Leukemia count 278 × 10 /L, platelet count 367 × 10 /L, activated partial thromboplastin time (aPTT) Myeloid ratio 2.10, and international normalized ratio (INR) 1.60. A blood smear showed 62% of Acute myeloblasts. The immunophenotype of the blasts was positive for CD117, HLA-DR, CD13, Leukostasis CD56, CD64, CD11c and CD14. Lactate dehydrogenase was 2384 U/L and creatinine 2.4 mg/dL (normal range: 0.7–1.6 mg/dL). Two sessions of leukapheresis were performed. At the end of the second session, the patient presented hemodynamic instability that culminated in cir- culatory shock and death. -

Requirements for Blood and Blood Components Intended for Transfusion Or for Further Manufacturing Use
Requirements for Blood and Blood Components Intended for Transfusion or for Further Manufacturing Use Office of Blood Research and Review CBER, FDA Ask the FDA Session – AABB Annual Meeting October 26, 2015 1 Outline • Overview – Historical Background – Intent of the Rule – Organization of the Rule • Selected Provisions – Relevant Transfusion-Transmitted Infection – Control of Bacterial Contamination of Platelets – Medical Supervision – Donor Acknowledgement – Alternative Procedures – Donor Eligibility • Implementation 2 GAO Oversight and FDA Concerns • Publish in the form of regulations the guidelines that FDA deems essential to ensure the safety of the blood supply • Concern about the delay in requiring testing for emerging infectious agents, e.g. HTLV • Concern about blood safety and the regulations being out-of-date • Concerns about donor safety 3 Intent of the Final Rule • To better assure the safety of the blood supply and to help protect donor health • To make donor eligibility and testing requirements more consistent with current practices and to provide flexibility with regard to emerging infectious diseases • To accommodate technological advances • To establish requirements for donor education, donor history, and donor testing 4 Organization of the Final Rule - 1 • A. General • B. Definitions (§§ 606.3, 610.39, 630.3, 640.125) • C. Standard Operating Procedures (§ 606.100) • D. Control of Bacterial Contamination of Platelets (§ 606.145) • E. Records (§ 606.160) • F. Test Requirements (§§ 610.40, 640.5, 640.71(a)) • G. Donor Deferral (§ 610.41) 5 Organization of the Final Rule - 2 • H. Purpose and Scope (§ 630.1) • I. Medical Supervision (§ 630.5) • J. General Donor Eligibility Requirements(§ 630.10) • K. Donor Eligibility Requirements Specific to Whole Blood, Red Blood Cells and Plasma Collected by Apheresis (§ 630.15) • L. -

27. Clinical Indications for Cryoprecipitate And
27. CLINICAL INDICATIONS FOR CRYOPRECIPITATE AND FIBRINOGEN CONCENTRATE Cryoprecipitate is indicated in the treatment of fibrinogen deficiency or dysfibrinogenaemia.1 Fibrinogen concentrate is licenced for the treatment of acute bleeding episodes in patients with congenital fibrinogen deficiency, including afibrinogenaemia and hypofibrinogenaemia,2 and is currently funded under the National Blood Agreement. Key messages y Fibrinogen is an essential component of the coagulation system, due to its role in initial platelet aggregation and formation of a stable fibrin clot.3 y The decision to transfuse cryoprecipitate or fibrinogen concentrate to an individual patient should take into account the relative risks and benefits.3 y The routine use of cryoprecipitate or fibrinogen concentrate is not advised in medical or critically ill patients.2,4 y Cryoprecipitate or fibrinogen concentrate may be indicated in critical bleeding if fibrinogen levels are not maintained using FFP. In the setting of major obstetric haemorrhage, early administration of cryoprecipitate or fibrinogen concentrate may be necessary.3 Clinical implications y The routine use of cryoprecipitate or fibrinogen concentrate in medical or critically ill patients with coagulopathy is not advised. The underlying causes of coagulopathy should be identified; where transfusion is considered necessary, the risks and benefits should be considered for each patient. Specialist opinion is advised for the management of disseminated intravascular coagulopathy (MED-PP18, CC-PP7).2,4 y Cryoprecipitate or fibrinogen concentrate may be indicated in critical bleeding if fibrinogen levels are not maintained using FFP. In patients with critical bleeding requiring massive transfusion, suggested doses of blood components is 3-4g (CBMT-PP10)3 in adults or as per the local Massive Transfusion Protocol. -

Unintentional Platelet Removal by Plasmapheresis
Journal of Clinical Apheresis 16:55–60 (2001) Unintentional Platelet Removal by Plasmapheresis Jedidiah J. Perdue,1 Linda K. Chandler,2 Sara K. Vesely,1 Deanna S. Duvall,2 Ronald O. Gilcher,2 James W. Smith,2 and James N. George1* 1Hematology-Oncology Section, Department of Medicine, University of Oklahoma Health Sciences Center, Oklahoma City, Oklahoma 2Oklahoma Blood Institute, Oklahoma City, Oklahoma Therapeutic plasmapheresis may remove platelets as well as plasma. Unintentional platelet loss, if not recognized, may lead to inappropriate patient assessment and treatment. A patient with thrombotic thrombocytopenic purpura- hemolytic uremic syndrome (TTP-HUS) is reported in whom persistent thrombocytopenia was interpreted as continuing active disease; thrombocytopenia resolved only after plasma exchange treatments were stopped. This observation prompted a systematic study of platelet loss with plasmapheresis. Data are reported on platelet loss during 432 apheresis procedures in 71 patients with six disease categories using three different instruments. Com- paring the first procedure recorded for each patient, there was a significant difference among instrument types ,than with the COBE Spectra (1.6% (21 ס P<0.001); platelet loss was greater with the Fresenius AS 104 (17.5%, N) .With all procedures, platelet loss ranged from 0 to 71% .(24 ס or the Haemonetics LN9000 (2.6%, N (26 ס N Among disease categories, platelet loss was greater in patients with dysproteinemias who were treated for hyper- viscosity symptoms. Absolute platelet loss with the first recorded apheresis procedure, in the 34 patients who had a normal platelet count before the procedure, was also greater with the AS 104 (2.23 × 1011 platelets) than with the Spectra (0.29 × 1011 platelets) or the LN9000 (0.37 × 1011 platelets). -

DRUGS REQUIRING PRIOR AUTHORIZATION in the MEDICAL BENEFIT Page 1
Effective Date: 08/01/2021 DRUGS REQUIRING PRIOR AUTHORIZATION IN THE MEDICAL BENEFIT Page 1 Therapeutic Category Drug Class Trade Name Generic Name HCPCS Procedure Code HCPCS Procedure Code Description Anti-infectives Antiretrovirals, HIV CABENUVA cabotegravir-rilpivirine C9077 Injection, cabotegravir and rilpivirine, 2mg/3mg Antithrombotic Agents von Willebrand Factor-Directed Antibody CABLIVI caplacizumab-yhdp C9047 Injection, caplacizumab-yhdp, 1 mg Cardiology Antilipemic EVKEEZA evinacumab-dgnb C9079 Injection, evinacumab-dgnb, 5 mg Cardiology Hemostatic Agent BERINERT c1 esterase J0597 Injection, C1 esterase inhibitor (human), Berinert, 10 units Cardiology Hemostatic Agent CINRYZE c1 esterase J0598 Injection, C1 esterase inhibitor (human), Cinryze, 10 units Cardiology Hemostatic Agent FIRAZYR icatibant J1744 Injection, icatibant, 1 mg Cardiology Hemostatic Agent HAEGARDA c1 esterase J0599 Injection, C1 esterase inhibitor (human), (Haegarda), 10 units Cardiology Hemostatic Agent ICATIBANT (generic) icatibant J1744 Injection, icatibant, 1 mg Cardiology Hemostatic Agent KALBITOR ecallantide J1290 Injection, ecallantide, 1 mg Cardiology Hemostatic Agent RUCONEST c1 esterase J0596 Injection, C1 esterase inhibitor (recombinant), Ruconest, 10 units Injection, lanadelumab-flyo, 1 mg (code may be used for Medicare when drug administered under Cardiology Hemostatic Agent TAKHZYRO lanadelumab-flyo J0593 direct supervision of a physician, not for use when drug is self-administered) Cardiology Pulmonary Arterial Hypertension EPOPROSTENOL (generic) -
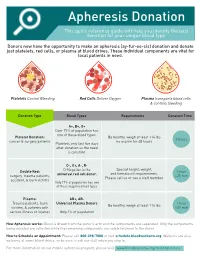
Apheresis Donation This Quick Reference Guide Will Help You Identify the Best Donation for Your Unique Blood Type
Apheresis Donation This quick reference guide will help you identify the best donation for your unique blood type. Donors now have the opportunity to make an apheresis (ay-fur-ee-sis) donation and donate just platelets, red cells, or plasma at blood drives. These individual components are vital for local patients in need. Platelets Control Bleeding Red Cells Deliver Oxygen Plasma transports blood cells & controls bleeding Donation Type Blood Types Requirements Donation Time A+, B+, O+ Over 75% of population has one of these blood types. Platelet Donation: Be healthy, weigh at least 114 lbs 2 hours cancer & surgery patients no aspirin for 48 hours Platelets only last five days after donation so the need is constant. O-, O+, A-, B- Special height, weight, Double Red: O-Negative is the 1 hour and hematocrit requirements. surgery, trauma patients, universal red cell donor. +25 min Please call us or see a staff member accident, & burn victims Only 17% of population has one of these negative blood types Plasma: AB+, AB- Trauma patients, burn Universal Plasma Donors 1 hour Be healthy, weigh at least 114 lbs victims, & patients with +30 min serious illness or injuries Only 4% of population How Apheresis works: Blood is drawn from the donor’s arm and the components are separated. Only the components being donated are collected while the remaining components are safely returned to the donor How to Schedule an Appointment: Please call 800-398-7888 or visit schedule.bloodworksnw.org. Walk-ins are also welcome at some blood drives, so be sure to ask our staff when you stop in. -

Changes to the Technical Manual, 18Th Edition Monday, November 17, 2014 12:00 P.M
Changes to the Technical Manual, 18th Edition Monday, November 17, 2014 12:00 p.m. – 1:30 p.m. (ET) / 5:00p.m. – 6:30 p.m. (GMT) When this file is opened, Acrobat Reader will, by default, display the slides including the Acrobat reader controls. To return to full screen mode, hit Ctrl-L on your computer keyboard or use your mouse to click View>Full Screen on the menu bar of the Acrobat Reader program. To take the slides out of full screen mode and display the Acrobat Reader controls, simply hit the Esc key on your computer keyboard. To advance slides during the program, use the Enter, Page Down, down arrow or right arrow on your computer keyboard. To back up slides during the program, use the Page Up, up arrow of left arrow on your computer keyboard. Please remember to logon to the Live Learning Center using your email address and password to complete an evaluation of the program and speakers. At this time, advance to the next slide and wait for the audioconference to begin. A 2014 Audioconference presented to you by AABB The AABB Technical Manual 18th Edition What’s new? www.aabb.org Technical Manual by the Numbers • 1953 =year of first edition • 69 = number of authors/editors this edition • 370,378 = word count • 96 = number of methods • 60 = number of countries where TM is used www.aabb.org It’s a Process www.aabb.org Overview of Changes Major • Molecular testing • Patient blood management • Cellular therapy • Methods Minor • Throughout www.aabb.org 1: Quality Systems 2: Facilities and Safety • These 2 chapters are comprehensive discussions -

Blood Product Modifications: Leukofiltration, Irradiation and Washing
Blood Product Modifications: Leukofiltration, Irradiation and Washing 1. Leukocyte Reduction Definitions and Standards: o Process also known as leukoreduction, or leukofiltration o Applicable AABB Standards, 25th ed. Leukocyte-reduced RBCs At least 85% of original RBCs < 5 x 106 WBCs in 95% of units tested . Leukocyte-reduced Platelet Concentrates: At least 5.5 x 1010 platelets in 75% of units tested < 8.3 x 105 WBCs in 95% of units tested pH≥6.2 in at least 90% of units tested . Leukocyte-reduced Apheresis Platelets: At least 3.0 x 1011 platelets in 90% of units tested < 5.0 x 106 WBCs 95% of units tested pH≥6.2 in at least 90% of units tested Methods o Filter: “Fourth-generation” filters remove 99.99% WBCs o Apheresis methods: most apheresis machines have built-in leukoreduction mechanisms o Less efficient methods of reducing WBC content . Washing, deglycerolizing after thawing a frozen unit, centrifugation . These methods do not meet requirement of < 5.0 x 106 WBCs per unit of RBCs/apheresis platelets. Types of leukofiltration/leukoreduction o “Pre-storage” . Done within 24 hours of collection . May use inline filters at time of collection (apheresis) or post collection o “Pre-transfusion” leukoreduction/bedside leukoreduction . Done prior to transfusion . “Bedside” leukoreduction uses gravity-based filters at time of transfusion. Least desirable given variability in practice and absence of proficiency . Alternatively performed by transfusion service prior to issuing Benefits of leukoreduction o Prevention of alloimmunization to donor HLA antigens . Anti-HLA can mediate graft rejection and immune mediated destruction of platelets o Leukoreduced products are indicated for transplant recipients or patients who are likely platelet transfusion dependent o Prevention of febrile non-hemolytic transfusion reactions (FNHTR) . -
Hyperleukocytosis (Re)Visited- Is It Case Series Always Leukaemia: a Report of Two Pathology Section Cases and Review of Literature Short Communication
Review Article Clinician’s corner Original Article Images in Medicine Experimental Research Miscellaneous Letter to Editor DOI: 10.7860/JCDR/2020/40556.13409 Case Report Postgraduate Education Hyperleukocytosis (Re)Visited- Is it Case Series always Leukaemia: A Report of Two Pathology Section Cases and Review of Literature Short Communication ASHUTOSH RATH1, RICHA GUPTA2 ABSTRACT Hyperleukocytosis is defined as total leukocyte count of more than 100×109/L. Commonly seen in leukaemic conditions, non- leukaemic causes are usually not encountered and thought of. We report two such non-malignant cases of hyperleukocytosis. A six-year old girl presented with fever, cough and respiratory distress with a leukocyte count of 125.97×109/L. Another case is of a two-month old female infant, who presented with fever and respiratory distress and a leukocyte count of 112.27×109/L. The present case thrives to highlight various possible causes of hyperleukocytosis with an emphasis on non-malignant causes. Also, important complications and management of hyperleukocytosis are discussed. Keywords: Benign, Leukocytosis, Leukostasis CASE REPORT 1 for methicillin-resistant Staphylococcus aureus and was started A six-year-old girl was admitted with complaints of fever, non- on intravenous Vancomycin along with supportive care. Serial productive cough for one week and severe respiratory distress for monitoring of TLC revealed a gradual reduction and it returned to the the past one day. There was no other significant history. On physical baseline of 15×109/L after eight days. The patient was discharged examination, the patient had mild pallor. Respiratory examination after 10 days of hospital stay. -

Novel Emergency Medicine Curriculum Utilizing Self- Directed Learning and the Flipped Classroom Method: Hematologic/Oncologic Emergencies Small Group
CURRICULUM Novel Emergency Medicine Curriculum Utilizing Self- Directed Learning and the Flipped Classroom Method: Hematologic/Oncologic Emergencies Small Group Module Michael G Barrie, MD*, Chad L Mayer, MD*, Colin Kaide, MD*, Emily Kauffman, DO*, Jennifer Mitzman, MD*, Matthew Malone, MD*, Daniel Bachmann, MD*, Ashish Panchal, MD*, Howard Werman, MD*, Benjamin Ostro, MD* and Andrew King, MD* *The Ohio State University Wexner Medical Center, Department of Emergency Medicine, Columbus, OH Correspondence should be addressed to Andrew King, MD, FACEP at [email protected], Twitter: @akingermd Submitted: August 6, 2018; Accepted: November 21, 2018; Electronically Published: January 15, 2019; https://doi.org/10.21980/J8VW56 Copyright: © 2019 Barrie, et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/licenses/by/4.0/ ABSTRACT: Audience: This curriculum, created and implemented at The Ohio State University Wexner Medical Center, was designed to educate our emergency medicine (EM) residents, intern to senior resident level, as well as medical students and attending physicians. Introduction: Disorders of the hematologic system represent a wide spectrum of disease, including bleeding disorders, coagulopathies, blood cell disorders, and oncology related disorders. Hematologic related problems often complicate other disease processes, including malignancy. The number of patients diagnosed with malignancy is increasing due to many factors, including, but not limited to, an aging population and the increased ability for early detection.1 Patients with oncologic conditions can present in a variety of ways with a variety of complications,2 and understanding the steps in diagnosis and management are important components of any emergency physician’s training. -
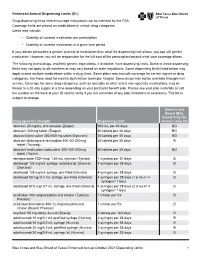
Enhanced Annual Drug List Dispensing Limits
Enhanced Annual Dispensing Limits (DL) Drug dispensing limits help encourage medication use as intended by the FDA. Coverage limits are placed on medications in certain drug categories. Limits may include: • Quantity of covered medication per prescription • Quantity of covered medication in a given time period If your doctor prescribes a greater quantity of medication than what the dispensing limit allows, you can still get the medication. However, you will be responsible for the full cost of the prescription beyond what your coverage allows. The following brand drugs, and their generic equivalents, if available, have dispensing limits. Some of these dispensing limits may not apply to all members or may vary based on state regulations. Some dispensing limits listed below may apply across multiple medications within a drug class. Some plans may exclude coverage for certain agents or drug categories, like those used for erectile dysfunction (example: Viagra). Some drugs may not be available through mail service. Coverage for some drug categories, such as specialty or other select non‑specialty medications, may be limited to a 30‑day supply at a time depending on your particular benefit plan. Please see your plan materials or call the number on the back of your ID card to verify if you are uncertain of any plan limitations or exclusions. This list is subject to change. Generic and Brand (BG), Brand Only (B), Drug (generic) strength Dispensing Limit Generic only (G) abacavir 20 mg/mL oral solution (Ziagen) 960 mL per 30 days BG abacavir 300 -

Digitalcommons@UNMC Granulocytopenia
University of Nebraska Medical Center DigitalCommons@UNMC MD Theses Special Collections 5-1-1936 Granulocytopenia Howard E. Mitchell University of Nebraska Medical Center This manuscript is historical in nature and may not reflect current medical research and practice. Search PubMed for current research. Follow this and additional works at: https://digitalcommons.unmc.edu/mdtheses Part of the Medical Education Commons Recommended Citation Mitchell, Howard E., "Granulocytopenia" (1936). MD Theses. 457. https://digitalcommons.unmc.edu/mdtheses/457 This Thesis is brought to you for free and open access by the Special Collections at DigitalCommons@UNMC. It has been accepted for inclusion in MD Theses by an authorized administrator of DigitalCommons@UNMC. For more information, please contact [email protected]. G PA~lULOCYTOPENI A SENIOR THESIS By Howard E. Mitchell April 17, 1936 TABLE OF CONT'ENTS Introduction Definition • · 1 History . • • • 1 Nomenclature • • • • • 4 ClassificBtion • • • • 6 Physiology • • • • .10 Etiology • • 22 Geographic Distribution • 23 Age, Sex, and R9ce • • ·• 23 Occupation • .. • • • • .. • 23 Ba.cteria • • • • .. 24 Glandu18.r Dysfunction • • • 27 Radiation • • • • 28 Allergy • • • 28 Chemotactic and Maturation Factors • • 28 Chemicals • • • • • 30 Pathology • • • • • 36 Symptoms • • • • • • • 43 DiEtgnosis • • • • • .. • • • • • .. • 4'7 Prognosis 48 '" • • • • • • • • • • • • Treatment • • • • • • • • 49 Non"'specific Therapy • • • • .. 50 Transfusion • • • • .. 51 X-Ray • • • • • • • • • 52 Liver ·Extract • • • • • • • 53 Nucleotides • • • • • • • • • • • 53 General Ca.re • • • • • • • • 57 Conclusion • • • • • • • • • 58 480805 INTHODUCTION Although t~ere is reference in literature of the Nineteenth Century to syndromes similating the disease (granulocytopenia) 9.8 W(~ know it todes, it "vas not un til the year 1922 that Schultz 8ctually described his C8se as a disease entity and by so doing, stimulated the interest of tne medical profession to further in vestigation.