Aron Berhanie Abera
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

In Thyroid Cancer
Metere et al. Cancer Cell Int (2018) 18:7 https://doi.org/10.1186/s12935-018-0504-4 Cancer Cell International PRIMARY RESEARCH Open Access A possible role for selenoprotein glutathione peroxidase (GPx1) and thioredoxin reductases (TrxR1) in thyroid cancer: our experience in thyroid surgery Alessio Metere1* , Francesca Frezzotti1, Claire Elizabeth Graves2, Massimo Vergine1, Alessandro De Luca1, Donatella Pietraforte3 and Laura Giacomelli1 Abstract Background: Oxidative stress is responsible for some alterations in the chemical structure and, consequently, in the function of proteins, lipids, and DNA. Recent studies have linked oxidative stress to cancers, particularly thyroid cancer, but the mechanisms remain unclear. Here, we further characterize the role of oxidative stress in thyroid cancer by analyzing the expression of two selenium antioxidant molecules, glutathione peroxidase (GPx1) and thioredoxin reductase (TrxR1) in thyroid cancer cells. Methods: Samples of both healthy thyroid tissue and thyroid tumor were taken for analysis after total thyroidectomy. The expression of GPx1 and TrxR1 was revealed by Western blot analysis and quantifed by densitometric analy- ses, while the evaluation of free radicals was performed by Electron Paramagnetic Resonance (EPR)-spin trapping technique. Results: Our results show a decrease in the expression of GPx1 and TrxR1 ( 45.7 and 43.2% respectively, p < 0.01) in the thyroid cancer cells compared to the healthy cells. In addition, the EPR− technique− shows an increase of free radicals in tumor tissue, signifcantly higher than that found in healthy thyroid tissue ( 116.3%, p < 0.01). + Conclusions: Our fndings underscore the relationship between thyroid cancer and oxidative stress, showing the imbalance of the oxidant/antioxidant system in thyroid cancer tissue. -
SUPPLEMENTARY DATA Supplementary Figure 1. The
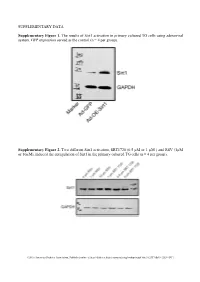
SUPPLEMENTARY DATA Supplementary Figure 1. The results of Sirt1 activation in primary cultured TG cells using adenoviral system. GFP expression served as the control (n = 4 per group). Supplementary Figure 2. Two different Sirt1 activators, SRT1720 (0.5 µM or 1 µM ) and RSV (1µM or 10µM), induced the upregulation of Sirt1 in the primary cultured TG cells (n = 4 per group). ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. Primers used in qPCR Gene Name Primer Sequences Product Size (bp) Sirt1 F: tgccatcatgaagccagaga 241 (NM_001159589) R: aacatcgcagtctccaagga NOX4 F: tgtgcctttattgtgcggag 172 (NM_001285833.1) R: gctgatacactggggcaatg Supplementary Table 2. Antibodies used in Western blot or Immunofluorescence Antibody Company Cat. No Isotype Dilution Sirt1 Santa Cruz * sc-15404 Rabbit IgG 1/200 NF200 Sigma** N5389 Mouse IgG 1/500 Tubulin R&D# MAB1195 Mouse IgG 1/500 NOX4 Abcam† Ab133303 Rabbit IgG 1/500 NOX2 Abcam Ab129068 Rabbit IgG 1/500 phospho-AKT CST‡ #4060 Rabbit IgG 1/500 EGFR CST #4267 Rabbit IgG 1/500 Ki67 Santa Cruz sc-7846 Goat IgG 1/500 * Santa Cruz Biotechnology, Santa Cruz, CA, USA ** Sigma aldrich, Shanghai, China # R&D Systems Inc, Minneapolis, MN, USA † Abcam, Inc., Cambridge, MA, USA ‡ Cell Signaling Technology, Inc., Danvers, MA, USA ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary -

Effect of Prostanoids on Human Platelet Function: an Overview
International Journal of Molecular Sciences Review Effect of Prostanoids on Human Platelet Function: An Overview Steffen Braune, Jan-Heiner Küpper and Friedrich Jung * Institute of Biotechnology, Molecular Cell Biology, Brandenburg University of Technology, 01968 Senftenberg, Germany; steff[email protected] (S.B.); [email protected] (J.-H.K.) * Correspondence: [email protected] Received: 23 October 2020; Accepted: 23 November 2020; Published: 27 November 2020 Abstract: Prostanoids are bioactive lipid mediators and take part in many physiological and pathophysiological processes in practically every organ, tissue and cell, including the vascular, renal, gastrointestinal and reproductive systems. In this review, we focus on their influence on platelets, which are key elements in thrombosis and hemostasis. The function of platelets is influenced by mediators in the blood and the vascular wall. Activated platelets aggregate and release bioactive substances, thereby activating further neighbored platelets, which finally can lead to the formation of thrombi. Prostanoids regulate the function of blood platelets by both activating or inhibiting and so are involved in hemostasis. Each prostanoid has a unique activity profile and, thus, a specific profile of action. This article reviews the effects of the following prostanoids: prostaglandin-D2 (PGD2), prostaglandin-E1, -E2 and E3 (PGE1, PGE2, PGE3), prostaglandin F2α (PGF2α), prostacyclin (PGI2) and thromboxane-A2 (TXA2) on platelet activation and aggregation via their respective receptors. Keywords: prostacyclin; thromboxane; prostaglandin; platelets 1. Introduction Hemostasis is a complex process that requires the interplay of multiple physiological pathways. Cellular and molecular mechanisms interact to stop bleedings of injured blood vessels or to seal denuded sub-endothelium with localized clot formation (Figure1). -

Thiol Peroxidases Mediate Specific Genome-Wide Regulation of Gene Expression in Response to Hydrogen Peroxide
Thiol peroxidases mediate specific genome-wide regulation of gene expression in response to hydrogen peroxide Dmitri E. Fomenkoa,1,2, Ahmet Koca,1, Natalia Agishevaa, Michael Jacobsena,b, Alaattin Kayaa,c, Mikalai Malinouskia,c, Julian C. Rutherfordd, Kam-Leung Siue, Dong-Yan Jine, Dennis R. Winged, and Vadim N. Gladysheva,c,2 aDepartment of Biochemistry, University of Nebraska, Lincoln, NE 68588-0664; bDepartment of Life Sciences, Wayne State College, Wayne, NE 68787; dDepartment of Medicine, University of Utah Health Sciences Center, Salt Lake City, UT 84132; eDepartment of Biochemistry, University of Hong Kong, Hong Kong, China; and cDivision of Genetics, Department of Medicine, Brigham and Women’s Hospital and Harvard Medical School, Boston, MA 02115 Edited by Joan Selverstone Valentine, University of California, Los Angeles, CA, and approved December 22, 2010 (received for review July 21, 2010) Hydrogen peroxide is thought to regulate cellular processes by and could withstand significant oxidative stress. It responded to direct oxidation of numerous cellular proteins, whereas antioxi- several redox stimuli by robust transcriptional reprogramming. dants, most notably thiol peroxidases, are thought to reduce However, it was unable to transcriptionally respond to hydrogen peroxides and inhibit H2O2 response. However, thiol peroxidases peroxide. The data suggested that thiol peroxidases transfer have also been implicated in activation of transcription factors oxidative signals from peroxides to target proteins, thus activating and signaling. It remains unclear if these enzymes stimulate or various transcriptional programs. This study revealed a previously inhibit redox regulation and whether this regulation is widespread undescribed function of these proteins, in addition to their roles or limited to a few cellular components. -

Activation of the Murine EP3 Receptor for PGE2 Inhibits Camp Production and Promotes Platelet Aggregation
Activation of the murine EP3 receptor for PGE2 inhibits cAMP production and promotes platelet aggregation Jean-Etienne Fabre, … , Thomas M. Coffman, Beverly H. Koller J Clin Invest. 2001;107(5):603-610. https://doi.org/10.1172/JCI10881. Article The importance of arachidonic acid metabolites (termed eicosanoids), particularly those derived from the COX-1 and COX-2 pathways (termed prostanoids), in platelet homeostasis has long been recognized. Thromboxane is a potent agonist, whereas prostacyclin is an inhibitor of platelet aggregation. In contrast, the effect of prostaglandin E2 (PGE2) on platelet aggregation varies significantly depending on its concentration. Low concentrations of PGE2 enhance platelet aggregation, whereas high PGE2 levels inhibit aggregation. The mechanism for this dual action of PGE2 is not clear. This study shows that among the four PGE2 receptors (EP1–EP4), activation of EP3 is sufficient to mediate the proaggregatory actions of low PGE2 concentration. In contrast, the prostacyclin receptor (IP) mediates the inhibitory effect of higher PGE2 concentrations. Furthermore, the relative activation of these two receptors, EP3 and IP, regulates the intracellular level of cAMP and in this way conditions the response of the platelet to aggregating agents. Consistent with these findings, loss of the EP3 receptor in a model of venous inflammation protects against formation of intravascular clots. Our results suggest that local production of PGE2 during an inflammatory process can modulate ensuing platelet responses. Find the latest version: https://jci.me/10881/pdf Activation of the murine EP3 receptor for PGE2 inhibits cAMP production and promotes platelet aggregation Jean-Etienne Fabre,1 MyTrang Nguyen,1 Krairek Athirakul,2 Kenneth Coggins,1 John D. -

MK+10-025-Mouse-Model-For-Inflammatory-Bowel-Diseases
Intellectual Property (Non-confidential) NATIONAL MEDICAL CENTER AND BECKMAN RESEARCH INSTITUTE Mouse Model for Inflammatory Bowel Diseases DESCRIPTION This is a transgenic double-knockout (DKO) mouse (Gpx1 and Gpx2). Both GPX1 and GPX2 belong to a selenium-dependent glutathione peroxidase (GPXs) family, which are enzymes that are efficient in the reduction of hydroperoxides. Gpx1 is expressed ubiquitously, and Gpx2 is highly expressed in the epithelium of the gastrointestinal (GI) tract. Gpx1/2-DKO mice have early-onset spontaneous ileocolitis beginning around weaning. These mice are an excellent model for human inflammatory bowel diseases (IBD) because they share similar disease etiology, which includes that the disease severity is highly influenced by genetic background, gut microflora, diet, and higher cancer risk in the distal GI tract. Unlike most mouse IBD models which have disrupted genes playing an important role in regulation of adaptive immunity, these Gpx1/2-DKO mice have intact immunity- which resembles human IBD. KEY ASPECTS • Homozygous disruption of both the endogenous Gpx1 and Gpx2 genes. • Disease characteristics include ileitis, colitis, hypothermia, growth retardation, runting, perianal ulceration, diarrhea, wasting syndrome,, IBD histopathology, tumors/cancer in the ileum or colon PUBLISHED DATA • Esworthy RS, Binder SW, Doroshow JH, and Chu FF, Microflora trigger colitis in mice deficient in selenium-dependent glutathione peroxidase and induce Gpx2 gene expression. Biol. Chem. 384: 597- 607, 2003 • Lee D-H, Esworthy RS, Chu, C, Pfeifer GP, and Chu, F-F. Mutation accumulation in the intestine and colon of mice deficient in two intracellular glutathione peroxidases. Cancer Res. 66: 9845-9851, 2006 • Hahn MA, Hahn T, Lee DH, Esworthy RS, Kim B, Riggs AD, Chu F-F, and Pfeifer GP. -

Characterization of Cytosolic Glutathione Peroxidase And
Aquatic Toxicology 130–131 (2013) 97–111 Contents lists available at SciVerse ScienceDirect Aquatic Toxicology jou rnal homepage: www.elsevier.com/locate/aquatox Characterization of cytosolic glutathione peroxidase and phospholipid-hydroperoxide glutathione peroxidase genes in rainbow trout (Oncorhynchus mykiss) and their modulation by in vitro selenium exposure a a b a d c a,∗ D. Pacitti , T. Wang , M.M. Page , S.A.M. Martin , J. Sweetman , J. Feldmann , C.J. Secombes a Scottish Fish Immunology Research Centre, Institute of Biological and Environmental Sciences, University of Aberdeen, Aberdeen AB24 2TZ, United Kingdom b Integrative and Environmental Physiology, Institute of Biological and Environmental Sciences, University of Aberdeen, Aberdeen AB24 2TZ, United Kingdom c Trace Element Speciation Laboratory, Department of Chemistry, University of Aberdeen, Aberdeen AB24 3UE, United Kingdom d Alltech Biosciences Centre, Sarney, Summerhill Rd, Dunboyne, Country Meath, Ireland a r t i c l e i n f o a b s t r a c t Article history: Selenium (Se) is an oligonutrient with both essential biological functions and recognized harmful effects. Received 4 July 2012 As the selenocysteine (SeCys) amino acid, selenium is integrated in several Se-containing proteins Received in revised form (selenoproteins), many of which are fundamental for cell homeostasis. Nevertheless, selenium may exert 19 December 2012 toxic effects at levels marginally above those required, mainly through the generation of reactive oxygen Accepted 20 December 2012 species (ROS). The selenium chemical speciation can strongly affect the bioavailability of this metal and its impact on metabolism, dictating the levels that can be beneficial or detrimental towards an organism. -

Ramatroban As a Novel Immunotherapy for COVID-19
UC Irvine UC Irvine Previously Published Works Title Ramatroban as a Novel Immunotherapy for COVID-19. Permalink https://escholarship.org/uc/item/94x5m059 Journal Journal of molecular and genetic medicine : an international journal of biomedical research, 14(3) ISSN 1747-0862 Authors Gupta, Ajay Kalantar-Zadeh, Kamyar Reddy, Srinivasa T Publication Date 2020 DOI 10.37421/jmgm.2020.14.457 Peer reviewed eScholarship.org Powered by the California Digital Library University of California HHS Public Access Author manuscript Author ManuscriptAuthor Manuscript Author J Mol Genet Manuscript Author Med. Author Manuscript Author manuscript; available in PMC 2020 September 18. Published in final edited form as: J Mol Genet Med. 2020 ; 14(3): . doi:10.37421/jmgm.2020.14.457. Ramatroban as a Novel Immunotherapy for COVID-19 Ajay Gupta1,*, Kamyar Kalantar-Zadeh1, Srinivasa T. Reddy2 1Division of Nephrology, Hypertension and Kidney Transplantation and Department of Medicine, University of California Irvine (UCI) School of Medicine, United States 2Departments of Medicine, and Molecular and Medical Pharmacology, David Geffen School of Medicine at UCLA, Los Angeles, CA 90095, United States Abstract SARS-CoV-2 virus suppresses host innate and adaptive immune responses, thereby allowing the virus to proliferate, and cause multiorgan failure, especially in the elderly. Respiratory viruses stimulate cyclooxygenase-2 (COX-2) to generate prostanoids including Prostaglandin D2 (PGD2) and thromboxane A2. Furthermore, PGD2 concentrations in the airways increase with aging. PGD2 action mediated via DP2 receptors suppresses both innate and adaptive immune responses, by inhibiting interferon-λ and stimulation of myeloid monocyte-derived suppressor cells respectively. PGD2 and thromboxane A2 actions via the TP receptors activate platelets leading to a prothrombotic state. -

Thromboxane A2 Acts As Tonic Immunoregulator by Preferential Disruption of Low-Avidity CD4+ T Cell–Dendritic Cell Interactions
Published December 8, 2014 Brief Definitive Report Thromboxane A2 acts as tonic immunoregulator by preferential disruption of low-avidity CD4+ T cell–dendritic cell interactions Federica Moalli,1 Jovana Cupovic,2 Flavian Thelen,1 Pascal Halbherr,1 Yoshinori Fukui,3,4 Shuh Narumiya,5 Burkhard Ludewig,2 and Jens V. Stein1 1Theodor Kocher Institute, University of Bern, 3012 Bern, Switzerland 2Institute of Immunobiology, Cantonal Hospital St. Gallen, CH-9007 St. Gallen, Switzerland 3Division of Immunogenetics, Department of Immunobiology and Neuroscience, Medical Institute of Bioregulation and 4Research Center for Advanced Immunology, Kyushu University, Fukuoka 812-8582, Japan 5Department of Pharmacology, Faculty of Pharmaceutical Sciences, Kyoto University, Kyoto 606-8501, Japan Downloaded from Interactions between dendritic cells (DCs) and T cells control the decision between activa- tion and tolerance induction. Thromboxane A2 (TXA2) and its receptor TP have been sug- gested to regulate adaptive immune responses through control of T cell–DC interactions. Here, we show that this control is achieved by selectively reducing expansion of low-avidity CD4+ T cells. During inflammation, weak tetramer-binding TP-deficient CD4+ T cells were jem.rupress.org preferentially expanded compared with TP-proficient CD4+ T cells. Using intravital imaging of cellular interactions in reactive peripheral lymph nodes (PLNs), we found that TXA2 led to disruption of low- but not high-avidity interactions between DCs and CD4+ T cells. Lack of TP correlated with higher expression of activation markers on stimulated CD4+ T cells and with augmented accumulation of follicular helper T cells (TFH), which correlated with on December 9, 2014 increased low-avidity IgG responses. -

Structures of the Human PGD2 Receptor CRTH2 Reveal Novel Mechanisms for Ligand Recognition
Article Structures of the Human PGD2 Receptor CRTH2 Reveal Novel Mechanisms for Ligand Recognition Graphical Abstract Authors Lei Wang, Dandan Yao, R.N.V. Krishna Deepak, ..., Weimin Gong, Zhiyi Wei, Cheng Zhang Correspondence [email protected] (Z.W.), [email protected] (C.Z.) In Brief Wang et al. reported crystal structures of antagonist-bound human CRTH2 as a new asthma drug target. Chemically diverse antagonists occupy a similar semi-occluded pocket with distinct binding modes. Structural analysis suggests a potential ligand entry port and an opposite charge attraction-facilitated binding process for the endogenous CRTH2 ligand prostaglandin D2. Highlights d Crystal structures of antagonist-bound human CRTH2 are solved d A well-structured N terminus covers ligand binding pocket d Conserved and divergent binding features of CRTH2 antagonists are revealed d A multiple-step binding process of prostaglandin D2 is proposed Wang et al., 2018, Molecular Cell 72, 48–59 October 4, 2018 ª 2018 Elsevier Inc. https://doi.org/10.1016/j.molcel.2018.08.009 Molecular Cell Article Structures of the Human PGD2 Receptor CRTH2 Reveal Novel Mechanisms for Ligand Recognition Lei Wang,1,7 Dandan Yao,2,3,7 R.N.V. Krishna Deepak,4 Heng Liu,1 Qingpin Xiao,1,5 Hao Fan,4 Weimin Gong,2,6 Zhiyi Wei,5,* and Cheng Zhang1,8,* 1Department of Pharmacology and Chemical Biology, School of Medicine, University of Pittsburgh, Pittsburgh, PA 15261, USA 2Key Laboratory of RNA Biology, Institute of Biophysics, Chinese Academy of Sciences, Beijing 100101, China 3University -

Glutathione Peroxidase-2 Protects from Allergen-Induced Airway Inflammation in Mice
Eur Respir J 2010; 35: 1148–1154 DOI: 10.1183/09031936.00026108 CopyrightßERS Journals Ltd 2010 Glutathione peroxidase-2 protects from allergen-induced airway inflammation in mice A.M. Dittrich*,#,e, H-A. Meyer*,e, M. Krokowski*, D. Quarcoo*, B. Ahrens*, S.M. Kube", M. Witzenrath", R.S. Esworthy+, F-F. Chu+ and E. Hamelmann*,1 ABSTRACT: The aim of the present study was to identify and validate the biological significance AFFILIATIONS of new genes/proteins involved in the development of allergic airway disease in a murine asthma *Dept of Paediatric Pneumology and Immunology, Charite´ Humboldt model. University, Gene microarrays were used to identify genes with at least a two-fold increase in gene "Dept of Internal Medicine/Infectious expression in lungs of two separate mouse strains with high and low allergic susceptibility. Diseases and Respiratory Medicine, Validation of mRNA data was obtained by western blotting and immunohistochemistry, followed Charite´-Universita¨tsmedizin Berlin, Berlin, by functional analysis of one of the identified genes in mice with targeted disruption of specific #Junior Research Group SFB 587, gene expression. Hannover Medical School, Hannover, Expression of two antioxidant enzymes, glutathione peroxidase-2 (GPX2) and glutathione S- 1University Children’s Hospital, Ruhr transferase omega (GSTO) 1-1 was increased in both mouse strains after induction of allergic University, Bochum, Germany. +Dept of Radiation Biology, Beckman airway disease and localised in lung epithelial cells. Mice with targeted disruption of the Gpx-2 Research Institute of City of Hope, gene showed significantly enhanced airway inflammation compared to sensitised and challenged Duarte, CA, USA. wild-type mice. -

Oxygenated Fatty Acids Enhance Hematopoiesis Via the Receptor GPR132
Oxygenated Fatty Acids Enhance Hematopoiesis via the Receptor GPR132 The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation Lahvic, Jamie L. 2017. Oxygenated Fatty Acids Enhance Hematopoiesis via the Receptor GPR132. Doctoral dissertation, Harvard University, Graduate School of Arts & Sciences. Citable link http://nrs.harvard.edu/urn-3:HUL.InstRepos:42061504 Terms of Use This article was downloaded from Harvard University’s DASH repository, and is made available under the terms and conditions applicable to Other Posted Material, as set forth at http:// nrs.harvard.edu/urn-3:HUL.InstRepos:dash.current.terms-of- use#LAA Oxygenated Fatty Acids Enhance Hematopoiesis via the Receptor GPR132 A dissertation presented by Jamie L. Lahvic to The Division of Medical Sciences in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the subject of Developmental and Regenerative Biology Harvard University Cambridge, Massachusetts May 2017 © 2017 Jamie L. Lahvic All rights reserved. Dissertation Advisor: Leonard I. Zon Jamie L. Lahvic Oxygenated Fatty Acids Enhance Hematopoiesis via the Receptor GPR132 Abstract After their specification in early development, hematopoietic stem cells (HSCs) maintain the entire blood system throughout adulthood as well as upon transplantation. The processes of HSC specification, renewal, and homing to the niche are regulated by protein, as well as lipid signaling molecules. A screen for chemical enhancers of marrow transplant in the zebrafish identified the endogenous lipid signaling molecule 11,12-epoxyeicosatrienoic acid (11,12-EET). EET has vasodilatory properties, but had no previously described function on HSCs.