Node of Ranvier Length As a Potential Regulator of Myelinated Axon
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Vocabulario De Morfoloxía, Anatomía E Citoloxía Veterinaria
Vocabulario de Morfoloxía, anatomía e citoloxía veterinaria (galego-español-inglés) Servizo de Normalización Lingüística Universidade de Santiago de Compostela COLECCIÓN VOCABULARIOS TEMÁTICOS N.º 4 SERVIZO DE NORMALIZACIÓN LINGÜÍSTICA Vocabulario de Morfoloxía, anatomía e citoloxía veterinaria (galego-español-inglés) 2008 UNIVERSIDADE DE SANTIAGO DE COMPOSTELA VOCABULARIO de morfoloxía, anatomía e citoloxía veterinaria : (galego-español- inglés) / coordinador Xusto A. Rodríguez Río, Servizo de Normalización Lingüística ; autores Matilde Lombardero Fernández ... [et al.]. – Santiago de Compostela : Universidade de Santiago de Compostela, Servizo de Publicacións e Intercambio Científico, 2008. – 369 p. ; 21 cm. – (Vocabularios temáticos ; 4). - D.L. C 2458-2008. – ISBN 978-84-9887-018-3 1.Medicina �������������������������������������������������������������������������veterinaria-Diccionarios�������������������������������������������������. 2.Galego (Lingua)-Glosarios, vocabularios, etc. políglotas. I.Lombardero Fernández, Matilde. II.Rodríguez Rio, Xusto A. coord. III. Universidade de Santiago de Compostela. Servizo de Normalización Lingüística, coord. IV.Universidade de Santiago de Compostela. Servizo de Publicacións e Intercambio Científico, ed. V.Serie. 591.4(038)=699=60=20 Coordinador Xusto A. Rodríguez Río (Área de Terminoloxía. Servizo de Normalización Lingüística. Universidade de Santiago de Compostela) Autoras/res Matilde Lombardero Fernández (doutora en Veterinaria e profesora do Departamento de Anatomía e Produción Animal. -

Clustering of Na+ Channels and Node of Ranvier Formation in Remyelinating Axons
The Journal of Neuroscience, January 1995, 15(l): 492503 Clustering of Na+ Channels and Node of Ranvier Formation in Remyelinating Axons Sanja Dugandgija-NovakoviC,’ Adam G. Koszowski,2 S. Rock Levinson,2 and Peter Shragerl ‘Department of Physiology, University of Rochester Medical Center, Rochester, New York 14642 and 2Department of Physiology, University of Colorado Health Sciences Center, Denver, Colorado 80262 Polyclonal antibodies were raised against a well conserved nodal regions(Black et al., 1990). The density of Na+ channels, region of the vertebrate Na+ channel and were affinity pu- in particular, is about 25 times higher at nodesof Ranvier than rified for use in immunocytochemistry. Focal demyelination at internodal sites (Shrager, 1989). There has been vigorous of rat sciatic axons was initiated by an intraneural injection debate over the mechanism of Na+ channel clustering during of lysolecithin and Na+ channel clustering was followed at myelination, particularly with respect to the role of Schwann several stages of myelin removal and repair. At 1 week post- cells, and studies have included both developing nerve and injection axons contained long, fully demyelinated regions. pathological conditions (Ellisman, 1979; Rosenbluth, 1979; Ro- Na+ channel clusters appeared only at heminodes forming senbluth and Blakemore, 1984; Le Beau et al., 1987; England the borders of these zones, and at widely spaced isolated et al., 1990, 1991; Joe and Angelides, 1992, 1993).There remain sites that may represent former nodes of Ranvier. Over the many interesting questions, particularly regarding remodeling next few days proliferating Schwann cells adhered to axons that occurs following myelin disruption. When axons are de- and began to extend processes. -

Myelin Biogenesis Is Associated with Pathological Ultrastructure That Is
bioRxiv preprint doi: https://doi.org/10.1101/2021.02.02.429485; this version posted February 4, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 1 Myelin biogenesis is associated with pathological ultrastructure that 2 is resolved by microglia during development 3 4 5 Minou Djannatian1,2*, Ulrich Weikert3, Shima Safaiyan1,2, Christoph Wrede4, Cassandra 6 Deichsel1,2, Georg Kislinger1,2, Torben Ruhwedel3, Douglas S. Campbell5, Tjakko van Ham6, 7 Bettina Schmid2, Jan Hegermann4, Wiebke Möbius3, Martina Schifferer2,7, Mikael Simons1,2,7* 8 9 1Institute of Neuronal Cell Biology, Technical University Munich, 80802 Munich, Germany 10 2German Center for Neurodegenerative Diseases (DZNE), 81377 Munich, Germany 11 3Max-Planck Institute of Experimental Medicine, 37075 Göttingen, Germany 12 4Institute of Functional and Applied Anatomy, Research Core Unit Electron Microscopy, 13 Hannover Medical School, 30625, Hannover, Germany 14 5Department of Neuronal Remodeling, Graduate School of Pharmaceutical Sciences, Kyoto 15 University, Sakyo-ku, Kyoto 606-8501, Japan. 16 6Department of Clinical Genetics, Erasmus MC, University Medical Center Rotterdam, 3015 CN, 17 the Netherlands 18 7Munich Cluster of Systems Neurology (SyNergy), 81377 Munich, Germany 19 20 *Correspondence: [email protected] or [email protected] 21 Keywords 22 Myelination, degeneration, phagocytosis, microglia, oligodendrocytes, phosphatidylserine 23 24 1 bioRxiv preprint doi: https://doi.org/10.1101/2021.02.02.429485; this version posted February 4, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. -
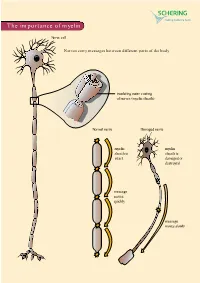
The Importance of Myelin
The importance of myelin Nerve cell Nerves carry messages between different parts of the body insulating outer coating of nerves (myelin sheath) Normal nerve Damaged nerve myelin myelin sheath is sheath is intact damaged or destroyed message moves quickly message moves slowly Nerve cells transmit impulses Nerve cells have a long, thin, flexible fibre that transmits impulses. These impulses are electrical signals that travel along the length of the nerve. Nerve fibres are long to enable impulses to travel between distant parts of the body, such as the spinal cord and leg muscles. Myelin speeds up impulses Most nerve fibres are surrounded by an insulating, fatty sheath called myelin, which acts to speed up impulses. The myelin sheath contains periodic breaks called nodes of Ranvier. By jumping from node to node, the impulse can travel much more quickly than if it had to travel along the entire length of the nerve fibre. Myelinated nerves can transmit a signal at speeds as high as 100 metres per second – as fast as a Formula One racing car. normal damaged nerve nerve Loss of myelin leads to a variety of symptoms If the myelin sheath surrounding nerve fibres is damaged or destroyed, transmission of nerve impulses is slowed or blocked. The impulse now has to flow continuously along the whole nerve fibre – a process that is much slower than jumping from node to node. Loss of myelin can also lead to ‘short-circuiting’ of nerve impulses. An area where myelin has been destroyed is called a lesion or plaque. This slowing and ‘short-circuiting’ of nerve impulses by lesions leads to a variety of symptoms related to nervous system activity. -

Regulation of Myelin Structure and Conduction Velocity by Perinodal Astrocytes
Correction NEUROSCIENCE Correction for “Regulation of myelin structure and conduc- tion velocity by perinodal astrocytes,” by Dipankar J. Dutta, Dong Ho Woo, Philip R. Lee, Sinisa Pajevic, Olena Bukalo, William C. Huffman, Hiroaki Wake, Peter J. Basser, Shahriar SheikhBahaei, Vanja Lazarevic, Jeffrey C. Smith, and R. Douglas Fields, which was first published October 29, 2018; 10.1073/ pnas.1811013115 (Proc. Natl. Acad. Sci. U.S.A. 115,11832–11837). The authors note that the following statement should be added to the Acknowledgments: “We acknowledge Dr. Hae Ung Lee for preliminary experiments that informed the ultimate experimental approach.” Published under the PNAS license. Published online June 10, 2019. www.pnas.org/cgi/doi/10.1073/pnas.1908361116 12574 | PNAS | June 18, 2019 | vol. 116 | no. 25 www.pnas.org Downloaded by guest on October 2, 2021 Regulation of myelin structure and conduction velocity by perinodal astrocytes Dipankar J. Duttaa,b, Dong Ho Wooa, Philip R. Leea, Sinisa Pajevicc, Olena Bukaloa, William C. Huffmana, Hiroaki Wakea, Peter J. Basserd, Shahriar SheikhBahaeie, Vanja Lazarevicf, Jeffrey C. Smithe, and R. Douglas Fieldsa,1 aSection on Nervous System Development and Plasticity, The Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health, Bethesda, MD 20892; bThe Henry M. Jackson Foundation for the Advancement of Military Medicine, Inc., Bethesda, MD 20817; cMathematical and Statistical Computing Laboratory, Office of Intramural Research, Center for Information -
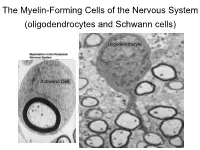
The Myelin-Forming Cells of the Nervous System (Oligodendrocytes and Schwann Cells)
The Myelin-Forming Cells of the Nervous System (oligodendrocytes and Schwann cells) Oligodendrocyte Schwann Cell Oligodendrocyte function Saltatory (jumping) nerve conduction Oligodendroglia PMD PMD Saltatory (jumping) nerve conduction Investigating the Myelinogenic Potential of Individual Oligodendrocytes In Vivo Sparse Labeling of Oligodendrocytes CNPase-GFP Variegated expression under the MBP-enhancer Cerebral Cortex Corpus Callosum Cerebral Cortex Corpus Callosum Cerebral Cortex Caudate Putamen Corpus Callosum Cerebral Cortex Caudate Putamen Corpus Callosum Corpus Callosum Cerebral Cortex Caudate Putamen Corpus Callosum Ant Commissure Corpus Callosum Cerebral Cortex Caudate Putamen Piriform Cortex Corpus Callosum Ant Commissure Characterization of Oligodendrocyte Morphology Cerebral Cortex Corpus Callosum Caudate Putamen Cerebellum Brain Stem Spinal Cord Oligodendrocytes in disease: Cerebral Palsy ! CP major cause of chronic neurological morbidity and mortality in children ! CP incidence now about 3/1000 live births compared to 1/1000 in 1980 when we started intervening for ELBW ! Of all ELBW {gestation 6 mo, Wt. 0.5kg} , 10-15% develop CP ! Prematurely born children prone to white matter injury {WMI}, principle reason for the increase in incidence of CP ! ! 12 Cerebral Palsy Spectrum of white matter injury ! ! Macro Cystic Micro Cystic Gliotic Khwaja and Volpe 2009 13 Rationale for Repair/Remyelination in Multiple Sclerosis Oligodendrocyte specification oligodendrocytes specified from the pMN after MNs - a ventral source of oligodendrocytes -

Myelin Oligodendrocyte Glycoprotein (MOG) Antibody Disease
MOG ANTIBODY DISEASE Myelin Oligodendrocyte Glycoprotein (MOG) Antibody-Associated Encephalomyelitis WHAT IS MOG ANTIBODY-ASSOCIATED DEMYELINATION? Myelin oligodendrocyte glycoprotein (MOG) antibody-associated demyelination is an immune-mediated inflammatory process that affects the central nervous system (brain and spinal cord). MOG is a protein that exists on the outer surface of cells that create myelin (an insulating layer around nerve fibers). In a small number of patients, an initial episode of inflammation due to MOG antibodies may be the first manifestation of multiple sclerosis (MS). Most patients may only experience one episode of inflammation, but repeated episodes of central nervous system demyelination can occur in some cases. WHAT ARE THE SYMPTOMS? • Optic neuritis (inflammation of the optic nerve(s)) may be a symptom of MOG antibody- associated demyelination, which may result in painful loss of vision in one or both eyes. • Transverse myelitis (inflammation of the spinal cord) may cause a variety of symptoms that include: o Abnormal sensations. Numbness, tingling, coldness or burning in the arms and/or legs. Some are especially sensitive to the light touch of clothing or to extreme heat or cold. You may feel as if something is tightly wrapping the skin of your chest, abdomen or legs. o Weakness in your arms or legs. Some people notice that they're stumbling or dragging one foot, or heaviness in the legs. Others may develop severe weakness or even total paralysis. o Bladder and bowel problems. This may include needing to urinate more frequently, urinary incontinence, difficulty urinating and constipation. • Acute disseminated encephalomyelitis (ADEM) may cause loss of vision, weakness, numbness, and loss of balance, and altered mental status. -

Targeting Myelin Lipid Metabolism As a Potential Therapeutic Strategy in a Model of CMT1A Neuropathy
ARTICLE DOI: 10.1038/s41467-018-05420-0 OPEN Targeting myelin lipid metabolism as a potential therapeutic strategy in a model of CMT1A neuropathy R. Fledrich 1,2,3, T. Abdelaal 1,4,5, L. Rasch1,4, V. Bansal6, V. Schütza1,3, B. Brügger7, C. Lüchtenborg7, T. Prukop1,4,8, J. Stenzel1,4, R.U. Rahman6, D. Hermes 1,4, D. Ewers 1,4, W. Möbius 1,9, T. Ruhwedel1, I. Katona 10, J. Weis10, D. Klein11, R. Martini11, W. Brück12, W.C. Müller3, S. Bonn 6,13, I. Bechmann2, K.A. Nave1, R.M. Stassart 1,3,12 & M.W. Sereda1,4 1234567890():,; In patients with Charcot–Marie–Tooth disease 1A (CMT1A), peripheral nerves display aberrant myelination during postnatal development, followed by slowly progressive demye- lination and axonal loss during adult life. Here, we show that myelinating Schwann cells in a rat model of CMT1A exhibit a developmental defect that includes reduced transcription of genes required for myelin lipid biosynthesis. Consequently, lipid incorporation into myelin is reduced, leading to an overall distorted stoichiometry of myelin proteins and lipids with ultrastructural changes of the myelin sheath. Substitution of phosphatidylcholine and phosphatidylethanolamine in the diet is sufficient to overcome the myelination deficit of affected Schwann cells in vivo. This treatment rescues the number of myelinated axons in the peripheral nerves of the CMT rats and leads to a marked amelioration of neuropathic symptoms. We propose that lipid supplementation is an easily translatable potential therapeutic approach in CMT1A and possibly other dysmyelinating neuropathies. 1 Department of Neurogenetics, Max-Planck-Institute of Experimental Medicine, Göttingen 37075, Germany. -

Cytochemical Differentiation of the Axon Membrane in A- Andc-Fibres
J Neurol Neurosurg Psychiatry: first published as 10.1136/jnnp.40.4.379 on 1 April 1977. Downloaded from Journal ofNeurology, Neurosurgery, and Psychiatry, 1977, 40, 379-385 Cytochemical differentiation of the axon membrane in A- and C-fibres S. G. WAXMAN AND D. C. QUICK From the Department ofNeurology, Harvard Medical School, Beth Israel Hospital, Boston, and Program in Health Sciences and Technology, and Research Laboratory ofElectronics, Massachusetts Institute of Technology, Cambridge, Massachusetts, USA SUMMARY Guinea pig and rat sciatic nerves were fixed with cacodylate-buffered aldehydes and OS04, and were stained with ferric ion and ferrocyanide. Cytoplasmic surfaces of the non-myelinated nodal axon membrane ofA-fibres display distinct electron-dense aggregates of stain. These aggregates were not observed in association with the paranodal or internodal axolemma. The membranes of C-fibres exhibit no staining under these conditions. Thus, the nodal axolemma of normal myelinated fibres is structurally distinct from both the myelinated internodal membrane, and from the axolemma of C-fibres. The ferric ion-ferrocyanide technique may provide a method for marking axonal mem- brane with normal nodal properties. Protected by copyright. In previous studies (Quick and Waxman, 1977) we therefore, extended our studies on the distribution of demonstrated that, under appropriate conditions, ferric ion binding to peripheral nerve axon mem- ferric ion is bound to the cytoplasmic surface of the branes. In the present paper we show that cytoplasmic axon membrane at nodes of Ranvier in mammalian ferric ion binding occurs only for A-fibre nodal peripheral myelinated axons. The binding of ferric membrane, and does not occur for C-fibres in the ion occurred specifically at the nodal axon membrane, peripheral nervous system. -

A Translation Insight Into the Scientific Textbook
NEUROPHYSIOLOGY: A TRANSLATION INSIGHT INTO THE SCIENTIFIC TEXTBOOK MASTER’S DISSERTATION ON MEDICAL TRANSLATION PRACTICE MÁSTER UNIVERSITARIO EN TRADUCCIÓN MÉDICO-SANITARIA (2017/2018) Esther Andrés Caballo Supervisors: Dr. Laura Carasusán Senosiáin (Universitat Jaume I) Dr. Rocío Baños-Piñero (CenTraS-UCL) Acknowledgments This dissertation would have not been possible but for the support of many people. In the first place, I am particularly grateful to the Master’s faculty at UJI who gave me the insight and educational input into the medical translation that is needed for competence and subject-knowledge acquisition to enter into this profession. I would like to thank them all personally since I have most learnt from their lectures, feedback on my translation work, and recommendations during the master’s course of studies. Secondly, I am extremely grateful to the Erasmus+ Master Exchange Programme, whereby a Higher Education Learning Agreement for Studies was signed by and between Universitat Jaume I (Spain) and University College London (UK), which gave me the great opportunity of a five-month stay at University College London. In this prestigious university, particularly in the Centre for Translation Studies (CenTraS), I have done my translation practice on-line, conducted my research and written down this dissertation, while making full employ of the numberless resources available at the Main and Science Libraries and the Institute of Physiology at UCL. I highly appreciate the welcoming and availability of CenTraS’ administrators and teaching staff, and specially, the priceless support of my dissertation supervisor. Thirdly, I must acknowledge the wisdom of the masters, and devotedly thank Dr. Ignacio Navascués and their team, Dr. -

The Effects of Normal Aging on Myelin and Nerve Fibers: a Review∗
Journal of Neurocytology 31, 581–593 (2002) The effects of normal aging on myelin and nerve fibers: A review∗ ALAN PETERS Department of Anatomy and Neurobiology, Boston University School of Medicine, 715, Albany Street, Boston, MA 02118, USA [email protected] Received 7 January 2003; revised 4 March 2003; accepted 5 March 2003 Abstract It was believed that the cause of the cognitive decline exhibited by human and non-human primates during normal aging was a loss of cortical neurons. It is now known that significant numbers of cortical neurons are not lost and other bases for the cognitive decline have been sought. One contributing factor may be changes in nerve fibers. With age some myelin sheaths exhibit degenerative changes, such as the formation of splits containing electron dense cytoplasm, and the formation on myelin balloons. It is suggested that such degenerative changes lead to cognitive decline because they cause changes in conduction velocity, resulting in a disruption of the normal timing in neuronal circuits. Yet as degeneration occurs, other changes, such as the formation of redundant myelin and increasing thickness suggest of sheaths, suggest some myelin formation is continuing during aging. Another indication of this is that oligodendrocytes increase in number with age. In addition to the myelin changes, stereological studies have shown a loss of nerve fibers from the white matter of the cerebral hemispheres of humans, while other studies have shown a loss of nerve fibers from the optic nerves and anterior commissure in monkeys. It is likely that such nerve fiber loss also contributes to cognitive decline, because of the consequent decrease in connections between neurons. -

Differential Distribution of Closely Related Potassium Channels in Rat Schwann Cells
The Journal of Neuroscience, May 1995, 15(5): 3761-3774 Differential Distribution of Closely Related Potassium Channels in Rat Schwann Cells Huaiyu Mi,’ Thomas J. Deerinck,2 Mark H. Ellisman, and Thomas L. Schwarz’ ‘Department of Molecular and Cellular Physiology, Beckman Center, Stanford University, Stanford, California 94305 and ?San Diego Microscopy and Imaging Resource, Department of Neuroscience, School of Medicine, UCSD, La Jolla, California 92093 Closely related K+ channels can coassemble to form het- 1992; Salkoff et al., 1992). The function of these molecularly eromultimers in expression systems, as well as in vivo. defined channels within their normal environment is less well Whether in vivo this coassembly is random and inevitable understood. It has been difficult to correlate cloned and ex- or whether highly homologous channels can be segregated pressed channels with currents that are observed in vivo. More- and targeted independently within a given cell has not been over, it is not known how channels become distributed in ap- determined. In this study, we address these questions by propriate domains within the cell or how the association of sub- characterizing and localizing voltage-dependent K+ chan- units is regulated. Finally, in many cell types, the function of nels in Schwann cells. Transcripts for three closely related the channel in the physiology of the cell is not understood. This members of the Shaker-like family of K+ channels are is particularly true in the case of voltage-dependent K+ channels found in adult rat sciatic nerve: Kvl.1, Kv1.2, and Kv1.5. in electrically inexcitable cells such as Schwann cells.