KRSA: Network-Based Prediction of Differential Kinase Activity from Kinome Array Data
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supplemental Information to Mammadova-Bach Et Al., “Laminin Α1 Orchestrates VEGFA Functions in the Ecosystem of Colorectal Carcinogenesis”
Supplemental information to Mammadova-Bach et al., “Laminin α1 orchestrates VEGFA functions in the ecosystem of colorectal carcinogenesis” Supplemental material and methods Cloning of the villin-LMα1 vector The plasmid pBS-villin-promoter containing the 3.5 Kb of the murine villin promoter, the first non coding exon, 5.5 kb of the first intron and 15 nucleotides of the second villin exon, was generated by S. Robine (Institut Curie, Paris, France). The EcoRI site in the multi cloning site was destroyed by fill in ligation with T4 polymerase according to the manufacturer`s instructions (New England Biolabs, Ozyme, Saint Quentin en Yvelines, France). Site directed mutagenesis (GeneEditor in vitro Site-Directed Mutagenesis system, Promega, Charbonnières-les-Bains, France) was then used to introduce a BsiWI site before the start codon of the villin coding sequence using the 5’ phosphorylated primer: 5’CCTTCTCCTCTAGGCTCGCGTACGATGACGTCGGACTTGCGG3’. A double strand annealed oligonucleotide, 5’GGCCGGACGCGTGAATTCGTCGACGC3’ and 5’GGCCGCGTCGACGAATTCACGC GTCC3’ containing restriction site for MluI, EcoRI and SalI were inserted in the NotI site (present in the multi cloning site), generating the plasmid pBS-villin-promoter-MES. The SV40 polyA region of the pEGFP plasmid (Clontech, Ozyme, Saint Quentin Yvelines, France) was amplified by PCR using primers 5’GGCGCCTCTAGATCATAATCAGCCATA3’ and 5’GGCGCCCTTAAGATACATTGATGAGTT3’ before subcloning into the pGEMTeasy vector (Promega, Charbonnières-les-Bains, France). After EcoRI digestion, the SV40 polyA fragment was purified with the NucleoSpin Extract II kit (Machery-Nagel, Hoerdt, France) and then subcloned into the EcoRI site of the plasmid pBS-villin-promoter-MES. Site directed mutagenesis was used to introduce a BsiWI site (5’ phosphorylated AGCGCAGGGAGCGGCGGCCGTACGATGCGCGGCAGCGGCACG3’) before the initiation codon and a MluI site (5’ phosphorylated 1 CCCGGGCCTGAGCCCTAAACGCGTGCCAGCCTCTGCCCTTGG3’) after the stop codon in the full length cDNA coding for the mouse LMα1 in the pCIS vector (kindly provided by P. -

Gene Symbol Gene Description ACVR1B Activin a Receptor, Type IB
Table S1. Kinase clones included in human kinase cDNA library for yeast two-hybrid screening Gene Symbol Gene Description ACVR1B activin A receptor, type IB ADCK2 aarF domain containing kinase 2 ADCK4 aarF domain containing kinase 4 AGK multiple substrate lipid kinase;MULK AK1 adenylate kinase 1 AK3 adenylate kinase 3 like 1 AK3L1 adenylate kinase 3 ALDH18A1 aldehyde dehydrogenase 18 family, member A1;ALDH18A1 ALK anaplastic lymphoma kinase (Ki-1) ALPK1 alpha-kinase 1 ALPK2 alpha-kinase 2 AMHR2 anti-Mullerian hormone receptor, type II ARAF v-raf murine sarcoma 3611 viral oncogene homolog 1 ARSG arylsulfatase G;ARSG AURKB aurora kinase B AURKC aurora kinase C BCKDK branched chain alpha-ketoacid dehydrogenase kinase BMPR1A bone morphogenetic protein receptor, type IA BMPR2 bone morphogenetic protein receptor, type II (serine/threonine kinase) BRAF v-raf murine sarcoma viral oncogene homolog B1 BRD3 bromodomain containing 3 BRD4 bromodomain containing 4 BTK Bruton agammaglobulinemia tyrosine kinase BUB1 BUB1 budding uninhibited by benzimidazoles 1 homolog (yeast) BUB1B BUB1 budding uninhibited by benzimidazoles 1 homolog beta (yeast) C9orf98 chromosome 9 open reading frame 98;C9orf98 CABC1 chaperone, ABC1 activity of bc1 complex like (S. pombe) CALM1 calmodulin 1 (phosphorylase kinase, delta) CALM2 calmodulin 2 (phosphorylase kinase, delta) CALM3 calmodulin 3 (phosphorylase kinase, delta) CAMK1 calcium/calmodulin-dependent protein kinase I CAMK2A calcium/calmodulin-dependent protein kinase (CaM kinase) II alpha CAMK2B calcium/calmodulin-dependent -
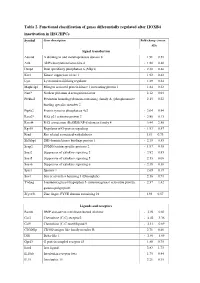
Table 2. Functional Classification of Genes Differentially Regulated After HOXB4 Inactivation in HSC/Hpcs
Table 2. Functional classification of genes differentially regulated after HOXB4 inactivation in HSC/HPCs Symbol Gene description Fold-change (mean ± SD) Signal transduction Adam8 A disintegrin and metalloprotease domain 8 1.91 ± 0.51 Arl4 ADP-ribosylation factor-like 4 - 1.80 ± 0.40 Dusp6 Dual specificity phosphatase 6 (Mkp3) - 2.30 ± 0.46 Ksr1 Kinase suppressor of ras 1 1.92 ± 0.42 Lyst Lysosomal trafficking regulator 1.89 ± 0.34 Mapk1ip1 Mitogen activated protein kinase 1 interacting protein 1 1.84 ± 0.22 Narf* Nuclear prelamin A recognition factor 2.12 ± 0.04 Plekha2 Pleckstrin homology domain-containing. family A. (phosphoinosite 2.15 ± 0.22 binding specific) member 2 Ptp4a2 Protein tyrosine phosphatase 4a2 - 2.04 ± 0.94 Rasa2* RAS p21 activator protein 2 - 2.80 ± 0.13 Rassf4 RAS association (RalGDS/AF-6) domain family 4 3.44 ± 2.56 Rgs18 Regulator of G-protein signaling - 1.93 ± 0.57 Rrad Ras-related associated with diabetes 1.81 ± 0.73 Sh3kbp1 SH3 domain kinase bindings protein 1 - 2.19 ± 0.53 Senp2 SUMO/sentrin specific protease 2 - 1.97 ± 0.49 Socs2 Suppressor of cytokine signaling 2 - 2.82 ± 0.85 Socs5 Suppressor of cytokine signaling 5 2.13 ± 0.08 Socs6 Suppressor of cytokine signaling 6 - 2.18 ± 0.38 Spry1 Sprouty 1 - 2.69 ± 0.19 Sos1 Son of sevenless homolog 1 (Drosophila) 2.16 ± 0.71 Ywhag 3-monooxygenase/tryptophan 5- monooxygenase activation protein. - 2.37 ± 1.42 gamma polypeptide Zfyve21 Zinc finger. FYVE domain containing 21 1.93 ± 0.57 Ligands and receptors Bambi BMP and activin membrane-bound inhibitor - 2.94 ± 0.62 -

Allosteric Activation of Functionally Asymmetric RAF Kinase Dimers
Allosteric Activation of Functionally Asymmetric RAF Kinase Dimers Jiancheng Hu,1,2 Edward C. Stites,1 Haiyang Yu,1 Elizabeth A. Germino,1 Hiruy S. Meharena,3,4 Philip J.S. Stork,6 Alexandr P. Kornev,3,4,5 Susan S. Taylor,3,4,5,* and Andrey S. Shaw1,2,* 1Department of Pathology and Immunology 2Howard Hughes Medical Institute Washington University School of Medicine, 660 South Euclid, Box 8118, St. Louis, MO 63110, USA 3Department of Chemistry/Biochemistry 4Department of Pharmacology 5Howard Hughes Medical Institute University of California San Diego, 9500 Gilman Drive, Mail code 0654, La Jolla, CA 92093, USA 6Vollum Institute, Oregon Health & Science University, 3181 SW Sam Jackson Park Road, Portland, OR 97239, USA *Correspondence: [email protected] (S.S.T.), [email protected] (A.S.S.) http://dx.doi.org/10.1016/j.cell.2013.07.046 SUMMARY There are three isoforms of RAF: A-RAF, B-RAF, and C-RAF. Each appears to have a distinct mechanism of activation (Baljuls Although RAF kinases are critical for controlling et al., 2007; Galabova-Kovacs et al., 2006; Wimmer and Baccar- cell growth, their mechanism of activation is incom- ini, 2010). BRAF is considered to be more active and have a pletely understood. Recently, dimerization was simpler mechanism of activation in comparison to CRAF and shown to be important for activation. Here we show ARAF (Chong et al., 2001; Rebocho and Marais, 2013; Zhang that the dimer is functionally asymmetric with one and Guan, 2000). This difference can potentially explain why kinase functioning as an activator to stimulate activ- BRAF mutations are so common in cancer, in contrast to CRAF and ARAF, where mutations are rarely found (Davies ity of the partner, receiver kinase. -

Anti-SEK1 / MKK4 Phospho (Ser80) Antibody (ARG51673)
Product datasheet [email protected] ARG51673 Package: 100 μl, 50 μl anti-SEK1 / MKK4 phospho (Ser80) antibody Store at: -20°C Summary Product Description Rabbit Polyclonal antibody recognizes SEK1 / MKK4 phospho (Ser80) Tested Reactivity Hu, Ms, Rat Tested Application ICC/IF, IHC-P, WB Host Rabbit Clonality Polyclonal Isotype IgG Target Name SEK1 / MKK4 Antigen Species Human Immunogen Peptide sequence around phosphorylation site of serine 80 (T-H-S(p)-I-E) derived from Human SEK1/MKK4. Conjugation Un-conjugated Alternate Names MEK 4; MAPK/ERK kinase 4; PRKMK4; SAPKK-1; SAPK/ERK kinase 1; SKK1; JNK-activating kinase 1; EC 2.7.12.2; MEK4; MAP kinase kinase 4; c-Jun N-terminal kinase kinase 1; SEK1; SAPKK1; MAPKK4; Stress- activated protein kinase kinase 1; JNKK1; MKK4; SERK1; SAPK kinase 1; Dual specificity mitogen- activated protein kinase kinase 4; JNKK; MAPKK 4 Application Instructions Application table Application Dilution ICC/IF 1:100 - 1:200 IHC-P 1:50 - 1:100 WB 1:500 - 1:1000 Application Note * The dilutions indicate recommended starting dilutions and the optimal dilutions or concentrations should be determined by the scientist. Calculated Mw 44 kDa Properties Form Liquid Purification Antibodies were produced by immunizing rabbits with KLH-conjugated synthetic phosphopeptide. Antibodies were purified by affinity-chromatography using epitope-specific phosphopeptide. In addition, non-phospho specific antibodies were removed by chromatogramphy using non- phosphopeptide. Buffer PBS (without Mg2+ and Ca2+, pH 7.4), 150mM NaCl, 0.02% Sodium azide and 50% Glycerol. Preservative 0.02% Sodium azide Stabilizer 50% Glycerol www.arigobio.com 1/3 Concentration 1 mg/ml Storage instruction For continuous use, store undiluted antibody at 2-8°C for up to a week. -

Map2k1 and Map2k2 Genes Contribute to the Normal Development of Syncytiotrophoblasts During Placentation
RESEARCH ARTICLE 1363 Development 136, 1363-1374 (2009) doi:10.1242/dev.031872 Map2k1 and Map2k2 genes contribute to the normal development of syncytiotrophoblasts during placentation Valérie Nadeau*, Stéphanie Guillemette*, Louis-François Bélanger, Olivier Jacob, Sophie Roy and Jean Charron† The mammalian genome contains two ERK/MAP kinase kinase genes, Map2k1 and Map2k2, which encode dual-specificity kinases responsible for ERK/MAP kinase activation. In the mouse, loss of Map2k1 function causes embryonic lethality, whereas Map2k2 mutants survive with a normal lifespan, suggesting that Map2k1 masks the phenotype due to the Map2k2 mutation. To uncover the specific function of MAP2K2 and the threshold requirement of MAP2K proteins during embryo formation, we have successively ablated the Map2k gene functions. We report here that Map2k2 haploinsufficiency affects the normal development of placenta in the absence of one Map2k1 allele. Most Map2k1+/–Map2k2+/– embryos die during gestation because of placenta defects restricted to extra-embryonic tissues. The impaired viability of Map2k1+/–Map2k2+/– embryos can be rescued when the Map2k1 deletion is restricted to the embryonic tissues. The severity of the placenta phenotype is dependent on the number of Map2k mutant alleles, the deletion of the Map2k1 allele being more deleterious. Moreover, the deletion of one or both Map2k2 alleles in the context of one null Map2k1 allele leads to the formation of multinucleated trophoblast giant (MTG) cells. Genetic experiments indicate that these structures are derived from Gcm1-expressing syncytiotrophoblasts (SynT), which are affected in their ability to form the uniform SynT layer II lining the maternal sinuses. Thus, even though Map2k1 plays a predominant role, these results enlighten the function of Map2k2 in placenta development. -

Advancing a Clinically Relevant Perspective of the Clonal Nature of Cancer
Advancing a clinically relevant perspective of the clonal nature of cancer Christian Ruiza,b, Elizabeth Lenkiewicza, Lisa Eversa, Tara Holleya, Alex Robesona, Jeffrey Kieferc, Michael J. Demeurea,d, Michael A. Hollingsworthe, Michael Shenf, Donna Prunkardf, Peter S. Rabinovitchf, Tobias Zellwegerg, Spyro Moussesc, Jeffrey M. Trenta,h, John D. Carpteni, Lukas Bubendorfb, Daniel Von Hoffa,d, and Michael T. Barretta,1 aClinical Translational Research Division, Translational Genomics Research Institute, Scottsdale, AZ 85259; bInstitute for Pathology, University Hospital Basel, University of Basel, 4031 Basel, Switzerland; cGenetic Basis of Human Disease, Translational Genomics Research Institute, Phoenix, AZ 85004; dVirginia G. Piper Cancer Center, Scottsdale Healthcare, Scottsdale, AZ 85258; eEppley Institute for Research in Cancer and Allied Diseases, Nebraska Medical Center, Omaha, NE 68198; fDepartment of Pathology, University of Washington, Seattle, WA 98105; gDivision of Urology, St. Claraspital and University of Basel, 4058 Basel, Switzerland; hVan Andel Research Institute, Grand Rapids, MI 49503; and iIntegrated Cancer Genomics Division, Translational Genomics Research Institute, Phoenix, AZ 85004 Edited* by George F. Vande Woude, Van Andel Research Institute, Grand Rapids, MI, and approved June 10, 2011 (received for review March 11, 2011) Cancers frequently arise as a result of an acquired genomic insta- on the basis of morphology alone (8). Thus, the application of bility and the subsequent clonal evolution of neoplastic cells with purification methods such as laser capture microdissection does variable patterns of genetic aberrations. Thus, the presence and not resolve the complexities of many samples. A second approach behaviors of distinct clonal populations in each patient’s tumor is to passage tumor biopsies in tissue culture or in xenografts (4, 9– may underlie multiple clinical phenotypes in cancers. -

KSR1- and ERK-Dependent Translational Regulation of 2 the Epithelial-To-Mesenchymal Transition
bioRxiv preprint doi: https://doi.org/10.1101/2021.01.18.427224; this version posted January 21, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 1 KSR1- and ERK-dependent Translational Regulation of 2 the Epithelial-to-Mesenchymal Transition 3 Chaitra Rao1, Danielle E. Frodyma1, Siddesh Southekal2, Robert A. Svoboda3, Adrian R. Black1, 4 Chittibabu Guda2, Tomohiro Mizutani5, Hans Clevers5, Keith R. Johnson1,4, Kurt W. Fisher3, and 5 Robert E. Lewis1* 6 1 Eppley Institute, Fred & Pamela Buffett Cancer Center, University of Nebraska Medical Center, Omaha, NE 68198, USA. 7 2 Department of Genetics, Cell Biology and Anatomy, University of Nebraska Medical Center, Omaha, NE, University of 8 Nebraska Medical Center, Omaha, NE 68198, USA. 9 3 Department of Pathology and Microbiology, University of Nebraska Medical Center, Omaha, NE 68198, USA. 10 4 Department of Oral Biology, University of Nebraska Medical Center, Omaha, NE 68198, USA 11 5 Hubrecht Institute, Royal Netherlands Academy of Arts and Sciences (KNAW) and UMC Utrecht, 3584CT Utrecht, The 12 Netherlands 13 *Correspondence: [email protected] 14 Abstract: 15 The epithelial-to-mesenchymal transition (EMT) is considered a transcriptional process 16 that induces a switch in cells from a polarized state to a migratory phenotype. Here we show that 17 KSR1 and ERK promote EMT through the preferential translation of Epithelial-Stromal 18 Interaction 1 (EPSTI1), which is required to induce the switch from E- to N-cadherin and 19 coordinate migratory and invasive behavior. -

The Genetic Landscape of Clinical Resistance to RAF Inhibition in Metastatic Melanoma
CD-13-0617_PAP.indd Page OF1 19/11/13 9:36 PM user-f028 /Books-Arts/JOURNAL-Cancer%20Discovery/01-JAN-Issue-2014/PAP Published OnlineFirst November 21, 2013; DOI: 10.1158/2159-8290.CD-13-0617 RESEARCH ARTICLE The Genetic Landscape of Clinical Resistance to RAF Inhibition in Metastatic Melanoma Eliezer M. Van Allen 1 , 3 , Nikhil Wagle 1 , 3 , Antje Sucker 5 , 6 , Daniel J. Treacy 1 , Cory M. Johannessen 3 , Eva M. Goetz 1 , Chelsea S. Place 1 , 3 , Amaro Taylor-Weiner 3 , Steven Whittaker 3 , Gregory V. Kryukov 3 , Eran Hodis 1 , 3,4 , Mara Rosenberg 3 , Aaron McKenna 3 , 15 , Kristian Cibulskis 3 , Deborah Farlow 3 , Lisa Zimmer 5 , 6 , Uwe Hillen 5 , 6 , Ralf Gutzmer 8 , Simone M. Goldinger 16 , Selma Ugurel 9 , Helen J. Gogas 17 , Friederike Egberts 10 , Carola Berking 6 , 11 , Uwe Trefzer 6 , 12 , Carmen Loquai 6 , 13 , Benjamin Weide 6 , 14 , Jessica C. Hassel 6 , 7 , Stacey B. Gabriel 3 , Scott L. Carter 3 , Gad Getz 2 , 3 , Levi A. Garraway 1 , 3 , and Dirk Schadendorf 5 , 6 on behalf of the Dermatologic Cooperative Oncology Group of Germany (DeCOG) Downloaded from cancerdiscovery.aacrjournals.org on September 25, 2021. © 2013 American Association for Cancer Research. CD-13-0617_PAP.indd Page OF2 19/11/13 9:36 PM user-f028 /Books-Arts/JOURNAL-Cancer%20Discovery/01-JAN-Issue-2014/PAP Published OnlineFirst November 21, 2013; DOI: 10.1158/2159-8290.CD-13-0617 ABSTRACT Most patients with BRAF V600 -mutant metastatic melanoma develop resistance to selective RAF kinase inhibitors. The spectrum of clinical genetic resistance mechanisms to RAF inhibitors and options for salvage therapy are incompletely understood. -

PDF Download
MEK-7 Polyclonal Antibody Catalog No : YT2725 Reactivity : Human,Mouse,Rat Applications : WB,ELISA Gene Name : MAP2K7 Protein Name : Dual specificity mitogen-activated protein kinase kinase 7 Human Gene Id : 5609 Human Swiss Prot O14733 No : Mouse Gene Id : 26400 Mouse Swiss Prot Q8CE90 No : Rat Gene Id : 363855 Rat Swiss Prot No : Q4KSH7 Immunogen : The antiserum was produced against synthesized peptide derived from human MAP2K7. AA range:241-290 Specificity : MEK-7 Polyclonal Antibody detects endogenous levels of MEK-7 protein. Formulation : Liquid in PBS containing 50% glycerol, 0.5% BSA and 0.02% sodium azide. Source : Rabbit Dilution : Western Blot: 1/500 - 1/2000. ELISA: 1/20000. Not yet tested in other applications. Purification : The antibody was affinity-purified from rabbit antiserum by affinity- chromatography using epitope-specific immunogen. Concentration : 1 mg/ml 1 / 3 Storage Stability : -20°C/1 year Molecularweight : 47485 Observed Band : 47 Cell Pathway : MAPK_ERK_Growth,MAPK_G_Protein,ErbB_HER,Toll_Like,T_Cell_Receptor, Fc epsilon RI,Neurotrophin,GnRH, Background : mitogen-activated protein kinase kinase 7(MAP2K7) Homo sapiens The protein encoded by this gene is a dual specificity protein kinase that belongs to the MAP kinase kinase family. This kinase specifically activates MAPK8/JNK1 and MAPK9/JNK2, and this kinase itself is phosphorylated and activated by MAP kinase kinase kinases including MAP3K1/MEKK1, MAP3K2/MEKK2,MAP3K3/MEKK5, and MAP4K2/GCK. This kinase is involved in the signal transduction mediating the cell -

Application of a MYC Degradation
SCIENCE SIGNALING | RESEARCH ARTICLE CANCER Copyright © 2019 The Authors, some rights reserved; Application of a MYC degradation screen identifies exclusive licensee American Association sensitivity to CDK9 inhibitors in KRAS-mutant for the Advancement of Science. No claim pancreatic cancer to original U.S. Devon R. Blake1, Angelina V. Vaseva2, Richard G. Hodge2, McKenzie P. Kline3, Thomas S. K. Gilbert1,4, Government Works Vikas Tyagi5, Daowei Huang5, Gabrielle C. Whiten5, Jacob E. Larson5, Xiaodong Wang2,5, Kenneth H. Pearce5, Laura E. Herring1,4, Lee M. Graves1,2,4, Stephen V. Frye2,5, Michael J. Emanuele1,2, Adrienne D. Cox1,2,6, Channing J. Der1,2* Stabilization of the MYC oncoprotein by KRAS signaling critically promotes the growth of pancreatic ductal adeno- carcinoma (PDAC). Thus, understanding how MYC protein stability is regulated may lead to effective therapies. Here, we used a previously developed, flow cytometry–based assay that screened a library of >800 protein kinase inhibitors and identified compounds that promoted either the stability or degradation of MYC in a KRAS-mutant PDAC cell line. We validated compounds that stabilized or destabilized MYC and then focused on one compound, Downloaded from UNC10112785, that induced the substantial loss of MYC protein in both two-dimensional (2D) and 3D cell cultures. We determined that this compound is a potent CDK9 inhibitor with a previously uncharacterized scaffold, caused MYC loss through both transcriptional and posttranslational mechanisms, and suppresses PDAC anchorage- dependent and anchorage-independent growth. We discovered that CDK9 enhanced MYC protein stability 62 through a previously unknown, KRAS-independent mechanism involving direct phosphorylation of MYC at Ser . -

Comparative Analysis of Protein Expression Concomitant with DNA Methyltransferase 3A Depletion in a Melanoma Cell Line
American Journal of Analytical Chemistry, 2011, 2, 539-572 doi:10.4236/ajac.2011.25064 Published Online September 2011 (http://www.SciRP.org/journal/ajac) Comparative Analysis of Protein Expression Concomitant with DNA Methyltransferase 3A Depletion in a Melanoma Cell Line Shengnan Tang1,#, Xiaoyan Liu1,#, Tonghua Li1*, Haoyue Wang2, Jiangming Sun1, Qian Qiao1, Jun Yao3, Jian Fei2 1Department of Chemistry, Tongji University, Shanghai, China 2School of Life Science & Technology, Tongji University, Shanghai, China 3School of Medicine, Fudan University, Shanghai, China E-mail: *[email protected] Received March 17, 2011; revised May 3, 2011; accepted June 1, 2011 Abstract DNA methyltransferase 3A (Dnmt3a), a de novo methyltransferase, has attracted a great deal of attention for its important role played in tumorigenesis. We have previously demonstrated that melanoma is unable to grow in-vivo in conditions of Dnmt3a depletion in a mouse model. In this study, we cultured the Dnmt3a depletion B16 melanoma (Dnmt3a-D) cell line to conduct a comparative analysis of protein expression con-comitant with Dnmt3a depletion in a melanoma cell line. After two-dimensional separation, by gel elec- tro-phoresis and liquid chromatography, combined with mass spectrometry analysis (1DE-LC-MS/MS), the re-sults demonstrated that 467 proteins were up-regulated and 535 proteins were down-regulated in the Dnmt3a-D cell line compared to the negative control (NC) cell line. The Genome Ontology (GO) and KEGG pathway were used to further analyze the altered proteins. KEGG pathway analysis indicated that the MAPK signaling pathway exhibited a greater alteration in proteins, an interesting finding due to the close rela- tion-ship with tumorigenesis.