Double Anaerobic Coverage: What Is the Role in Clinical Practice?
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Role of Nanobiosensors in Therapeutic Drug Monitoring
Journal of Personalized Medicine Review Personalized Medicine for Antibiotics: The Role of Nanobiosensors in Therapeutic Drug Monitoring Vivian Garzón 1, Rosa-Helena Bustos 2 and Daniel G. Pinacho 2,* 1 PhD Biosciences Program, Universidad de La Sabana, Chía 140013, Colombia; [email protected] 2 Therapeutical Evidence Group, Clinical Pharmacology, Universidad de La Sabana, Chía 140013, Colombia; [email protected] * Correspondence: [email protected]; Tel.: +57-1-8615555 (ext. 23309) Received: 21 August 2020; Accepted: 7 September 2020; Published: 25 September 2020 Abstract: Due to the high bacterial resistance to antibiotics (AB), it has become necessary to adjust the dose aimed at personalized medicine by means of therapeutic drug monitoring (TDM). TDM is a fundamental tool for measuring the concentration of drugs that have a limited or highly toxic dose in different body fluids, such as blood, plasma, serum, and urine, among others. Using different techniques that allow for the pharmacokinetic (PK) and pharmacodynamic (PD) analysis of the drug, TDM can reduce the risks inherent in treatment. Among these techniques, nanotechnology focused on biosensors, which are relevant due to their versatility, sensitivity, specificity, and low cost. They provide results in real time, using an element for biological recognition coupled to a signal transducer. This review describes recent advances in the quantification of AB using biosensors with a focus on TDM as a fundamental aspect of personalized medicine. Keywords: biosensors; therapeutic drug monitoring (TDM), antibiotic; personalized medicine 1. Introduction The discovery of antibiotics (AB) ushered in a new era of progress in controlling bacterial infections in human health, agriculture, and livestock [1] However, the use of AB has been challenged due to the appearance of multi-resistant bacteria (MDR), which have increased significantly in recent years due to AB mismanagement and have become a global public health problem [2]. -

Polymyxin B in Combination with Doripenem Against Heteroresistant Acinetobacter Baumannii: Pharmacodynamics of New Dosing Strategies
J Antimicrob Chemother 2016; 71: 3148–3156 doi:10.1093/jac/dkw293 Advance Access publication 3 August 2016 Polymyxin B in combination with doripenem against heteroresistant Acinetobacter baumannii: pharmacodynamics of new dosing strategies Gauri G. Rao1,2, Neang S. Ly1,2,Ju¨rgen B. Bulitta1,3, Rachel L. Soon1,2, Marie D. San Roman1,2, Patricia N. Holden1,2, Cornelia B. Landersdorfer4, Roger L. Nation4, Jian Li4, Alan Forrest1,5 and Brian T. Tsuji1,2* 1Laboratory for Antimicrobial Pharmacodynamics, School of Pharmacy and Pharmaceutical Sciences, University at Buffalo, The State University of New York, Buffalo, NY, USA; 2The New York State Center of Excellence in Bioinformatics & Life Sciences, University at Buffalo, The State University of New York, Buffalo, NY, USA; 3Center for Pharmacometrics and Systems Pharmacology, College of Pharmacy, University of Florida, Orlando, FL, USA; 4Drug Delivery, Disposition and Dynamics, Monash Institute of Pharmaceutical Sciences, Monash University, Melbourne, Australia; 5Eshelman School of Pharmacy, The University of North Carolina at Chapel Hill, NC, USA *Corresponding author. Tel: +1-716-881-7543; Fax: +1-716-849-6890; E-mail: [email protected] Received 13 October 2015; returned 3 January 2016; revised 14 June 2016; accepted 19 June 2016 Objectives: Polymyxin B is being increasingly utilized as a last resort against resistant Gram-negative bacteria. We examined the pharmacodynamics of novel dosing strategies for polymyxin B combinations to maximize efficacy and minimize the emergence of resistance and drug exposure against Acinetobacter baumannii. Methods: The pharmacodynamics of polymyxin B together with doripenem were evaluated in time–kill experi- ments over 48 h against 108 cfu/mL of two polymyxin-heteroresistant A. -

Medical Review(S) Clinical Review
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 200327 MEDICAL REVIEW(S) CLINICAL REVIEW Application Type NDA Application Number(s) 200327 Priority or Standard Standard Submit Date(s) December 29, 2009 Received Date(s) December 30, 2009 PDUFA Goal Date October 30, 2010 Division / Office Division of Anti-Infective and Ophthalmology Products Office of Antimicrobial Products Reviewer Name(s) Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD Review Completion October 29, 2010 Date Established Name Ceftaroline fosamil for injection (Proposed) Trade Name Teflaro Therapeutic Class Cephalosporin; ß-lactams Applicant Cerexa, Inc. Forest Laboratories, Inc. Formulation(s) 400 mg/vial and 600 mg/vial Intravenous Dosing Regimen 600 mg every 12 hours by IV infusion Indication(s) Acute Bacterial Skin and Skin Structure Infection (ABSSSI); Community-acquired Bacterial Pneumonia (CABP) Intended Population(s) Adults ≥ 18 years of age Template Version: March 6, 2009 Reference ID: 2857265 Clinical Review Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD NDA 200327: Teflaro (ceftaroline fosamil) Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT ......................................... 9 1.1 Recommendation on Regulatory Action ........................................................... 10 1.2 Risk Benefit Assessment.................................................................................. 10 1.3 Recommendations for Postmarketing Risk Evaluation and Mitigation Strategies ........................................................................................................................ -

National Antibiotic Consumption for Human Use in Sierra Leone (2017–2019): a Cross-Sectional Study
Tropical Medicine and Infectious Disease Article National Antibiotic Consumption for Human Use in Sierra Leone (2017–2019): A Cross-Sectional Study Joseph Sam Kanu 1,2,* , Mohammed Khogali 3, Katrina Hann 4 , Wenjing Tao 5, Shuwary Barlatt 6,7, James Komeh 6, Joy Johnson 6, Mohamed Sesay 6, Mohamed Alex Vandi 8, Hannock Tweya 9, Collins Timire 10, Onome Thomas Abiri 6,11 , Fawzi Thomas 6, Ahmed Sankoh-Hughes 12, Bailah Molleh 4, Anna Maruta 13 and Anthony D. Harries 10,14 1 National Disease Surveillance Programme, Sierra Leone National Public Health Emergency Operations Centre, Ministry of Health and Sanitation, Cockerill, Wilkinson Road, Freetown, Sierra Leone 2 Department of Community Health, Faculty of Clinical Sciences, College of Medicine and Allied Health Sciences, University of Sierra Leone, Freetown, Sierra Leone 3 Special Programme for Research and Training in Tropical Diseases (TDR), World Health Organization, 1211 Geneva, Switzerland; [email protected] 4 Sustainable Health Systems, Freetown, Sierra Leone; [email protected] (K.H.); [email protected] (B.M.) 5 Unit for Antibiotics and Infection Control, Public Health Agency of Sweden, Folkhalsomyndigheten, SE-171 82 Stockholm, Sweden; [email protected] 6 Pharmacy Board of Sierra Leone, Central Medical Stores, New England Ville, Freetown, Sierra Leone; [email protected] (S.B.); [email protected] (J.K.); [email protected] (J.J.); [email protected] (M.S.); [email protected] (O.T.A.); [email protected] (F.T.) Citation: Kanu, J.S.; Khogali, M.; 7 Department of Pharmaceutics and Clinical Pharmacy & Therapeutics, Faculty of Pharmaceutical Sciences, Hann, K.; Tao, W.; Barlatt, S.; Komeh, College of Medicine and Allied Health Sciences, University of Sierra Leone, Freetown 0000, Sierra Leone 8 J.; Johnson, J.; Sesay, M.; Vandi, M.A.; Directorate of Health Security & Emergencies, Ministry of Health and Sanitation, Sierra Leone National Tweya, H.; et al. -

Pharmacodynamic Evaluation of Suppression of in Vitro Resistance In
www.nature.com/scientificreports OPEN Pharmacodynamic evaluation of suppression of in vitro resistance in Acinetobacter baumannii strains using polymyxin B‑based combination therapy Nayara Helisandra Fedrigo1, Danielle Rosani Shinohara1, Josmar Mazucheli2, Sheila Alexandra Belini Nishiyama1, Floristher Elaine Carrara‑Marroni3, Frederico Severino Martins4, Peijuan Zhu5, Mingming Yu6, Sherwin Kenneth B. Sy2 & Maria Cristina Bronharo Tognim1* The emergence of polymyxin resistance in Gram‑negative bacteria infections has motivated the use of combination therapy. This study determined the mutant selection window (MSW) of polymyxin B alone and in combination with meropenem and fosfomycin against A. baumannii strains belonging to clonal lineages I and III. To evaluate the inhibition of in vitro drug resistance, we investigate the MSW‑derived pharmacodynamic indices associated with resistance to polymyxin B administrated regimens as monotherapy and combination therapy, such as the percentage of each dosage interval that free plasma concentration was within the MSW (%TMSW) and the percentage of each dosage interval that free plasma concentration exceeded the mutant prevention concentration (%T>MPC). The MSW of polymyxin B varied between 1 and 16 µg/mL for polymyxin B‑susceptible strains. The triple combination of polymyxin B with meropenem and fosfomycin inhibited the polymyxin B‑resistant subpopulation in meropenem‑resistant isolates and polymyxin B plus meropenem as a double combination sufciently inhibited meropenem‑intermediate, and susceptible strains. T>MPC 90% was reached for polymyxin B in these combinations, while %TMSW was 0 against all strains. TMSW for meropenem and fosfomycin were also reduced. Efective antimicrobial combinations signifcantly reduced MSW. The MSW‑derived pharmacodynamic indices can be used for the selection of efective combination regimen to combat the polymyxin B‑resistant strain. -

Cusumano-Et-Al-2017.Pdf
International Journal of Infectious Diseases 63 (2017) 1–6 Contents lists available at ScienceDirect International Journal of Infectious Diseases journal homepage: www.elsevier.com/locate/ijid Rapidly growing Mycobacterium infections after cosmetic surgery in medical tourists: the Bronx experience and a review of the literature a a b c b Lucas R. Cusumano , Vivy Tran , Aileen Tlamsa , Philip Chung , Robert Grossberg , b b, Gregory Weston , Uzma N. Sarwar * a Albert Einstein College of Medicine, Montefiore Medical Center, Bronx, New York, USA b Division of Infectious Diseases, Department of Medicine, Albert Einstein College of Medicine, Montefiore Medical Center, Bronx, New York, USA c Department of Pharmacy, Nebraska Medicine, Omaha, Nebraska, USA A R T I C L E I N F O A B S T R A C T Article history: Background: Medical tourism is increasingly popular for elective cosmetic surgical procedures. However, Received 10 May 2017 medical tourism has been accompanied by reports of post-surgical infections due to rapidly growing Received in revised form 22 July 2017 mycobacteria (RGM). The authors’ experience working with patients with RGM infections who have Accepted 26 July 2017 returned to the USA after traveling abroad for cosmetic surgical procedures is described here. Corresponding Editor: Eskild Petersen, Methods: Patients who developed RGM infections after undergoing cosmetic surgeries abroad and who ?Aarhus, Denmark presented at the Montefiore Medical Center (Bronx, New York, USA) between August 2015 and June 2016 were identified. A review of patient medical records was performed. Keywords: Results: Four patients who presented with culture-proven RGM infections at the sites of recent cosmetic Mycobacterium abscessus complex procedures were identified. -

Crystal Structure and Stability of Gyrase–Fluoroquinolone Cleaved Complexes from Mycobacterium Tuberculosis
Crystal structure and stability of gyrase–fluoroquinolone cleaved complexes from Mycobacterium tuberculosis Tim R. Blowera,1, Benjamin H. Williamsonb, Robert J. Kernsb, and James M. Bergera,2 aDepartment of Biophysics and Biophysical Chemistry, Johns Hopkins University School of Medicine, Baltimore, MD 21205; and bDivision of Medicinal and Natural Products Chemistry, University of Iowa, Iowa City, IA 52242 This contribution is part of the special series of Inaugural Articles by members of the National Academy of Sciences elected in 2013. Contributed by James M. Berger, December 22, 2015 (sent for review October 28, 2015; reviewed by Benjamin Bax and Yuk-Ching Tse-Dinh) Mycobacterium tuberculosis (Mtb) infects one-third of the world’s threatens both first-line and second-line use (11). The wide- population and in 2013 accounted for 1.5 million deaths. Fluoro- spread testing of fluoroquinolones against TB has revealed quinolone antibacterials, which target DNA gyrase, are critical considerable variation in efficacy of different drug variants agents used to halt the progression from multidrug-resistant tu- against Mtb. For example, ciprofloxacin is only marginally active, berculosis to extensively resistant disease; however, fluoroquino- and its early use with Mtb was halted in favor of ofloxacin and lone resistance is emerging and new ways to bypass resistance are levofloxacin (7). These two agents are now proving to be less required. To better explain known differences in fluoroquinolone effective than moxifloxacin and gatifloxacin (7, 10); however, the action, the crystal structures of the WT Mtb DNA gyrase cleavage newest two compounds also exhibit some nonideality. For ex- core and a fluoroquinolone-sensitized mutant were determined in ample, gatifloxacin can elicit side effects such as hypo/hypergly- complex with DNA and five fluoroquinolones. -

General Items
Essential Medicines List (EML) 2019 Application for the inclusion of imipenem/cilastatin, meropenem and amoxicillin/clavulanic acid in the WHO Model List of Essential Medicines, as reserve second-line drugs for the treatment of multidrug-resistant tuberculosis (complementary lists of anti-tuberculosis drugs for use in adults and children) General items 1. Summary statement of the proposal for inclusion, change or deletion This application concerns the updating of the forthcoming WHO Model List of Essential Medicines (EML) and WHO Model List of Essential Medicines for Children (EMLc) to include the following medicines: 1) Imipenem/cilastatin (Imp-Cln) to the main list but NOT the children’s list (it is already mentioned on both lists as an option in section 6.2.1 Beta Lactam medicines) 2) Meropenem (Mpm) to both the main and the children’s lists (it is already on the list as treatment for meningitis in section 6.2.1 Beta Lactam medicines) 3) Clavulanic acid to both the main and the children’s lists (it is already listed as amoxicillin/clavulanic acid (Amx-Clv), the only commercially available preparation of clavulanic acid, in section 6.2.1 Beta Lactam medicines) This application makes reference to amendments recommended in particular to section 6.2.4 Antituberculosis medicines in the latest editions of both the main EML (20th list) and the EMLc (6th list) released in 2017 (1),(2). On the basis of the most recent Guideline Development Group advising WHO on the revision of its guidelines for the treatment of multidrug- or rifampicin-resistant (MDR/RR-TB)(3), the applicant considers that the three agents concerned be viewed as essential medicines for these forms of TB in countries. -

Severe Sepsis and Septic Shock Antibiotic Guide
Stanford Health Issue Date: 05/2017 Stanford Antimicrobial Safety and Sustainability Program Severe Sepsis and Septic Shock Antibiotic Guide Table 1: Antibiotic selection options for healthcare associated and/or immunocompromised patients • Healthcare associated: intravenous therapy, wound care, or intravenous chemotherapy within the prior 30 days, residence in a nursing home or other long-term care facility, hospitalization in an acute care hospital for two or more days within the prior 90 days, attendance at a hospital or hemodialysis clinic within the prior 30 days • Immunocompromised: Receiving chemotherapy, known systemic cancer not in remission, ANC <500, severe cell-mediated immune deficiency Table 2: Antibiotic selection options for community acquired, immunocompetent patients Table 3: Antibiotic selection options for patients with simple sepsis, community acquired, immunocompetent patients requiring hospitalization. Risk Factors for Select Organisms P. aeruginosa MRSA Invasive Candidiasis VRE (and other resistant GNR) Community acquired: • Known colonization with MDROs • Central venous catheter • Liver transplant • Prior IV antibiotics within 90 day • Recent MRSA infection • Broad-spectrum antibiotics • Known colonization • Known colonization with MDROs • Known MRSA colonization • + 1 of the following risk factors: • Prolonged broad antibacterial • Skin & Skin Structure and/or IV access site: ♦ Parenteral nutrition therapy Hospital acquired: ♦ Purulence ♦ Dialysis • Prolonged profound • Prior IV antibiotics within 90 days ♦ Abscess -

Tetracycline and Sulfonamide Antibiotics in Soils: Presence, Fate and Environmental Risks
processes Review Tetracycline and Sulfonamide Antibiotics in Soils: Presence, Fate and Environmental Risks Manuel Conde-Cid 1, Avelino Núñez-Delgado 2 , María José Fernández-Sanjurjo 2 , Esperanza Álvarez-Rodríguez 2, David Fernández-Calviño 1,* and Manuel Arias-Estévez 1 1 Soil Science and Agricultural Chemistry, Faculty Sciences, University Vigo, 32004 Ourense, Spain; [email protected] (M.C.-C.); [email protected] (M.A.-E.) 2 Department Soil Science and Agricultural Chemistry, Engineering Polytechnic School, University Santiago de Compostela, 27002 Lugo, Spain; [email protected] (A.N.-D.); [email protected] (M.J.F.-S.); [email protected] (E.Á.-R.) * Correspondence: [email protected] Received: 30 October 2020; Accepted: 13 November 2020; Published: 17 November 2020 Abstract: Veterinary antibiotics are widely used worldwide to treat and prevent infectious diseases, as well as (in countries where allowed) to promote growth and improve feeding efficiency of food-producing animals in livestock activities. Among the different antibiotic classes, tetracyclines and sulfonamides are two of the most used for veterinary proposals. Due to the fact that these compounds are poorly absorbed in the gut of animals, a significant proportion (up to ~90%) of them are excreted unchanged, thus reaching the environment mainly through the application of manures and slurries as fertilizers in agricultural fields. Once in the soil, antibiotics are subjected to a series of physicochemical and biological processes, which depend both on the antibiotic nature and soil characteristics. Adsorption/desorption to soil particles and degradation are the main processes that will affect the persistence, bioavailability, and environmental fate of these pollutants, thus determining their potential impacts and risks on human and ecological health. -
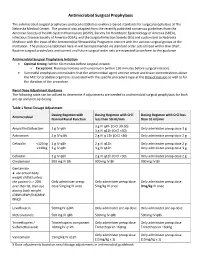
Antimicrobial Surgical Prophylaxis
Antimicrobial Surgical Prophylaxis The antimicrobial surgical prophylaxis protocol establishes evidence-based standards for surgical prophylaxis at The Nebraska Medical Center. The protocol was adapted from the recently published consensus guidelines from the American Society of Health-System Pharmacists (ASHP), Society for Healthcare Epidemiology of America (SHEA), Infectious Disease Society of America (IDSA), and the Surgical Infection Society (SIS) and customized to Nebraska Medicine with the input of the Antimicrobial Stewardship Program in concert with the various surgical groups at the institution. The protocol established here-in will be implemented via standard order sets utilized within One Chart. Routine surgical prophylaxis and current and future surgical order sets are expected to conform to this guidance. Antimicrobial Surgical Prophylaxis Initiation Optimal timing: Within 60 minutes before surgical incision o Exceptions: Fluoroquinolones and vancomycin (within 120 minutes before surgical incision) Successful prophylaxis necessitates that the antimicrobial agent achieve serum and tissue concentrations above the MIC for probable organisms associated with the specific procedure type at the time of incision as well as for the duration of the procedure. Renal Dose Adjustment Guidance The following table can be utilized to determine if adjustments are needed to antimicrobial surgical prophylaxis for both pre-op and post-op dosing. Table 1 Renal Dosage Adjustment Dosing Regimen with Dosing Regimen with CrCl Dosing Regimen with -

In Vitro Susceptibilities of Escherichia Coli and Klebsiella Spp. To
Jpn. J. Infect. Dis., 60, 227-229, 2007 Short Communication In Vitro Susceptibilities of Escherichia coli and Klebsiella Spp. to Ampicillin-Sulbactam and Amoxicillin-Clavulanic Acid Birgul Kacmaz* and Nedim Sultan1 Department of Central Microbiology and 1Department of Microbiology, Faculty of Medicine, Gazi University, Ankara, Turkey (Received January 30, 2007. Accepted April 13, 2007) SUMMARY: Ampicillin-sulbactam (A/S) and amoxicillin-clavulanic acid (AUG) are thought to be equally efficacious clinically against the Enterobacteriaceae family. In this study, the in vitro activities of the A/S and AUG were evaluated and compared against Escherichia coli and Klebsiella spp. Antimicrobial susceptibility tests were performed by standard agar dilution and disc diffusion techniques according to the Clinical and Laboratory Standards Institute (CLSI). During the study period, 973 strains were isolated. Of the 973 bacteria isolated, 823 were E. coli and 150 Klebsiella spp. More organisms were found to be susceptible to AUG than A/S, regardless of the susceptibility testing methodology. The agar dilution results of the isolates that were found to be sensitive or resistant were also compatible with the disc diffusion results. However, some differences were seen in the agar dilution results of some isolates that were found to be intermediately resistant with disc diffusion. In E. coli isolates, 17 of the 76 AUG intermediately resistant isolates (by disc diffusion), and 17 of the 63 A/S intermediately resistant isolates (by disc diffusion) showed different resistant patterns by agar dilution. When the CLSI breakpoint criteria are applied it should be considered that AUG and A/S sensitivity in E. coli and Klebsiella spp.