Certificate of Analysis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
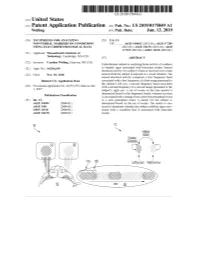
ANNNNNNNNNNNNNNNNNNNN 100A 006 Left Eye Input Right Eye Input
US 20190175049A1 ( 19) United States (12 ) Patent Application Publication (10 ) Pub. No. : US 2019 /0175049 A1 Welling ( 43 ) Pub . Date : Jun . 13 , 2019 ( 54 ) TECHNIQUES FOR ANALYZING (52 ) U . S . CI. NON -VERBAL MARKERS OF CONDITIONS CPC . .. A61B 5 /04842 (2013 . 01 ) ; A61B 5 / 7289 USING ELECTROPHYSIOLOGICAL DATA (2013 . 01) ; A61B 5 /0478 ( 2013 .01 ) ; A61B 5 /7225 ( 2013. 01 ) ; G06N 20 / 10 (2019 .01 ) (71 ) Applicant: Massachusetts Institute of Technology , Cambridge , MA (US ) ( 57 ) ABSTRACT (72 ) Inventor : Caroline Welling, Hanover, NH (US ) Embodiments related to analyzing brain activity of a subject to identify signs associated with binocular rivalry . Sensed ( 21 ) Appl. No. : 16 / 206, 639 electrical activity of a subject' s brain is received over a time period while the subject is exposed to a visual stimulus. The ( 22 ) Filed : Nov. 30 , 2018 sensed electrical activity comprises a first frequency band Related U . S . Application Data associated with a first frequency of a first image presented to the subject ' s left eye , a second frequency band associated (60 ) Provisional application No .62 / 593 , 535, filed on Dec . with a second frequency of a second image presented to the 1 , 2017 subject ' s right eye . A set of events in the time period is determined based on the frequency bands, wherein an event Publication Classification is associated with a change from a previous perceptual event (51 ) Int. Ci. to a new perceptual event. A metric for the subject is A61B 5 /0484 ( 2006 .01 ) determined based on the set of events . The metric is ana A61B 5 /00 ( 2006 .01 ) lyzed to determine whether the subject exhibits signs asso GO6N 20 / 10 (2006 .01 ) ciated with a condition that is associated with binocular A61B 5 /0478 ( 2006 .01 ) rivalry . -

Campro Catalog Stable Isotope
Introduction & Welcome Dear Valued Customer, We are pleased to present to you our Stable Isotopes Catalog which contains more than three thousand (3000) high quality labeled compounds. You will find new additions that are beneficial for your research. Campro Scientific is proud to work together with Isotec, Inc. for the distribution and marketing of their stable isotopes. We have been working with Isotec for more than twenty years and know that their products meet the highest standard. Campro Scientific was founded in 1981 and we provide services to some of the most prestigious universities, research institutes and laboratories throughout Europe. We are a research-oriented company specialized in supporting the requirements of the scientific community. We are the exclusive distributor of some of the world’s leading producers of research chemicals, radioisotopes, stable isotopes and environmental standards. We understand the requirements of our customers, and work every day to fulfill them. In working with us you are guaranteed to receive: - Excellent customer service - High quality products - Dependable service - Efficient distribution The highly educated staff at Campro’s headquarters and sales office is ready to assist you with your questions and product requirements. Feel free to call us at any time. Sincerely, Dr. Ahmad Rajabi General Manager 180/280 = unlabeled 185/285 = 15N labeled 181/281 = double labeled (13C+15N, 13C+D, 15N+18O etc.) 186/286 = 12C labeled 182/282 = d labeled 187/287 = 17O labeled 183/283 = 13C labeleld 188/288 = 18O labeled 184/284 = 16O labeled, 14N labeled 189/289 = Noble Gases Table of Contents Ordering Information.................................................................................................. page 4 - 5 Packaging Information .............................................................................................. -

5994392 Tion of Application No. 67375.734 Eb3-1685, PEN. T
USOO5994392A United States Patent (19) 11 Patent Number: 5,994,392 Shashoua (45) Date of Patent: Nov.30, 1999 54 ANTIPSYCHOTIC PRODRUGS COMPRISING 5,120,760 6/1992 Horrobin ................................. 514/458 AN ANTIPSYCHOTICAGENT COUPLED TO 5,141,958 8/1992 Crozier-Willi et al. ................ 514/558 AN UNSATURATED FATTY ACID 5,216,023 6/1993 Literati et al. .......................... 514/538 5,246,726 9/1993 Horrobin et al. ....................... 424/646 5,516,800 5/1996 Horrobin et al. ....................... 514/560 75 Inventor: Victor E. Shashoua, Brookline, Mass. 5,580,556 12/1996 Horrobin ................................ 424/85.4 73 Assignee: Neuromedica, Inc., Conshohocken, Pa. FOREIGN PATENT DOCUMENTS 30009 6/1981 European Pat. Off.. 21 Appl. No.: 08/462,820 009 1694 10/1983 European Pat. Off.. 22 Filed: Jun. 5, 1995 09 1694 10/1983 European Pat. Off.. 91694 10/1983 European Pat. Off.. Related U.S. Application Data 59-025327 2/1984 Japan. 1153629 6/1989 Japan. 63 Continuation of application No. 08/080,675, Jun. 21, 1993, 1203331 8/1989 Japan. abandoned, which is a continuation of application No. 07/952,191, Sep. 28, 1992, abandoned, which is a continu- (List continued on next page.) ation of application No. 07/577,329, Sep. 4, 1990, aban doned, which is a continuation-in-part of application No. OTHER PUBLICATIONS 07/535,812,tion of application Jun. 11, No. 1990, 67,375.734 abandoned, Eb3-1685, which is a continu-PEN. T. Higuchi et al. 66 Prodrugs as Noye Drug Delivery Sys 4,933,324, which is a continuation-in-part of application No. -

O'neill2020 Redacted.Pdf (7.617Mb)
This thesis has been submitted in fulfilment of the requirements for a postgraduate degree (e.g. PhD, MPhil, DClinPsychol) at the University of Edinburgh. Please note the following terms and conditions of use: This work is protected by copyright and other intellectual property rights, which are retained by the thesis author, unless otherwise stated. A copy can be downloaded for personal non-commercial research or study, without prior permission or charge. This thesis cannot be reproduced or quoted extensively from without first obtaining permission in writing from the author. The content must not be changed in any way or sold commercially in any format or medium without the formal permission of the author. When referring to this work, full bibliographic details including the author, title, awarding institution and date of the thesis must be given. Functional Characterisation of Spontaneously Active GABAA Receptors in Rat Dentate Gyrus Granule Cells Nathanael O’Neill B.Medsc. (Hons) Doctor of Philosophy The University of Edinburgh 2020 ii Abstract GABAA receptors (GABAARs) are the principal inhibitory neurotransmitter receptors in the adult mammalian central nervous system. GABAARs mediate two forms of inhibition: fast, phasic conductance; and slow, tonic conductance. Tonic conductance arises due to the persistent activation of GABAARs. This persistent activation can occur by GABA-dependent or GABA- independent mechanisms. Low concentrations of ambient GABA activate high affinity GABAARs located outside the synapse – at peri-/extra-synaptic sites – to generate GABA-dependent tonic conductance. In contrast, GABA-independent tonic conductance is generated by GABAARs that activate spontaneously, in the absence of GABA, due to constitutive receptor gating. -

The Gabaa Receptor 8 Subunit Gene Promoter Fragments to Direct Long - Term Neuron - Specific Expression
THE GABAa RECEPTOR 8 SUBUNIT GENE PROMOTER : CHARACTERISATION AND USE Alexa Brett Roberts Division of Molecular Genetics Institute of Biomedical and Life Sciences University of Glasgow Glasgow A dissertation submitted for the degree of Doctor of Philosophy of the University of Glasgow February 1998 ProQuest Number: 13818612 All rights reserved INFORMATION TO ALL USERS The quality of this reproduction is dependent upon the quality of the copy submitted. In the unlikely event that the author did not send a com plete manuscript and there are missing pages, these will be noted. Also, if material had to be removed, a note will indicate the deletion. uest ProQuest 13818612 Published by ProQuest LLC(2018). Copyright of the Dissertation is held by the Author. All rights reserved. This work is protected against unauthorized copying under Title 17, United States C ode Microform Edition © ProQuest LLC. ProQuest LLC. 789 East Eisenhower Parkway P.O. Box 1346 Ann Arbor, Ml 48106- 1346 GIASGOW UNIVERSITY LIBRARY 11110 (coh O f GLASGOW 1 IUNIVERSITT I [LQgAW I For my husband Stuart and for my children Ross and Ailie with love Also to JR (Grandad) in loving memory Preface The work presented in this thesis was performed entirely by the author, except where acknowledged. I declare that my thesis contains unique work and will not be submitted for any other degree, diploma or qualification at any other University. A. Brett Roberts December 1997 Acknowledgements I would like to thank Professor R.W. Davies for giving me the opportunity to study for this degree and for his help throughout the last three years. -

Tonic Gabaergic Inhibition As a New Way to Regulate Mesolimbic Dopamine System
Tonic GABAergic inhibition as a new way to regulate mesolimbic dopamine system Elena Vashchinkina Institute of Biomedicine, Pharmacology University of Helsinki Academic Dissertation To be presented with the permission of the Medical Faculty of the University of Helsinki, for public examination in lecture hall 3, Biomedicum Helsinki 1, Haartmaninkatu 8, on December 13th 2013 at 12 noon Helsinki 2013 Supervisor Professor Esa R. Korpi, MD PhD Institute of Biomedicine, Pharmacology Faculty of Medicine University of Helsinki, Finland Reviewers Docent Mikko Airavaara, PhD Institute of Biotechnology University of Helsinki, Finland Docent Tarja Stenberg, MD PhD Institute of Biomedicine, Physiology University of Helsinki, Finland Dissertation Opponent Professor Kimmo Jensen, MD PhD Department of Biomedicine Aarhus University, Denmark ISBN 978-952-10-9636-5 (paperback) ISBN 978-952-10-9637-2 (PDF) http://ethesis.helsinki.fi Unigrafia OY Helsinki 2013 TABLE OF CONTENTS Abstract ......................................................................................................................... 1 Original publications ................................................................................................... 2 Abbreviations ............................................................................................................... 3 Glossary of terms ......................................................................................................... 4 1 INTRODUCTION .................................................................................................... -

Neuroscience Products
Neuroscience Products CATALOG CATALOG NUMBER U.S. $ NUMBER U.S. $ -A- 3-(N-ACETYLAMINO)-5-(N-DECYL-N- 1 mg 27.50 159549 METHYLAMINO)BENZYL ALCOHOL 5 mg 89.40 o A23187 0-5 C [103955-90-4] (ADMB) See: Antibiotic A23187 A Protein Kinase C activator. Ref.: Proc. Nat. Acad. Sci. USA, 83, 4214 AA-861 20 mg 72.70 (1986). 159061 Purity: 95% 100 mg 326.40 C20H34N2O2 MW 334.5 0oC Orally active, specific and potent inhibitor of 5-lipoxygenase. N-ACETYL-ASP-GLU 25 mg 45.00 153036 [3106-85-2] 100 mg 156.00 Ref.: 1. Yoshimoto, T., et.al., Biochim. o Biophys. Acta, 713, 470 (1982). 2. Ashida, -20-0 C An endogenous neuropeptide with high 250 mg 303.65 Y., et.al., Prostaglandins, 26, 955 (1983). 3. affinity for a brain "Glutamate" receptor. Ancill, R.J., et.al., J. Int. Med. Res., 18, 75 Ref: Zaczek, R., et al., Proc. Natl. Acad. (1990). Sci. (USA), 80, 1116 (1983). C21H26O3 MW 326.4 C11H16N2O8 MW 304.3 ABL PROTEIN TYROSINE KINASE 250 U 47.25 N-ACETYL-2-BENZYLTRYPTAMINE 195876 (v-abl) 1 KU 162.75 See: Luzindole -70oC Recombinant Expressed in E. coli ACETYL-DL-CARNITINE 250 mg 60.00 A truncated form of the v-abl protein 154690 [2504-11-2] 1 g 214.00 tyrosine kinase which contains the 0oC Hydrochloride minimum region needed for kinase activity Crystalline and fibroblast transformation. Suppresses C9H17NO4 • HCl MW 239.7 apoptosis and induces resistance to anti-cancer compounds. O-ACETYL-L-CARNITINE CHLORIDE 500 mg 11.45 Activity: 100 KU/ml 159062 [5080-50-2] 1 g 20.65 Unit Definition: one unit is the amount of 0-5oC (R-(-)-2-Acetyloxy-3-carboxy-N,N,N-trimethyl 5 g 97.45 enzyme which catalyzes the transfer of 1 -1-propanaminium chloride) pmol of phosphate to EAIYAAPFAKKK per Purity: >88% minute at 30°C, pH 7.5. -

David R. Curtis 171
EDITORIAL ADVISORY COMMITTEE Giovanni Berlucchi Mary B. Bunge Robert E. Burke Larry E Cahill Stanley Finger Bernice Grafstein Russell A. Johnson Ronald W. Oppenheim Thomas A. Woolsey (Chairperson) The History of Neuroscience in" Autob~ograp" by VOLUME 5 Edited by Larry R. Squire AMSTERDAM 9BOSTON 9HEIDELBERG 9LONDON NEW YORK 9OXFORD ~ PARIS 9SAN DIEGO SAN FRANCISCO 9SINGAPORE 9SYDNEY 9TOKYO ELSEVIER Academic Press is an imprint of Elsevier Elsevier Academic Press 30 Corporate Drive, Suite 400, Burlington, Massachusetts 01803, USA 525 B Street, Suite 1900, San Diego, California 92101-4495, USA 84 Theobald's Road, London WC1X 8RR, UK This book is printed on acid-free paper. O Copyright 92006 by the Society for Neuroscience. All rights reserved. No part of this publication may be reproduced or transmitted in any form or by any means, electronic or mechanical, including photocopy, recording, or any information storage and retrieval system, without permission in writing from the publisher. Permissions may be sought directly from Elsevier's Science & Technology Rights Department in Oxford, UK: phone: (+44) 1865 843830, fax: (+44) 1865 853333, E-mail: [email protected]. You may also complete your request on-line via the Elsevier homepage (http://elsevier.com), by selecting "Support & Contact" then "Copyright and Permission" and then "Obtaining Permissions." Library of Congress Catalog Card Number: 2003 111249 British Library Cataloguing in Publication Data A catalogue record for this book is available from the British Library ISBN 13:978-0-12-370514-3 ISBN 10:0-12-370514-2 For all information on all Elsevier Academic Press publications visit our Web site at www.books.elsevier.com Printed in the United States of America 06 07 08 09 10 11 9 8 7 6 5 4 3 2 1 Working together to grow libraries in developing countries www.elsevier.com ] ww.bookaid.org ] www.sabre.org ER BOOK AID ,~StbFC" " " =LSEVI lnt ..... -

Design and Syntheses of Potential Drugs Based on GABAA Receptor Pharmacophores
Design and Syntheses of Potential Drugs Based on GABAA Receptor Pharmacophores Ella Chow Clement Dissertation submitted to the Faculty of the Virginia Polytechnic Institute and State University in partial fulfillment of the requirements for the Degree of Doctor of Philosopy in Chemistry Dr. Paul R. Carlier, Chairman Dr. Jeffrey R. Bloomquist Dr. Richard D. Gandour Dr. David G. I. Kingston Dr. James M. Tanko June 28, 2005 Blacksburg, Virginia Keywords: GABAA receptor, Partial/full agonists, Superagonist, Antagonists, Non-zwitterionic GABA amide homodimers and heterodimers, 36Cl- Flux assay, [3H]Muscimol binding, ZAPA, PEG, Memory of Chirality Copyright 2005, Ella Chow Clement Design and Syntheses of Potential Drugs Based on GABAA Receptor Pharmocophores Ella Chow Clement ABSTRACT Numerous previous studies of GABAAR ligands have suggested that GABAAR agonists must be zwitterionic and feature an intercharge separation similar to that of GABA (approx. 4.7-6.0 Å). We have demonstrated that monomeric, homodimeric and heterodimeric non-zwitterionic GABA amides are partial, full, or superagonists at the murine GABAA receptor (GABAAR). The agonism of these GABA amides is comparable to that of THIP, as shown by in vitro assay results. The assay data indicate that the agonism of GABA amides is tether length-dependent. Optimum agonism is achieved with a tether length of four methylenes in GABA amide dimers and in GABA amides bearing pendant amide or amino groups. We have further investigated the structure-activity relationship for GABA amides on the GABAAR by performing structural modifications to both the superagonist 2c and the agonist 6c. Synergism and [3H]muscimol binding experiments show that 2c binds to the same sites as GABA. -

PROPERTIES of R-AMINOBUTYRIC ACID ACTIVATED CHLORIDE CHANNELS in MAMMALIAN NEURONES by Claire Fiona Newland a Thesis Subm Itted
PROPERTIES OF r-AMINOBUTYRIC ACID ACTIVATED CHLORIDE CHANNELS IN MAMMALIAN NEURONES b y Claire Fiona Newland A Thesis submitted for the degree of Doctor of Philosophy at the University of London. Department of Pharmacology University College London Supervisors: Dr. S. G. Cull-Candy & Prof. D. Colquhoun, FRS. 1990 1 ProQuest Number: 10609808 All rights reserved INFORMATION TO ALL USERS The quality of this reproduction is dependent upon the quality of the copy submitted. In the unlikely event that the author did not send a com plete manuscript and there are missing pages, these will be noted. Also, if material had to be removed, a note will indicate the deletion. uest ProQuest 10609808 Published by ProQuest LLC(2017). Copyright of the Dissertation is held by the Author. All rights reserved. This work is protected against unauthorized copying under Title 17, United States C ode Microform Edition © ProQuest LLC. ProQuest LLC. 789 East Eisenhower Parkway P.O. Box 1346 Ann Arbor, Ml 48106- 1346 ABSTRACT GABA a receptor-channels in dissociated rat sympathetic ganglion neurones have been studied by conventional patch clamp techniques, with both whole-cell and outside-out patch configurations* T hese GABA a receptor-channels are shown to be pharmacologically similar to those of mammalian central neurones, being inhibited by bicuculline, picrotoxin, picrotoxinin and penicillin, and potentiated by pentobarbitone. Furthermore, peripheral GABAA-channels resemble central ones in their cur rent-voltage relationship (whole-cell and Bingle-channel) and in the voltage-dependence of their kinetic properties (noise an aly sis). The mechanism of action of the well established GABA a antagonist, picrotoxin, has been further investigated in these neurones. -

(12) Patent Application Publication (10) Pub. No.: US 2010/0184806 A1 Barlow Et Al
US 20100184806A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2010/0184806 A1 Barlow et al. (43) Pub. Date: Jul. 22, 2010 (54) MODULATION OF NEUROGENESIS BY PPAR (60) Provisional application No. 60/826,206, filed on Sep. AGENTS 19, 2006. (75) Inventors: Carrolee Barlow, Del Mar, CA (US); Todd Carter, San Diego, CA Publication Classification (US); Andrew Morse, San Diego, (51) Int. Cl. CA (US); Kai Treuner, San Diego, A6II 3/4433 (2006.01) CA (US); Kym Lorrain, San A6II 3/4439 (2006.01) Diego, CA (US) A6IP 25/00 (2006.01) A6IP 25/28 (2006.01) Correspondence Address: A6IP 25/18 (2006.01) SUGHRUE MION, PLLC A6IP 25/22 (2006.01) 2100 PENNSYLVANIA AVENUE, N.W., SUITE 8OO (52) U.S. Cl. ......................................... 514/337; 514/342 WASHINGTON, DC 20037 (US) (57) ABSTRACT (73) Assignee: BrainCells, Inc., San Diego, CA (US) The instant disclosure describes methods for treating diseases and conditions of the central and peripheral nervous system (21) Appl. No.: 12/690,915 including by stimulating or increasing neurogenesis, neuro proliferation, and/or neurodifferentiation. The disclosure (22) Filed: Jan. 20, 2010 includes compositions and methods based on use of a peroxi some proliferator-activated receptor (PPAR) agent, option Related U.S. Application Data ally in combination with one or more neurogenic agents, to (63) Continuation-in-part of application No. 1 1/857,221, stimulate or increase a neurogenic response and/or to treat a filed on Sep. 18, 2007. nervous system disease or disorder. Patent Application Publication Jul. 22, 2010 Sheet 1 of 9 US 2010/O184806 A1 Figure 1: Human Neurogenesis Assay Ciprofibrate Neuronal Differentiation (TUJ1) 100 8090 Ciprofibrates 10-8.5 10-8.0 10-7.5 10-7.0 10-6.5 10-6.0 10-5.5 10-5.0 10-4.5 Conc(M) Patent Application Publication Jul. -

Potential Therapeutic Approaches for Modulating Expression and Accumulation of Defective Lamin a in Laminopathies and Age-Related Diseases
J Mol Med (2012) 90:1361–1389 DOI 10.1007/s00109-012-0962-4 REVIEW Potential therapeutic approaches for modulating expression and accumulation of defective lamin A in laminopathies and age-related diseases Alex Zhavoronkov & Zeljka Smit-McBride & Kieran J. Guinan & Maria Litovchenko & Alexey Moskalev Received: 6 May 2012 /Revised: 8 September 2012 /Accepted: 25 September 2012 /Published online: 23 October 2012 # The Author(s) 2012. This article is published with open access at Springerlink.com Abstract Scientific understanding of the genetic compo- rise to lethal, early-onset diseases known as laminopathies. nents of aging has increased in recent years, with several Here, we analyze the literature and conduct computational genes being identified as playing roles in the aging process analyses of lamin A signaling and intracellular interactions and, potentially, longevity. In particular, genes encoding in order to examine potential mechanisms for altering or components of the nuclear lamina in eukaryotes have been slowing down aberrant Lamin A expression and/or for re- increasingly well characterized, owing in part to their clin- storing the ratio of normal to aberrant lamin A. The ultimate ical significance in age-related diseases. This review focuses goal of such studies is to ameliorate or combat laminopa- on one such gene, which encodes lamin A, a key component thies and related diseases of aging, and we provide a dis- of the nuclear lamina. Genetic variation in this gene can give cussion of current approaches in this review. A. Zhavoronkov (*) : M. Litovchenko Keywords Lamin A Progeria Laminopathies Bioinformatics and Medical Information Technology Laboratory, Age-related diseases .