Campro Catalog Stable Isotope
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

United States July 2016 2 Table of Contents
Deuterium Labelled Compounds United States July 2016 2 Table of Contents International Distributors 3 Corporate Overview 4 General Information 5 Pricing and Payment 5 Quotations 5 Custom Synthesis 5 Shipping 5 Quality Control 6 Quotations 6 Custom Synthesis 6 Shipping 6 Quality Control 6 Chemical Abstract Service Numbers 6 Handling Hazardous Compounds 6 Our Products are Not Intended for Use in Humans 7 Limited Warranty 7 Packaging Information 7 Alphabetical Listings 8 Stock Clearance 236 Products by Category 242 n-Alkanes 243 α-Amino Acids, N-Acyl α-Amino Acids, N-t-BOC Protected α-Amino Acid 243 and N-FMOC Protected α-Amino Acids Buffers and Reagents for NMR Studies 245 Detergents 245 Environmental Standards 246 Fatty Acids and Fatty Acid Esters 249 Flavours and Fragrances 250 Gases 253 Medical Research Products 254 Nucleic Acid Bases and Nucleosides 255 Pesticides and Pesticide Metabolites 256 Pharmaceutical Standards 257 Polyaromatic Hydrocarbons (PAHs), Alkyl-PAHs, Amino-PAHs, 260 Hydroxy-PAHs and Nitro-PAHs Polychlorinated Biphenyls (PCBs) 260 Spin Labels 261 Steroids 261 3 International Distributors C Beijng Zhenxiang H EQ Laboratories GmbH Australia K Technology Company Graf-von-Seyssel-Str. 10 Rm. 15A01, Changyin Bld. 86199 Augsburg Austria H No. 88, YongDingLu Rd. Germany Beijing 100039 Tel.: (49) 821 71058246 Belgium J China Fax: (49) 821 71058247 Tel.: (86) 10-58896805 [email protected] China C Fax: (86) 10-58896158 www.eqlabs.de Czech Republic H [email protected] Germany, Austria, China Czech Republic, Greece, Denmark I Hungary, -

Prnu-BICALUTAMIDE
PRODUCT MONOGRAPH PrNU-BICALUTAMIDE Bicalutamide Tablets, 50 mg Non-Steroidal Antiandrogen NU-PHARM INC. DATE OF PREPARATION: 50 Mural Street, Units 1 & 2 October 16, 2009 Richmond Hill, Ontario L4B 1E4 Control#: 133521 Page 1 of 27 Table of Contents PART I: HEALTH PROFESSIONAL INFORMATION....................................................... 3 SUMMARY PRODUCT INFORMATION ............................................................................. 3 INDICATIONS AND CLINICAL USE ................................................................................... 3 CONTRAINDICATIONS ........................................................................................................ 3 WARNINGS AND PRECAUTIONS....................................................................................... 4 ADVERSE REACTIONS......................................................................................................... 5 DRUG INTERACTIONS ......................................................................................................... 9 DOSAGE AND ADMINISTRATION ................................................................................... 10 OVERDOSAGE...................................................................................................................... 10 ACTION AND CLINICAL PHARMACOLOGY.................................................................. 10 STORAGE AND STABILITY............................................................................................... 11 DOSAGE FORMS, COMPOSITION AND PACKAGING -

Aldrich FT-IR Collection Edition I Library
Aldrich FT-IR Collection Edition I Library Library Listing – 10,505 spectra This library is the original FT-IR spectral collection from Aldrich. It includes a wide variety of pure chemical compounds found in the Aldrich Handbook of Fine Chemicals. The Aldrich Collection of FT-IR Spectra Edition I library contains spectra of 10,505 pure compounds and is a subset of the Aldrich Collection of FT-IR Spectra Edition II library. All spectra were acquired by Sigma-Aldrich Co. and were processed by Thermo Fisher Scientific. Eight smaller Aldrich Material Specific Sub-Libraries are also available. Aldrich FT-IR Collection Edition I Index Compound Name Index Compound Name 3515 ((1R)-(ENDO,ANTI))-(+)-3- 928 (+)-LIMONENE OXIDE, 97%, BROMOCAMPHOR-8- SULFONIC MIXTURE OF CIS AND TRANS ACID, AMMONIUM SALT 209 (+)-LONGIFOLENE, 98+% 1708 ((1R)-ENDO)-(+)-3- 2283 (+)-MURAMIC ACID HYDRATE, BROMOCAMPHOR, 98% 98% 3516 ((1S)-(ENDO,ANTI))-(-)-3- 2966 (+)-N,N'- BROMOCAMPHOR-8- SULFONIC DIALLYLTARTARDIAMIDE, 99+% ACID, AMMONIUM SALT 2976 (+)-N-ACETYLMURAMIC ACID, 644 ((1S)-ENDO)-(-)-BORNEOL, 99% 97% 9587 (+)-11ALPHA-HYDROXY-17ALPHA- 965 (+)-NOE-LACTOL DIMER, 99+% METHYLTESTOSTERONE 5127 (+)-P-BROMOTETRAMISOLE 9590 (+)-11ALPHA- OXALATE, 99% HYDROXYPROGESTERONE, 95% 661 (+)-P-MENTH-1-EN-9-OL, 97%, 9588 (+)-17-METHYLTESTOSTERONE, MIXTURE OF ISOMERS 99% 730 (+)-PERSEITOL 8681 (+)-2'-DEOXYURIDINE, 99+% 7913 (+)-PILOCARPINE 7591 (+)-2,3-O-ISOPROPYLIDENE-2,3- HYDROCHLORIDE, 99% DIHYDROXY- 1,4- 5844 (+)-RUTIN HYDRATE, 95% BIS(DIPHENYLPHOSPHINO)BUT 9571 (+)-STIGMASTANOL -

(12) Patent Application Publication (10) Pub. No.: US 2009/0156618 A1 Tollefson (43) Pub
US 20090156618A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2009/0156618 A1 Tollefson (43) Pub. Date: Jun. 18, 2009 (54) 1-(1-(2-ETHOXYETHYL)-3-ETHYL-7- Related U.S. Application Data (4-METHYLPYRIDIN-2-YLAMINO) - 1H-PYRAZOLO 4.3-D PYRIMIDIN-5-YL) (60) Provisional application No. 60/735,320, filed on Nov. PPERDINE-4-CARBOXYLIC ACID AND 10, 2005. SALTS THEREOF Publication Classification (51) Int. Cl. (75) Inventor: Michael B. Tollefson, Dardenne A 6LX 3/59 (2006.01) Prairie, MO (US) C07D 487/04 (2006.01) A6IP 9/12 (2006.01) A6IP 25/00 (2006.01) Correspondence Address: A6IP 9/00 (2006.01) PFZER INC. PATENT DEPARTMENT, Bld 114 M/S 114, (52) U.S. Cl. ...................................... 514/262.1; 54.4/262 EASTERN PONT ROAD (57) ABSTRACT GROTON, CT 06340 (US) The present invention comprises 1-(1-(2-ethoxyethyl)-3- ethyl-7-(4-methylpyridin-2-ylamino)-1H-pyrazolo 4.3-d (73) Assignee: Pfizer Inc pyrimidin-5-yl)piperidine-4-carboxylic acid and its salts. The invention further comprises pharmaceutical composi tions, methods of treatment, and synthetic methods relating to (21) Appl. No.: 111558,306 1-(1-(2-ethoxyethyl)-3-ethyl-7-(4-methylpyridin-2- ylamino)-1H-pyrazolo 4.3-dipyrimidin-5-yl)piperidine-4- (22) Filed: Nov. 9, 2006 carboxylic acid and its salts. Patent Application Publication Jun. 18, 2009 Sheet 2 of 2 US 2009/0156618 A1 "SOIHZ US 2009/0156618 A1 Jun. 18, 2009 1-(1-(2-ETHOXYETHYL)-3-ETHYL-7- 1H-pyrazolo 4,3-dipyrimidin-5-yl)piperidine-4-carboxylic (4-METHYLPYRIDIN-2-YLAMINO) - acid and its pharmaceutically acceptable salts. -
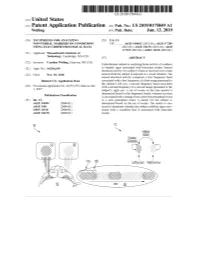
ANNNNNNNNNNNNNNNNNNNN 100A 006 Left Eye Input Right Eye Input
US 20190175049A1 ( 19) United States (12 ) Patent Application Publication (10 ) Pub. No. : US 2019 /0175049 A1 Welling ( 43 ) Pub . Date : Jun . 13 , 2019 ( 54 ) TECHNIQUES FOR ANALYZING (52 ) U . S . CI. NON -VERBAL MARKERS OF CONDITIONS CPC . .. A61B 5 /04842 (2013 . 01 ) ; A61B 5 / 7289 USING ELECTROPHYSIOLOGICAL DATA (2013 . 01) ; A61B 5 /0478 ( 2013 .01 ) ; A61B 5 /7225 ( 2013. 01 ) ; G06N 20 / 10 (2019 .01 ) (71 ) Applicant: Massachusetts Institute of Technology , Cambridge , MA (US ) ( 57 ) ABSTRACT (72 ) Inventor : Caroline Welling, Hanover, NH (US ) Embodiments related to analyzing brain activity of a subject to identify signs associated with binocular rivalry . Sensed ( 21 ) Appl. No. : 16 / 206, 639 electrical activity of a subject' s brain is received over a time period while the subject is exposed to a visual stimulus. The ( 22 ) Filed : Nov. 30 , 2018 sensed electrical activity comprises a first frequency band Related U . S . Application Data associated with a first frequency of a first image presented to the subject ' s left eye , a second frequency band associated (60 ) Provisional application No .62 / 593 , 535, filed on Dec . with a second frequency of a second image presented to the 1 , 2017 subject ' s right eye . A set of events in the time period is determined based on the frequency bands, wherein an event Publication Classification is associated with a change from a previous perceptual event (51 ) Int. Ci. to a new perceptual event. A metric for the subject is A61B 5 /0484 ( 2006 .01 ) determined based on the set of events . The metric is ana A61B 5 /00 ( 2006 .01 ) lyzed to determine whether the subject exhibits signs asso GO6N 20 / 10 (2006 .01 ) ciated with a condition that is associated with binocular A61B 5 /0478 ( 2006 .01 ) rivalry . -

(12) Patent Application Publication (10) Pub. No.: US 2007/0254920 A1 Delong Et Al
US 20070254920A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2007/0254920 A1 deLong et al. (43) Pub. Date: Nov. 1, 2007 (54) PRODRUG DERIVATIVES OF ACIDS USING (52) U.S. Cl. ......................... 514/319; 514/323: 514/573; ALCOHOLS WITH HOMOTOPIC HYDROXY 514/415: 514/443; 514/469; GROUPS AND METHODS FOR THEIR 546/196; 546/201: 546/206; PREPARATION AND USE 548/495; 549/49; 549/462 (75) Inventors: Mitchell A. deLong, Raleigh, NC (US); (57) ABSTRACT Jill M. McFadden, Chapel Hill, NC This invention relates to novel homotopic prodrugs and (US); Susan M. Royalty, Cary, NC medicaments and methods for their preparation, testing and (US); Eric J. Toone, Durham, NC use. In one embodiment, the homotopic prodrug has the (US); Jeffrey D. Yingling, Apex, NC general formula (US) Correspondence Address: O MICHAEL BEST & FRIEDRCH LLP 100 E WISCONSNAVENUE Rb, Suite 3300 --- MILWAUKEE, WI 53202 (US) wherein (73) Assignee: Aerie Pharmaceuticals, Inc., Research Triangle Park, NC (21) Appl. No.: 11/412,207 (22) Filed: Apr. 26, 2006 -- Publication Classification is a biologically-active moiety comprising a carboxylic acid functional group, and R is a homotopically-symmetrical (51) Int. Cl. alcohol bonded to the biologically-active moiety through the A6 IK 3/558 (2006.01) carboxylic acid functional group to form an ester linkage, as CO7D 409/02 (2006.01) well as optical isomers, enantiomers, pharmaceutically CO7D 405/02 (2006.01) acceptable salts, biohydrolyzable amides, esters, and imides CO7D 403/02 (2006.01) thereof and combinations thereof. US 2007/0254920 A1 Nov. 1, 2007 PRODRUG DERVATIVES OF ACDS USING WO 99/12896, and WO 99/12898.) However, such modifi ALCOHOLS WITH HOMOTOPIC HYDROXY cations have either resulted in only modest increases in GROUPS AND METHODS FOR THEIR half-life or resulted in compounds with diminished potency. -

European Journal of Chemistry 5 (4) (2014) 681‐694
European Journal of Chemistry 5 (4) (2014) 681‐694 European Journal of Chemistry Journal homepage: www.eurjchem.com Synthesis, reactions and applications of pyranotriazolopyrimidines Ashraf Hassan Fekry Abd El‐Wahab a,b,*, Ibrahim Ali Radini a and Hany Mostafa Mohamed a,b a Chemistry Department, Faculty of Science, Jazan University, 2097, Jazan, Saudi Arabia b Chemistry Department, Faculty of Science, Al‐Azhar University, 11884, Nasr City, Cairo, Egypt *Corresponding author at: Chemistry Department, Faculty of Science, Jazan University, 2097, Jazan, Saudi Arabia. Tel.: +966.054.0963753. Fax: +966.017.3230028. E‐mail address: [email protected] (A.H.F.A. El‐Wahab). REVIEW INFORMATION ABSTRACT This review deals with synthesis, reactions and their applications of pyranotriazolo‐ pyrimidines. The main purpose of this review is present a survey of literatures on the reactivity of amino imino derivatives and carboxylic acid derivatives. Some of these reactions have been applied successfully to the synthesis of biological important compounds. DOI: 10.5155/eurjchem.5.4.681‐694.1087 Received: 30 April 2014 Received in revised form: 27 May 2014 Accepted: 27 May 2014 Online: 31 December 2014 KEYWORDS Naphthols Pyrimidine Biological activity Pyranopyrimidines α‐Cyanocinnamonitriles Carboxylic acid derivatives 1. Introduction chemical and biological view points, due to their diverse pharmacological activities, such as antitumor potency [19,20], Pyran derivatives have attracted a great deal of interest inhibition of KDR kinase [21], antifungal effect [22] and owing to their antimicrobial activity [1‐7], inhibition of influ‐ macrophage activation [23]. enza, virus sialidase [8], mutagenic activity [9], activity as antiviral [10], anti‐proliferaction agents [11], sex pheromones [12], antitumor [13] and anti‐inflammatory agents [14]. -

Hypothalamic Principles Necessary for the Release of Ovulating Hormone from the Adenohypophysis
EFFECT OF AMINOGLUTETHIMIDE ON REPRODUCTIVE PROCESSES IN FEMALE RATS W. J. EVERSOLE and D. J. THOMPSON Department of Life Sciences, Indiana State University, Terre Haute, Indiana 47809, U.S.A. (Received "òlst January 1974) Summary. Adult female rats injected subcutaneously with 100 mg aminoglutethimide phosphate (AGP)/kg/day for 4 weeks failed to become pregnant when placed in cohabitation with males during the last 2 weeks of the injection period. Lower doses depressed the fertility rate and reduced the litter size: the average litter size of three of eleven rats given 50 mg/kg/day was 4\m=.\7 young and of four of eleven rats given 25 mg was 7\m=.\0 young. Twenty-nine controls averaged 10\m=.\1/litter. Doses of 100 mg AGP/kg/day stopped vaginal cycling, prevented ovulation in adults, and delayed dissolution of the vaginal membrane in pubertal rats. Histological studies of the ovaries from rats with initial ages of 17 or 21 days which were injected with 25 to 100 mg AGP/kg/day for 2 weeks showed an increase in the number and size of vesicular follicles. Treatment of 27-day-old rats with 25 mg/kg/day for 2 weeks reduced the number of CL and three of four rats given 50 mg had ovaries lacking such bodies; all control ovaries in this group contained CL. These findings are taken as evidence that AGP inhibits ovulation but does not prevent follicular maturation. INTRODUCTION Many pharmacological compounds originally developed as anaesthetics, tranquillizers and anticonvulsants modify endocrine structures and functions (see Gaunt, Chart & Renzi, 1965). -

Mechanisms of Action of Antiepileptic Drugs
Review Mechanisms of action of antiepileptic drugs Epilepsy affects up to 1% of the general population and causes substantial disability. The management of seizures in patients with epilepsy relies heavily on antiepileptic drugs (AEDs). Phenobarbital, phenytoin, carbamazepine and valproic acid have been the primary medications used to treat epilepsy for several decades. Since 1993 several AEDs have been approved by the US FDA for use in epilepsy. The choice of the AED is based primarily on the seizure type, spectrum of clinical activity, side effect profile and patient characteristics such as age, comorbidities and concurrent medical treatments. Those AEDs with broad- spectrum activity are often found to exert an action at more than one molecular target. This article will review the proposed mechanisms of action of marketed AEDs in the US and discuss the future of AEDs in development. 1 KEYWORDS: AEDs anticonvulsant drugs antiepileptic drugs epilepsy Aaron M Cook mechanism of action seizures & Meriem K Bensalem-Owen† The therapeutic armamentarium for the treat- patients with refractory seizures. The aim of this 1UK HealthCare, 800 Rose St. H-109, ment of seizures has broadened significantly article is to discuss the past, present and future of Lexington, KY 40536-0293, USA †Author for correspondence: over the past decade [1]. Many of the newer AED pharmacology and mechanisms of action. College of Medicine, Department of anti epileptic drugs (AEDs) have clinical advan- Neurology, University of Kentucky, 800 Rose Street, Room L-455, tages over older, so-called ‘first-generation’ First-generation AEDs Lexington, KY 40536, USA AEDs in that they are more predictable in their Broadly, the mechanisms of action of AEDs can Tel.: +1 859 323 0229 Fax: +1 859 323 5943 dose–response profile and typically are associ- be categorized by their effects on the neuronal [email protected] ated with less drug–drug interactions. -

WO 2018/190970 Al 18 October 2018 (18.10.2018) W !P O PCT
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2018/190970 Al 18 October 2018 (18.10.2018) W !P O PCT (51) International Patent Classification: GM, KE, LR, LS, MW, MZ, NA, RW, SD, SL, ST, SZ, TZ, CI2Q 1/32 (2006.01) UG, ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, RU, TJ, TM), European (AL, AT, BE, BG, CH, CY, CZ, DE, DK, (21) International Application Number: EE, ES, FI, FR, GB, GR, HR, HU, IE, IS, IT, LT, LU, LV, PCT/US2018/021 109 MC, MK, MT, NL, NO, PL, PT, RO, RS, SE, SI, SK, SM, (22) International Filing Date: TR), OAPI (BF, BJ, CF, CG, CI, CM, GA, GN, GQ, GW, 06 March 2018 (06.03.2018) KM, ML, MR, NE, SN, TD, TG). (25) Filing Language: English Declarations under Rule 4.17: (26) Publication Langi English — as to applicant's entitlement to apply for and be granted a patent (Rule 4.1 7(H)) (30) Priority Data: — as to the applicant's entitlement to claim the priority of the 62/484,141 11 April 2017 ( 11.04.2017) US earlier application (Rule 4.17(Hi)) (71) Applicant: REGENERON PHARMACEUTICALS, Published: INC. [US/US]; 777 Old Saw Mill River Road, Tarrytown, — with international search report (Art. 21(3)) New York 10591-6707 (US). — with sequence listing part of description (Rule 5.2(a)) (72) Inventors: STEVIS, Panayiotis; 777 Old Saw Mill Riv er Road, Tarrytown, New York 10591-6707 (US). -

5994392 Tion of Application No. 67375.734 Eb3-1685, PEN. T
USOO5994392A United States Patent (19) 11 Patent Number: 5,994,392 Shashoua (45) Date of Patent: Nov.30, 1999 54 ANTIPSYCHOTIC PRODRUGS COMPRISING 5,120,760 6/1992 Horrobin ................................. 514/458 AN ANTIPSYCHOTICAGENT COUPLED TO 5,141,958 8/1992 Crozier-Willi et al. ................ 514/558 AN UNSATURATED FATTY ACID 5,216,023 6/1993 Literati et al. .......................... 514/538 5,246,726 9/1993 Horrobin et al. ....................... 424/646 5,516,800 5/1996 Horrobin et al. ....................... 514/560 75 Inventor: Victor E. Shashoua, Brookline, Mass. 5,580,556 12/1996 Horrobin ................................ 424/85.4 73 Assignee: Neuromedica, Inc., Conshohocken, Pa. FOREIGN PATENT DOCUMENTS 30009 6/1981 European Pat. Off.. 21 Appl. No.: 08/462,820 009 1694 10/1983 European Pat. Off.. 22 Filed: Jun. 5, 1995 09 1694 10/1983 European Pat. Off.. 91694 10/1983 European Pat. Off.. Related U.S. Application Data 59-025327 2/1984 Japan. 1153629 6/1989 Japan. 63 Continuation of application No. 08/080,675, Jun. 21, 1993, 1203331 8/1989 Japan. abandoned, which is a continuation of application No. 07/952,191, Sep. 28, 1992, abandoned, which is a continu- (List continued on next page.) ation of application No. 07/577,329, Sep. 4, 1990, aban doned, which is a continuation-in-part of application No. OTHER PUBLICATIONS 07/535,812,tion of application Jun. 11, No. 1990, 67,375.734 abandoned, Eb3-1685, which is a continu-PEN. T. Higuchi et al. 66 Prodrugs as Noye Drug Delivery Sys 4,933,324, which is a continuation-in-part of application No. -

Supraphysiological Doses of Performance Enhancing Anabolic-Androgenic Steroids Exert Direct Toxic Effects on Neuron-Like Cells
ORIGINAL RESEARCH ARTICLE published: 09 May 2013 CELLULAR NEUROSCIENCE doi: 10.3389/fncel.2013.00069 Supraphysiological doses of performance enhancing anabolic-androgenic steroids exert direct toxic effects on neuron-like cells John R. Basile1,2, Nada O. Binmadi 1,3, Hua Zhou1, Ying-Hua Yang1, Antonio Paoli 4 and Patrizia Proia1,5* 1 Department of Oncology and Diagnostic Sciences, University of Maryland Dental School, Baltimore, MD, USA 2 Marlene and Stuart Greenebaum Cancer Center, University of Maryland, Baltimore, MD, USA 3 Department of Oral Basic and Clinical Sciences, King Abdulaziz University, Jeddah, Saudi Arabia 4 Department of Biomedical Sciences, University of Padova, Padova, Italy 5 Department of Sports Science (DISMOT), University of Palermo, Palermo, Italy Edited by: Anabolic-androgenic steroids (AAS) are lipophilic hormones often taken in excessive Chao Deng, University of quantities by athletes and bodybuilders to enhance performance and increase muscle Wollongong, Australia mass. AAS exert well known toxic effects on specific cell and tissue types and organ Reviewed by: systems. The attention that androgen abuse has received lately should be used as an Agata Copani, University of Catania, Italy opportunity to educate both athletes and the general population regarding their adverse Aram Megighian, University of effects. Among numerous commercially available steroid hormones, very few have been Padua, Italy specifically tested for direct neurotoxicity. We evaluated the effects of supraphysiological *Correspondence: doses of methandienone and 17-α-methyltestosterone on sympathetic-like neuron cells. Patrizia Proia, Department of Sports Vitality and apoptotic effects were analyzed, and immunofluorescence staining and Science (DISMOT), University of Palermo, Via Eleonora Duse 2, western blot performed.