"The Science for Diplomats" Annex on Chemicals
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Theoretical Study of Sarin Adsorption On
Chemical Physics Letters 738 (2020) 136816 Contents lists available at ScienceDirect Chemical Physics Letters journal homepage: www.elsevier.com/locate/cplett Research paper Theoretical study of sarin adsorption on (12,0) boron nitride nanotube doped with silicon atoms T ⁎ ⁎ Jeziel Rodrigues dos Santosa, , Elson Longo da Silvab, Osmair Vital de Oliveirac, , José Divino dos Santosa a Universidade Estadual de Goiás, Campus Anápolis, CEP: 75.132-903 GO, Brazil b INCTMN, LIEC, Departamento de Química da Universidade Federal de São Carlos, CEP: 13.565-905 São Carlos, SP, Brazil c Instituto Federal de Educação, Ciência e Tecnologia de São Paulo, Campus Catanduva, CEP: 15.808-305 Catanduva, SP, Brazil HIGHLIGHTS • DFT method was used to study the adsorption of nerve agent sarin by BNNT. • Electronic properties of pristine BNNT are improved by Si impurity atoms. • The adsorption of sarin by Si-doped BNNT is highest favorable than the pure BNNT. • Si-doped BNNT can be a new gas sensor for sarin gas detection and its derivatives. ARTICLE INFO ABSTRACT Keywords: Sarin gas is one of the most lethal nerve agent used in chemical warfare, which its detection is import to prevent Nerve agent sarin a chemical attack and to identify a contamination area. Herein, density functional theory was used to investigate Gas sensor the (12,0) boron nitride nanotube (BNNT) and Si–doped BNNT as possible candidates to sarin detection. The Si- Boron nitride nanotube atoms doped improve the electronic properties of nanotubes by altering the electrostatic potential, HOMO and DFT LUMO energies. Based in the adsorption energies and the conductivity increased to ~33 and 350%, respectively, for Si- and 2Si-BNNT imply that they can be used for sarin detection. -
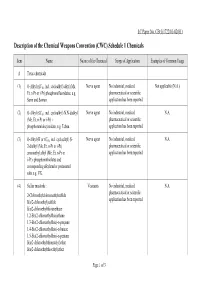
Description of the Chemical Weapons Convention (CWC) Schedule 1 Chemicals
LC Paper No. CB(1)1722/01-02(01) Description of the Chemical Weapons Convention (CWC) Schedule 1 Chemicals Item Name Nature of the Chemical Scope of Application Examples of Common Usage A Toxic chemicals (1) O-Alkyl (≤C10, incl. cycloalkyl) alkyl (Me, Nerve agent No industrial, medical, Not applicable (N.A.) Et, n-Pr or i-Pr) phosphonofluoridates, e.g. pharmaceutical or scientific Sarin and Soman. application has been reported. (2) O-Alkyl (≤C10, incl. cycloalkyl) N,N-dialkyl Nerve agent No industrial, medical, N.A. (Me, Et, n-Pr or i-Pr) - pharmaceutical or scientific phosphoramidocyanidate, e.g. Tabun. application has been reported. (3) O-Alkyl (H or ≤C10, incl. cycloalkyl) S- Nerve agent No industrial, medical, N.A. 2-dialkyl (Me, Et, n-Pr or i-Pr) pharmaceutical or scientific aminoethyl alkyl (Me, Et, n-Pr or application has been reported. i-Pr)- phosphonothiolates and corresponding alkylated or protonated salts e.g. VX. (4) Sulfur mustards : Vesicants No industrial, medical, N.A. pharmaceutical or scientific 2-Chloroethylchloromethylsulfide application has been reported. Bis(2-chloroethyl)sulfide Bis(2-chloroethylthio)methane 1,2-Bis(2-chloroethylthio)ethane 1,3-Bis(2-chloroethylthio)-n-propane 1,4-Bis(2-chloroethylthio)-n-butane 1,5-Bis(2-chloroethylthio)-n-pentane Bis(2-chloroethylthiomethyl)ether Bis(2-chloroethylthioethyl)ether Page 1 of 3 Item Name Nature of the Chemical Scope of Application Examples of Common Usage (5) Lewisites : Vesicants No industrial, medical, N.A. pharmaceutical or scientific Lewisite 1 : 2-Chlorovinyldichloroarsine application has been reported. Lewisite 2 : Bis(2-chlorovinyl)chloroarsine Lewisite 3 : Tris(2-chlorovinyl)arsine (6) Nitrogen mustards : Vesicants The chemical has medical Only HN2 has been reported to application. -

Pacs by Chemical Name (Mg/M3) (Pdf)
Table 4: Protective Action Criteria (PAC) Rev 25 based on applicable 60-minute AEGLs, ERPGs, or TEELs. Values are presented in mg/m3. August 2009 Table 4 is an alphabetical listing of the chemicals in the PAC data set. It provides Chemical Abstract Service Registry Numbers (CASRNs)1, PAC values, and technical information on the source of the PAC values. Table 4 presents all values for TEEL-0, PAC-1, PAC-2, and PAC-3 in mg/m3. The conversion of ppm to mg/m3 is calculated assuming 25 ºC and 760 mm Hg. The columns presented in Table 4 provide the following information: Heading Definition No. The ordered numbering of the chemicals as they appear in this alphabetical listing. Chemical Name The chemical name given to the PAC Development Team. CASRN The Chemical Abstract Service Registry Number for this chemical. TEEL-0 This is the threshold concentration below which most people will experience no adverse health effects. This PAC is always based on TEEL-0 because AEGL-0 or ERPG-0 values do not exist. PAC-1 Based on the applicable AEGL-1, ERPG-1, or TEEL-1 value. PAC-2 Based on the applicable AEGL-2, ERPG-2, or TEEL-2 value. PAC-3 Based on the applicable AEGL-3, ERPG-3, or TEEL-3 value. Source of PACs: Technical comments provided by the PAC development team that TEEL-0, PAC-1, indicate the source of the data used to derive PAC values. Future efforts PAC-2, PAC-3 are directed at reviewing, revising, and enhancing this information. -

459 Part 121—The United States Munitions List
Department of State § 121.1 (h) Equipment is a combination of 121.1 The United States Munitions List. parts, components, accessories, attach- 121.2–121.15 [Reserved] ments, firmware, or software that oper- 121.16 Missile Technology Control Regime ate together to perform a function of, Annex. as, or for an end-item or system. AUTHORITY: Secs. 2, 38, and 71, Pub. L. 90– Equipment may be a subset of an end- 629, 90 Stat. 744 (22 U.S.C. 2752, 2778, 2797); 22 item based on the characteristics of U.S.C. 2651a; Pub. L. 105–261, 112 Stat. 1920; Section 1261, Pub. L. 112–239; E.O. 13637, 78 FR the equipment. Equipment that meets 16129. the definition of an end-item is an end- item. Equipment that does not meet SOURCE: 58 FR 39287, July 22, 1993, unless the definition of an end-item is a com- otherwise noted. ponent, accessory, attachment, ENUMERATION OF ARTICLES firmware, or software. [79 FR 61228, Oct. 10, 2014] § 121.1 The United States Munitions List. § 120.46 Classified. (a) U.S. Munitions List. In this part, Classified means classified pursuant articles, services, and related technical to Executive Order 13526, and a secu- data are designated as defense articles rity classification guide developed pur- or defense services pursuant to sections suant thereto or equivalent, or to the 38 and 47(7) of the Arms Export Control corresponding classification rules of Act and constitute the U.S. Munitions another government or international List (USML). Changes in designations organization. are published in the FEDERAL REG- ISTER. -

ITAR Category
Category XIV—Toxicological Agents, Including Chemical Agents, Biological Agents, and Associated Equipment *(a) Chemical agents, to include: (1) Nerve agents: (i) O-Alkyl (equal to or less than C10, including cycloalkyl) alkyl (Methyl, Ethyl, n-Propyl or Isopropyl)phosphonofluoridates, such as: Sarin (GB): O-Isopropyl methylphosphonofluoridate (CAS 107–44–8) (CWC Schedule 1A); and Soman (GD): O-Pinacolyl methylphosphonofluoridate (CAS 96–64–0) (CWC Schedule 1A); (ii) O-Alkyl (equal to or less than C10, including cycloalkyl) N,N-dialkyl (Methyl, Ethyl, n- Propyl or Isopropyl)phosphoramidocyanidates, such as: Tabun (GA): O-Ethyl N, N- dimethylphosphoramidocyanidate (CAS 77–81–6) (CWC Schedule 1A); (iii) O-Alkyl (H or equal to or less than C10, including cycloalkyl) S–2-dialkyl (Methyl, Ethyl, n- Propyl or Isopropyl)aminoethyl alkyl (Methyl, Ethyl, n-Propyl or Isopropyl)phosphonothiolates and corresponding alkylated and protonated salts, such as: VX: O-Ethyl S-2- diisopropylaminoethyl methyl phosphonothiolate (CAS 50782–69–9) (CWC Schedule 1A); (2) Amiton: O,O-Diethyl S-[2(diethylamino)ethyl] phosphorothiolate and corresponding alkylated or protonated salts (CAS 78–53–5) (CWC Schedule 2A); (3) Vesicant agents: (i) Sulfur mustards, such as: 2-Chloroethylchloromethylsulfide (CAS 2625–76–5) (CWC Schedule 1A); Bis(2-chloroethyl)sulfide (CAS 505–60–2) (CWC Schedule 1A); Bis(2- chloroethylthio)methane (CAS 63839–13–6) (CWC Schedule 1A); 1,2-bis (2- chloroethylthio)ethane (CAS 3563–36–8) (CWC Schedule 1A); 1,3-bis (2-chloroethylthio)-n- propane (CAS -

Development and Applications of Novel
DEVELOPMENT AND APPLICATIONS OF NOVEL SOLVENT-MINIMIZED TECHNIQUES IN THE DETERMINATION OF CHEMICAL WARFARE AGENTS AND THEIR DEGRADATION PRODUCTS LEE HOI SIM NANCY NATIONAL UNIVERSITY OF SINGAPORE 2008 DEVELOPMENT AND APPLICATIONS OF NOVEL SOLVENT-MINIMIZED TECHNIQUES IN THE DETERMINATION OF CHEMICAL WARFARE AGENTS AND THEIR DEGRADATION PRODUCTS LEE HOI SIM NANCY (M.Sc.), NUS A THESIS SUBMITTED FOR THE DEGREE OF DOCTOR OF PHILOSOPHY DEPARTMENT OF CHEMISTRY NATIONAL UNIVERSITY OF SINGAPORE 2008 ACKNOWLEDGEMENTS My most sincere gratitude goes to my bosses at DSO National Laboratories, Ms. Sng Mui Tiang, Dr. Lee Fook Kay and Assoc. Prof. Lionel Lee Kim Hock, who gave me the opportunity to pursue a part-time higher degree and provided me with much encouragement and support throughout the course of my study. I had the privilege of working under the expert guidance of Prof. Lee Hian Kee and Dr. Chanbasha Basheer of the Department of Chemistry at the National University of Singapore and would like to thank them for this enriching learning experience as well as their friendship over the years. I sincerely appreciate the help and support from everyone at the Defense Medical and Environmental Research Institute, DSO National Laboratories, especially from the Organic Synthesis Group for providing the analytes used in the study and all users of the GC MSD for sharing the use of the instrument. Even though people come and go, the memories of the exciting times especially during the Proficiency Tests, with Dr. Ang Kiam Wee, Ms. Chan Shu Cheng, Mr. Leonard Chay Yew Leong, Dr. Alex Chin Piao, Dr. Chua Guan Leong, Ms. -

Table of Contents
UNIVERSITY OF MISSOURI-KANSAS CITY CHEMICAL MANAGEMENT PLAN Revised May 2016 UMKC CHEMICAL MANAGEMENT PLAN This document constitutes the Chemical Management Plan (CMP) for the University of Missouri-Kansas City (UMKC). It was developed by the Environmental Health and Safety Department (EHS), to ensure the safe and proper use of hazardous and non- hazardous chemicals and to comply with applicable governmental regulations addressing the disposal of these chemicals. In addition, it was developed to foster waste minimization, and to provide the faculty and the staff with a management program to reduce the potential for accidents involving hazardous chemicals and/or wastes. Elements of the CMP include: a. a procedure for identifying potential or actual hazardous chemicals or wastes b. a procedure for periodic reexamination of those hazardous chemicals or wastes identified by the procedure in (a.) above as well as a systematic method for identification and evaluation of any new potential or actual hazardous chemicals or wastes c. procedures for labeling, and inventorying hazardous chemicals or wastes d. a procedure for identification and training of personnel directly responsible for ensuring that (a.), (b.), and (c.) are implemented e. a procedure for monitoring, recording, and reporting compliance with the CMP f. a procedure by which information generated by the CMP is provided to the persons performing waste analyses Each element is addressed as part of the complete CMP in the following paragraphs. 4 Table of Contents 1 Definitions 7 2 Identification -

Acetylcholinesterase: the “Hub” for Neurodegenerative Diseases And
Review biomolecules Acetylcholinesterase: The “Hub” for NeurodegenerativeReview Diseases and Chemical Weapons Acetylcholinesterase: The “Hub” for Convention Neurodegenerative Diseases and Chemical WeaponsSamir F. de A. Cavalcante Convention 1,2,3,*, Alessandro B. C. Simas 2,*, Marcos C. Barcellos 1, Victor G. M. de Oliveira 1, Roberto B. Sousa 1, Paulo A. de M. Cabral 1 and Kamil Kuča 3,*and Tanos C. C. França 3,4,* Samir F. de A. Cavalcante 1,2,3,* , Alessandro B. C. Simas 2,*, Marcos C. Barcellos 1, Victor1 Institute G. M. ofde Chemical, Oliveira Biological,1, Roberto Radiological B. Sousa and1, Paulo Nuclear A. Defense de M. Cabral (IDQBRN),1, Kamil Brazilian Kuˇca Army3,* and TanosTechnological C. C. França Center3,4,* (CTEx), Avenida das Américas 28705, Rio de Janeiro 23020-470, Brazil; [email protected] (M.C.B.); [email protected] (V.G.M.d.O.); [email protected] 1 Institute of Chemical, Biological, Radiological and Nuclear Defense (IDQBRN), Brazilian Army (R.B.S.); [email protected] (P.A.d.M.C.) Technological Center (CTEx), Avenida das Américas 28705, Rio de Janeiro 23020-470, Brazil; 2 [email protected] Mors Institute of Research (M.C.B.); on Natural [email protected] Products (IPPN), Federal (V.G.M.d.O.); University of Rio de Janeiro (UFRJ), CCS,[email protected] Bloco H, Rio de Janeiro (R.B.S.); 21941-902, [email protected] Brazil (P.A.d.M.C.) 32 DepartmentWalter Mors of Institute Chemistry, of Research Faculty of on Science, Natural Un Productsiversity (IPPN), -

Chemical Weapons Convention Inventory Report - 2017
University of British Columbia Risk Management Services CHEMICAL WEAPONS CONVENTION INVENTORY REPORT - 2017 Department: Name of Building: Room No. Principal Investigator: SCHEDULE 1 CHEMICALS Location Quantity Obtained /Stored / Used A.Toxic Chemicals 1 O-Alkyl (<=C10, incl. cycloalkyl) alkyl (Me, Et, n-Pr or i-Pr) – phosphonofluoridates, Sarin: O-Isopropyl methylphosphonofluoridate (CAS 107-44-8) Soman: O-Pinacolyl methylphosphonofluoridate (CAS 96-64-0) 2 O-Alkyl (H or <=C10, incl. cycloalkyl) S-2-dialkyl (Me, Et, n- Pr or i-Pr) -aminoethyl alkyl (Me, Et, n-Pr or i-Pr) phosphonothiolates, and corresponding alkylated or protonated salts VX: O-Ethyl S-2-diisopropylaminoethyl methyl phosphonothiolate (CAS 50782-69-9) 3 O-Alkyl (H or <=C10, incl. cycloalkyl) S-2-dialkyl (Me, Et, n- Pr or i-Pr) -aminoethyl alkyl (Me, Et, n-Pr or i-Pr) phosphonothiolates, and corresponding alkylated or protonated salts VX: O-Ethyl S-2-diisopropylaminoethyl methyl phosphonothiolate (CAS 50782-69-9) 4 Sulphur Mustards: 2-Chloroethylchloromethylsulfide (CAS 2625-76-5) Mustard gas: Bis (2-chloroethyl) sulfide (CAS 505-60-2) Bis (2-chloroethylthio)methane (CAS 63869-13-6) Sesquimustard: 1, 2-Bis (2-chloroethylthio) ethane (CAS 3563-36-8) 1, 3-Bis (2-chloroethylthio) -n-propane (CAS 63905-10-2) 1, 4-Bis (2-chloroethylthio) -n-butane (CAS 142868-93-7) 1, 5-Bis (2-chloroethylthio) -n-pentane (CAS 142868-94-8) Bis (2-chloroethylthiomethyl) ether (CAS 63918-90-1) O-Mustard: Bis (2-chloroethylthioethyl) ether (CAS 63918-89-8) 5 Lewisites: Lewisite 1: 2-Chlorovinyldichloroarsine (CAS 541-25-3) Lewisite 2: Bis (2-chlorovinyl) chloroarsine (CAS 40334-69-8) Lewisite 3: Tris (2-chlorovinyl) arsine (CAS 40334-70-1) 6 Nitrogen Mustards: HN1: Bis (2-chloroethyl) ethylamine (CAS 538-07-08) HN2: Bis (2-chloroethyl) methylamine (CAS 51-75-2) HN3: Tris (2-chloroethyl) amine (CAS 555-77-1) University of British Columbia Risk Management Services CHEMICAL WEAPONS CONVENTION INVENTORY REPORT - 2017 Department: Name of Building: Room No. -

Department of Homeland Security (Dhs) Appendix a Chemicals of Interest (Coi)
DEPARTMENT OF HOMELAND SECURITY (DHS) APPENDIX A CHEMICALS OF INTEREST (COI) Acetaldehyde Bromine trifluoride Acetone Cyanohydrin, stabilized Bromothrifluorethylene Acetyl bromide 1,3-Butadiene Acetyl chloride Butane Acetyl iodide Butene Acetylene 1-Butene Acrolein 2-Butene Acrylonitrile 2-Butene-cis Acrylyl chloride 2-Butene-trans Allyl alcohol Butyltrichlorosilane Allylamine Calcium hydrosulfite Allyltrichlorosilane, stabilized Calcium phosphide Aluminum (powder) Carbon disulfide Aluminum Bromide, anhydrous Carbon oxysulfide Aluminum Chloride, anhydrous Carbonyl fluoride Aluminum phosphide Carbonyl sulfide Ammonia (anhydrous) Chlorine Ammonia (conc 20% or greater) Chlorine dioxide Ammonium nitrate, note #1 Chlorine monoxide Ammonium nitrate, note #2 Chlorine pentafluoride Ammonium perchlorate Chlorine trifluoride Ammonium picrate Chloroacetyl chloride Amyltrichlorosilane 2-Chloroethylchloro-methylsulfide Antimony pentafluoride Chloroform Arsenic trichloride Chloromethyl ether Arsine Chloromethyl methyl ether Barium azide 1-Chloropropylene 1,4-Bis(2-chloroethylthio)-n-butane 2-Chloropropylene Bis(2-chloroethylthio) methane Chlorosarin Bis(2-chloroethylthiomethyl)ether Chlorosoman 1,5-Bis(2-chloroethylthio)-n-pentane Chlorosulfonic acid 1,3-Bis(2-chloroethylthio)-n-propane Chromium oxychloride Boron tribromide Crotonaldehyde Boron trichloride Crotonaldehyde, (E)- Boron trifluoride Cyanogen Boron trifluoride compound with methyl Cyanogen chloride ether (1:1) Cyclohexylamine Bromine Cyclohexyltrichlorosilane Bromine chloride Cyclopropane -

Export Control Handbook for Chemicals
Export Control Handbook for Chemicals -Dual-use control list -Common Military List -Explosives precursors -Syria restrictive list -Psychotropics and narcotics precursors ARNES-NOVAU, X 2019 EUR 29879 This publication is a Technical report by the Joint Research Centre (JRC), the European Commission’s science and knowledge service. It aims to provide evidence-based scientific support to the European policymaking process. The scientific output expressed does not imply a policy position of the European Commission. Neither the European Commission nor any person acting on behalf of the Commission is responsible for the use that might be made of this publication. Contact information Xavier Arnés-Novau Joint Research Centre, Via Enrico Fermi 2749, 21027 Ispra (VA), Italy [email protected] Tel.: +39 0332-785421 Filippo Sevini Joint Research Centre, Via Enrico Fermi 2749, 21027 Ispra (VA), Italy [email protected] Tel.: +39 0332-786793 EU Science Hub https://ec.europa.eu/jrc JRC 117839 EUR 29879 Print ISBN 978-92-76-11971-5 ISSN 1018-5593 doi:10.2760/844026 PDF ISBN 978-92-76-11970-8 ISSN 1831-9424 doi:10.2760/339232 Luxembourg: Publications Office of the European Union, 2019 © European Atomic Energy Community, 2019 The reuse policy of the European Commission is implemented by Commission Decision 2011/833/EU of 12 December 2011 on the reuse of Commission documents (OJ L 330, 14.12.2011, p. 39). Reuse is authorised, provided the source of the document is acknowledged and its original meaning or message is not distorted. The European Commission shall not be liable for any consequence stemming from the reuse. -

CSAT Top-Screen Questions OMB PRA # 1670-0007 Expires: 5/31/2011
CSAT Top-Screen Questions January 2009 Version 2.8 CSAT Top-Screen Questions OMB PRA # 1670-0007 Expires: 5/31/2011 Change Log .........................................................................................................3 CVI Authorizing Statements...............................................................................4 General ................................................................................................................6 Facility Description.................................................................................................................... 7 Facility Regulatory Mandates ................................................................................................... 7 EPA RMP Facility Identifier....................................................................................................... 9 Refinery Capacity....................................................................................................................... 9 Refinery Market Share ............................................................................................................. 10 Airport Fuels Supplier ............................................................................................................. 11 Military Installation Supplier................................................................................................... 11 Liquefied Natural Gas (LNG) Capacity................................................................................... 12 Liquefied Natural Gas Exclusion