Table of Contents
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

United States Patent Office Patented Aug
3,202,556 United States Patent Office Patented Aug. 24, 1965 2 mannan. As a consequence, crosslinking agents are used METHOD FOR GELiNGWATER-BEARNG3,282,555 EX to hasten the formation of gels, or they may be employed PLGSIVE COMPOSTIONS CONTAINNG GAYLAC to form gels which have desired physical properties at rel TOMANNAN (GUAS atively low gum concentrations, or even have properties Joseph D. Chrisp, Caymorf, Dai, assig or to E. I. du not obtainable by the use of the gum alone. An effective Poit de Neno Ers and Company, Wilmington, De., a and Well known crosslinking agent for the hydrated galac corporation of Delaware tomannans, especially for guar gum, is the borate ion No iPrawing. Fied July 23, 1963, Ser. No. 296,932 which acts as a crosslinking agent with hydrated guar gum 14 (Claims. (C. 49-20) to form cohesive structural gels. The process of cross 10 linking guar-containing compositions by adding borate This invention relates to a process for gelling aqueous ions has been employed in the manufacture of ammonium solutions of water-dispersible, non-ionic hydroxyl poly nitrate-based blasting agent compositions, as disclosed and mers. More particularly, this invention relates to a proc claimed in U.S. Patent No. 3,072,509. Such blasting ess for making water-bearing gels containing galactoman agents have a desirable body which resists dispersion of nans and to certain products obtained thereby including 5 the compositions by water and facilitates their handling explosives. and loading. Also, such crosslinked blasting agents are Water-soluble gums have been articles of commerce economical and safe to use, and have better storage prop for hundreds of years. -

Hexamethylenediamine (HMDA) from Fossil Vs. Bio-Based Routes: an Economic and Life Cycle Assessment Comparative Study
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is © The Royal Society of Chemistry 2015 Hexamethylenediamine (HMDA) from Fossil vs. Bio-Based Routes: an Economic and Life Cycle Assessment Comparative Study A. B. Dros,b O. Larue,b A. Reimond,b F. De Campoa and M. Pera-Titusa* a Eco-Efficient Products and Processes Laboratory (E2P2L), UMI 3464 CNRS-Solvay, 3966 Jin Du Road, Xin Zhuang Ind. Zone, 201108 Shanghai, China. b Solvay (China) Co., Ltd., 3966 Jin Du Rd., Xin Zhuang Industrial Zone, Shanghai 201108, PR China. * Corresponding author. Tel.: +86 (0) 472445368, Fax: +86 (0) 472445399, E-mail: marc.pera-titus- [email protected] ELECTRONIC SUPPORTING INFORMATION FIGURE AND TABLE CAPTIONS Fig. S1. Speculative routes for the production of bio-based HMDA using molecules issued from biomass. Fig. S2. Flowsheet for the fossil-based route 1. Fig. S3. Flowsheet for the route Starch HFS. The bold number corresponds to the base-case value for steam consumption used in the LCA sensitivity study. The w/o HFS drying scenario value is also indicated. Fig. S4. Flowsheet for the bio-based route 2. Fig. S5. Flowsheet for the bio-based route 3. The bold numbers correspond to the base-case values used in the LCA sensitivity study. For HFS, the best- and worst case scenario values are also indicated. Fig. S6. Flowsheet for the bio-based route 4. Fig. S7. Evolution of market price of butadiene and HFCS42%. Data obtained from ref.18 and ref.21, respectively. Fig. S8. Impact score breakdown for ozone depletion (midpoint category 2) for HMDA production in France and Germany. -

Theoretical Study of Sarin Adsorption On
Chemical Physics Letters 738 (2020) 136816 Contents lists available at ScienceDirect Chemical Physics Letters journal homepage: www.elsevier.com/locate/cplett Research paper Theoretical study of sarin adsorption on (12,0) boron nitride nanotube doped with silicon atoms T ⁎ ⁎ Jeziel Rodrigues dos Santosa, , Elson Longo da Silvab, Osmair Vital de Oliveirac, , José Divino dos Santosa a Universidade Estadual de Goiás, Campus Anápolis, CEP: 75.132-903 GO, Brazil b INCTMN, LIEC, Departamento de Química da Universidade Federal de São Carlos, CEP: 13.565-905 São Carlos, SP, Brazil c Instituto Federal de Educação, Ciência e Tecnologia de São Paulo, Campus Catanduva, CEP: 15.808-305 Catanduva, SP, Brazil HIGHLIGHTS • DFT method was used to study the adsorption of nerve agent sarin by BNNT. • Electronic properties of pristine BNNT are improved by Si impurity atoms. • The adsorption of sarin by Si-doped BNNT is highest favorable than the pure BNNT. • Si-doped BNNT can be a new gas sensor for sarin gas detection and its derivatives. ARTICLE INFO ABSTRACT Keywords: Sarin gas is one of the most lethal nerve agent used in chemical warfare, which its detection is import to prevent Nerve agent sarin a chemical attack and to identify a contamination area. Herein, density functional theory was used to investigate Gas sensor the (12,0) boron nitride nanotube (BNNT) and Si–doped BNNT as possible candidates to sarin detection. The Si- Boron nitride nanotube atoms doped improve the electronic properties of nanotubes by altering the electrostatic potential, HOMO and DFT LUMO energies. Based in the adsorption energies and the conductivity increased to ~33 and 350%, respectively, for Si- and 2Si-BNNT imply that they can be used for sarin detection. -

(A) 1) at First a Pair of Each 10G. of Sample Was Extracted with Ether by the Soxhlet's Apparatus for a Week
52 [vol. 6, THE DISTRIBUTION OF PHOSPHORUS IN COW'S MILK AND THE SCHEME FOR THE SEPARATION OF PHOSPHATIDES By Rinjiro SASAKI (From the Institute of Agricultural Chemistry, College of Agriculture, Tokyo Imperial University) (Received 9th September 1930) A. The Distribution of Phosphorus in Cow's milk In order to extract the phosphatides, it is necessary first to determine the distribution of phosphorus in cow's milk. Liquid milk can not be extracted with ether or with other organic solvents owing to its large content of water. The substance applied in this experiment is the milk powder, dried by the Buflovak drum drier below 70•Ž. Its content of moisture is 4.498%, deter mined by the method of drying in the air bath of 105•Ž. The content of total phosphorus was determined(5) in the ash of sample which was burned with a little of fusing mixture. In 100g. of milk powder In 100g. of dry matter Total phosphorus 0.745g. 0.781g. The amounts of phosphorus, which are soluble in the various organic sol vents, were determined by extracting with three solvents in the following orders. (A) Ether-Acetone-Alcohol (B) Acetone-Ether-Alcohol (C) Alcohol-Ether-Acetone Experiment (A) 1) At first a pair of each 10g. of sample was extracted with ether by the Soxhlet's apparatus for a week. After the extraction had been comp leted, the extract was freed from ether and weighed. In 100g.of milkpowder In 100g.of dry matter Total ether matters soluble 3.609g. 3.779g. 2) The above extract was dissolved in a very little of ether. -

Hexamethylenediamine Cas N°: 124-09-4
OECD SIDS HEXAMETHYLENEDIAMINE FOREWORD INTRODUCTION HEXAMETHYLENEDIAMINE CAS N°: 124-09-4 UNEP PUBLICATIONS Identifiers, Physical and Chemical properties 165 Substance End Point : IDENTIFIERS, PHYSICAL AND CHEMICAL PROPERTIES Chemical Name : 1,6-Hexanediamine Common Name : Hexamethylenediamine CAS Number : 124-09-4 RTECS Number : MO1180000 Synonyms 1,6-Diaminohexane .alpha.-.omega.-Hexanediamine Hexylenediamine 1,6-Hexylenediamine HMD HMDA Properties & Definitions Molecular Formula : C6H16N2 Molecular Weight : 116.24 Melting Point : 41C Boiling Point : 205C Flash Point : 0.9 - 7.6 volume % Vapour Pressure : 0.05 kPa (0.4 mmHg) at 25C CAL Octanol/Water Partition : log Pow = 0.02 Coefficient Water Solubility : 800 g/L at 15.6C Additives : None Impurities : None. Purity of industrial product: 100% General Comments : Flammability (solids/gases): 85C. Ignition temperature: 305C. Overall Evaluation SIDS INITIAL ASSESSMENT CURRENTLY OF LOW PRIORITY FOR FURTHER WORK Hexamethylendiamine (HMDA) is an isolated chemical intermediate which is used for the manufacture of polyamides. Information regarding uses, production levels, exposure, and emissions was available only from the DuPont Company in Canada and in the United States. Tasks involving the exposure to HMDA are of short duration; therefore, occupational exposure is expected to be limited. Air monitoring at plant sites has detected =< 0.07 ppm HMDA; personal monitoring values range from 0.01 to 3.7 ppm. It is also expected that consumer exposure is negligible since HMDA is generally incorporated into other products before reaching the consumer; however, consumer exposure will need to be reassessed when additional exposure data is received from other countries. Under environmental conditions, HMDA will exist in an ionic state (+2). -

United States Patent Office Patented Apr
3,030,421 United States Patent Office Patented Apr. 17, 1962 2 3,030,421 perature and under a reduced pressure. In the case of PROCESS FOR PREPARNG TRHYDROXY most of the catalysts the reaction products, which are free METHYL-PHOSPHINE s from solvents, solidify already when being cooled to room Martin Reuter and Ludwig Ortane, Frankfurt an Main, temperature. If, in the case of some catalysts, they Germany, assignors to Farbwerke Hoechst Aktienger 5 solidify only at a lower temperature and still contain to sellschaft vormas Meister Lucius & Briining, Frank a greater extent oily by-products and/or phosphonium furt am Main, Germany, a corporation of Gerinally hydroxide, they can be separated from the latter by filter No Drawing. Fied Jaa. 14, 1958, Ser. No. 708,764 ing or pressing. - - - Claims priority, application Germany Jan. 23, 1957 It is to be assumed that the crystalline main product 6 Canas. (C. 260-606.5) O of the present invention constitutes the hitherto unknown We have found that a new and valuable phosphorus trihydroxymethyl-phosphine. Main and by-products are compound carrying hydroxymethyl groups at the phos easily soluble in water and methanol and sparingly solu phorus atom can be prepared by reacting 1 mol of formal ble in fat dissolvers. dehyde with 4 mol of phosphine, preferably in the pres The reaction products of the invention can be used as ence of water, and in the presence of Small quantities of 5 insecticides, additives for lubricants, flame-proofing agents finely distributed metals that do not belong to the alkali for wood and textiles and as intermediates for these sub metals or alkaline earth metals and/or their compounds Staces. -
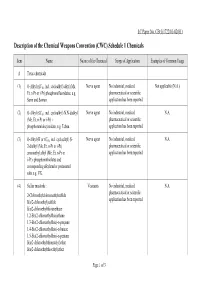
Description of the Chemical Weapons Convention (CWC) Schedule 1 Chemicals
LC Paper No. CB(1)1722/01-02(01) Description of the Chemical Weapons Convention (CWC) Schedule 1 Chemicals Item Name Nature of the Chemical Scope of Application Examples of Common Usage A Toxic chemicals (1) O-Alkyl (≤C10, incl. cycloalkyl) alkyl (Me, Nerve agent No industrial, medical, Not applicable (N.A.) Et, n-Pr or i-Pr) phosphonofluoridates, e.g. pharmaceutical or scientific Sarin and Soman. application has been reported. (2) O-Alkyl (≤C10, incl. cycloalkyl) N,N-dialkyl Nerve agent No industrial, medical, N.A. (Me, Et, n-Pr or i-Pr) - pharmaceutical or scientific phosphoramidocyanidate, e.g. Tabun. application has been reported. (3) O-Alkyl (H or ≤C10, incl. cycloalkyl) S- Nerve agent No industrial, medical, N.A. 2-dialkyl (Me, Et, n-Pr or i-Pr) pharmaceutical or scientific aminoethyl alkyl (Me, Et, n-Pr or application has been reported. i-Pr)- phosphonothiolates and corresponding alkylated or protonated salts e.g. VX. (4) Sulfur mustards : Vesicants No industrial, medical, N.A. pharmaceutical or scientific 2-Chloroethylchloromethylsulfide application has been reported. Bis(2-chloroethyl)sulfide Bis(2-chloroethylthio)methane 1,2-Bis(2-chloroethylthio)ethane 1,3-Bis(2-chloroethylthio)-n-propane 1,4-Bis(2-chloroethylthio)-n-butane 1,5-Bis(2-chloroethylthio)-n-pentane Bis(2-chloroethylthiomethyl)ether Bis(2-chloroethylthioethyl)ether Page 1 of 3 Item Name Nature of the Chemical Scope of Application Examples of Common Usage (5) Lewisites : Vesicants No industrial, medical, N.A. pharmaceutical or scientific Lewisite 1 : 2-Chlorovinyldichloroarsine application has been reported. Lewisite 2 : Bis(2-chlorovinyl)chloroarsine Lewisite 3 : Tris(2-chlorovinyl)arsine (6) Nitrogen mustards : Vesicants The chemical has medical Only HN2 has been reported to application. -

"The Science for Diplomats" Annex on Chemicals
ORGANISATION FOR THE PROHIBITION OF CHEMICAL WEAPONS "THE SCIENCE FOR DIPLOMATS" ANNEX ON CHEMICALS A user friendly and scientifically annotated version of the Chemical Weapons Convention Annex on Chemicals OPCW THE “SCIENCE FOR DIPLOMATS” ANNEX ON CHEMICALS A user friendly and scientifically annotated version of the Chemical Weapons Convention Annex on Chemicals1 CONTENTS A. GUIDELINES FOR SCHEDULES OF CHEMICALS B. VISUALISING AND READING MOLECULAR STRUCTURES C. SCHEDULES OF CHEMICALS D. RIOT CONTROL AGENTS 1 An official version of the Annex on Chemicals can be obtained from the OPCW public website, www.opcw.org/chemical-weapons-convention/annexes/annex-chemicals/annex-chemicals. Version 3.0 – 10 March 2019 A. GUIDELINES FOR SCHEDULES OF CHEMICALS Guidelines for Schedule 1 1. The following criteria shall be taken into account in considering whether a toxic chemical or precursor should be included in Schedule 1: (a) It has been developed, produced, stockpiled or used as a chemical weapon as defined in Article II; (b) It poses otherwise a high risk to the object and purpose of this Convention by virtue of its high potential for use in activities prohibited under this Convention because one or more of the following conditions are met: (i) It possesses a chemical structure closely related to that of other toxic chemicals listed in Schedule 1, and has, or can be expected to have, comparable properties; (ii) It possesses such lethal or incapacitating toxicity as well as other properties that would enable it to be used as a chemical weapon; (iii) It may be used as a precursor in the final single technological stage of production of a toxic chemical listed in Schedule 1, regardless of whether this stage takes place in facilities, in munitions or elsewhere; (c) It has little or no use for purposes not prohibited under this Convention. -

Thermodynamic Properties of Mixtures of Aqueous Solutions of Hydrochloric Acid and Cadmium Chloride, Copper Chloride, Manganese Chloride, and Zinc Chloride
Western Michigan University ScholarWorks at WMU Master's Theses Graduate College 12-1993 Thermodynamic Properties of Mixtures of Aqueous Solutions of Hydrochloric Acid and Cadmium Chloride, Copper Chloride, Manganese Chloride, and Zinc Chloride Samia A. Kosa Follow this and additional works at: https://scholarworks.wmich.edu/masters_theses Part of the Chemistry Commons Recommended Citation Kosa, Samia A., "Thermodynamic Properties of Mixtures of Aqueous Solutions of Hydrochloric Acid and Cadmium Chloride, Copper Chloride, Manganese Chloride, and Zinc Chloride" (1993). Master's Theses. 4363. https://scholarworks.wmich.edu/masters_theses/4363 This Masters Thesis-Open Access is brought to you for free and open access by the Graduate College at ScholarWorks at WMU. It has been accepted for inclusion in Master's Theses by an authorized administrator of ScholarWorks at WMU. For more information, please contact [email protected]. THERMODYNAMIC PROPERTIES OF MIXTURES OF AQUEOUS SOLUTIONS OF HYDROCHLORIC ACID AND CADMIUM CHLORIDE, COPPER CHLORIDE, MANGANESE CHLORIDE, AND ZINC CHLORIDE by Samia A. Kosa A Thesis Submitted to the Faculty of The Graduate College in partial fulfillment of the requirements for the Degree of Master of Arts Department of Chemistry Western Michigan University Kalamazoo, Michigan December 1993 ACKNOWLEDGEMENTS I wish to express my deep gratitude and sincere appreciation to my committee advisor, Dr. Donald Schriber, for his continual guidance, direction, and assistance in this research. I would also like to acknowledge the help of the other committee members, Dr. Thomass Houser and Dr. Ralph Steinhaus, towards this work. My family continuously provided me support and encouragement and to them I owe more than I can say. -

Download Author Version (PDF)
Journal of Materials Chemistry A Accepted Manuscript This is an Accepted Manuscript, which has been through the Royal Society of Chemistry peer review process and has been accepted for publication. Accepted Manuscripts are published online shortly after acceptance, before technical editing, formatting and proof reading. Using this free service, authors can make their results available to the community, in citable form, before we publish the edited article. We will replace this Accepted Manuscript with the edited and formatted Advance Article as soon as it is available. You can find more information about Accepted Manuscripts in the Information for Authors. Please note that technical editing may introduce minor changes to the text and/or graphics, which may alter content. The journal’s standard Terms & Conditions and the Ethical guidelines still apply. In no event shall the Royal Society of Chemistry be held responsible for any errors or omissions in this Accepted Manuscript or any consequences arising from the use of any information it contains. www.rsc.org/materialsA Page 1 of 9 Journal of Materials Chemistry A ARTICLE JMCA Safer Salts for CdTe Nanocrystal Solution Processed Solar Cells: The Dual Roles of Ligand Exchange and Grain Growth Received 00th January 20xx, a b c d e Accepted 00th January 20xx Troy K. Townsend, † William B. Heuer, Edward E. Foos, Eric Kowalski, Woojun Yoon and Joseph G. Tischler e DOI: 10.1039/x0xx00000x Inorganic CdSe/CdTe nanocrystals for solid-state photovoltaic devices are typically sintered into a bulk-like material after www.rsc.org/ annealing in the presence of solid cadmium chloride. -

United States Patent Office Patented Feb
3,235,600 United States Patent Office Patented Feb. 15, 1966 2 quality and color stability of the polyamide product re 3,235,600 Sulting from the polycondensation of hexamethylenedi REDUCTION OF DAMENOCYCLOHEXANE CON. CENTRATION IN CRUDE HEXAMETYLENED amine with dibasic carboxylic acids. The 1,2-diamino AMNE cyclohexane gives rise to salt solutions of hexamethylene Philip W. Evans, Pensacola, Fla., assignor to Monsanto diamine and dibasic carboxylic acids which have poor Company, a corporation of Delaware and variable color, and the polyamides prepared from No Drawing. Filed Nov. 28, 1962, Ser. No. 240,742 these Salt solutions not only have poor color but also are characterized by irregular tensile strength and nonuniform - 4 Claims. (C. 260-583) dyeing properties. This invention relates to the production of amines and Although one major cause for polyamide product more particularly, it relates to a process for the produc 10 quality, color, and dyeing problems has been ascertained tion of amines in a high degree of purity. and explained, the saisfactory removal on a commercial Hexamethylenediamine is now a well-known com Scale of the Small amounts of 1,2-diaminocyclohexane pound which may be prepared on a commercial scale present in manufactured crude hexamethylenediamine to most conveniently by catalytically hydrogenating adipo eliminate the cause presents a difficult problem. Accord nitrile in the presence of ammonia. A principal use of ing to present commercial practices, the 1,2-diaminocyclo hexamethylenediamine involves condensing -

Used at Rocky Flats
. TASK 1 REPORT (Rl) IDENTIFICATION OF CHEMICALS AND RADIONUCLIDES USED AT ROCKY FLATS I PROJECT BACKGROUND ChemRisk is conducting a Rocky Flats Toxicologic Review and Dose Reconstruction study for The Colorado Department of Health. The two year study will be completed by the fall of 1992. The ChemRisk study is composed of twelve tasks that represent the first phase of an independent investigation of off-site health risks associated with the operation of the Rocky Flats nuclear weapons plant northwest of Denver. The first eight tasks address the collection of historic information on operations and releases and a detailed dose reconstruction analysis. Tasks 9 through 12 address the compilation of information and communication of the results of the study. Task 1 will involve the creation of an inventory of chemicals and radionuclides that have been present at Rocky Flats. Using this inventory, chemicals and radionuclides of concern will be selected under Task 2, based on such factors as the relative toxicity of the materials, quantities used, how the materials might have been released into the environment, and the likelihood for transport of the materials off-site. An historical activities profile of the plant will be constructed under Task 3. Tasks 4, 5, and 6 will address the identification of where in the facility activities took place, how much of the materials of concern were released to the environment, and where these materials went after the releases. Task 7 addresses historic land-use in the vicinity of the plant and the location of off-site populations potentially affected by releases from Rocky Flats.