Kinship and Intra-Group Social Dynamics in Two Sympatric African
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
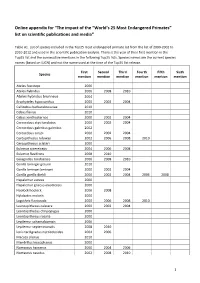
Online Appendix for “The Impact of the “World's 25 Most Endangered
Online appendix for “The impact of the “World’s 25 Most Endangered Primates” list on scientific publications and media” Table A1. List of species included in the Top25 most endangered primate list from the list of 2000-2002 to 2010-2012 and used in the scientific publication analysis. There is the year of their first mention in the Top25 list and the consecutive mentions in the following Top25 lists. Species names are the current species names (based on IUCN) and not the name used at the time of the Top25 list release. First Second Third Fourth Fifth Sixth Species mention mention mention mention mention mention Ateles fusciceps 2006 Ateles hybridus 2006 2008 2010 Ateles hybridus brunneus 2004 Brachyteles hypoxanthus 2000 2002 2004 Callicebus barbarabrownae 2010 Cebus flavius 2010 Cebus xanthosternos 2000 2002 2004 Cercocebus atys lunulatus 2000 2002 2004 Cercocebus galeritus galeritus 2002 Cercocebus sanjei 2000 2002 2004 Cercopithecus roloway 2002 2006 2008 2010 Cercopithecus sclateri 2000 Eulemur cinereiceps 2004 2006 2008 Eulemur flavifrons 2008 2010 Galagoides rondoensis 2006 2008 2010 Gorilla beringei graueri 2010 Gorilla beringei beringei 2000 2002 2004 Gorilla gorilla diehli 2000 2002 2004 2006 2008 Hapalemur aureus 2000 Hapalemur griseus alaotrensis 2000 Hoolock hoolock 2006 2008 Hylobates moloch 2000 Lagothrix flavicauda 2000 2006 2008 2010 Leontopithecus caissara 2000 2002 2004 Leontopithecus chrysopygus 2000 Leontopithecus rosalia 2000 Lepilemur sahamalazensis 2006 Lepilemur septentrionalis 2008 2010 Loris tardigradus nycticeboides -

REVIEW ARTICLE Agroecosystems and Primate Conservation in the Tropics: a Review
American Journal of Primatology 74:696–711 (2012) REVIEW ARTICLE Agroecosystems and Primate Conservation in The Tropics: A Review ∗ ALEJANDRO ESTRADA1 , BECKY E. RABOY2,3, AND LEONARDO C. OLIVEIRA3-6 1Estaci´on de Biolog´ıa Tropical Los Tuxtlas Instituto de Biolog´ıa, Universidad Nacional Aut´onoma de M´exico, Mexico City, Mexico 2Conservation Ecology Center, Smithsonian Conservation Biology Institute, National Zoological Park, Washington, DC 3Instituto de Estudos S´ocioambientais do Sul da Bahia (IESB), Ilh´eus-BA, Brazil 4Programa de P´os-Gradua¸c˜ao em Ecologia, Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brazil 5Programa de P´os-Gradua¸c˜ao em Ecologia e Conserva¸c˜ao da Biodiversidade, Universidade Estadual de Santa Cruz, Ilh´eus-BA, Brazil 6Bicho do Mato Instituto de Pesquisa, Belo Horizonte-MG, Brazil Agroecosystems cover more than one quarter of the global land area (ca. 50 million km2) as highly simplified (e.g. pasturelands) or more complex systems (e.g. polycultures and agroforestry systems) with the capacity to support higher biodiversity. Increasingly more information has been published about primates in agroecosystems but a general synthesis of the diversity of agroecosystems that primates use or which primate taxa are able to persist in these anthropogenic components of the landscapes is still lacking. Because of the continued extensive transformation of primate habitat into human-modified landscapes, it is important to explore the extent to which agroecosystems are used by primates. In this article, we reviewed published information on the use of agroecosystems by primates in habitat countries and also discuss the potential costs and benefits to human and nonhuman primates of primate use of agroecosystems. -

World's Most Endangered Primates
Primates in Peril The World’s 25 Most Endangered Primates 2016–2018 Edited by Christoph Schwitzer, Russell A. Mittermeier, Anthony B. Rylands, Federica Chiozza, Elizabeth A. Williamson, Elizabeth J. Macfie, Janette Wallis and Alison Cotton Illustrations by Stephen D. Nash IUCN SSC Primate Specialist Group (PSG) International Primatological Society (IPS) Conservation International (CI) Bristol Zoological Society (BZS) Published by: IUCN SSC Primate Specialist Group (PSG), International Primatological Society (IPS), Conservation International (CI), Bristol Zoological Society (BZS) Copyright: ©2017 Conservation International All rights reserved. No part of this report may be reproduced in any form or by any means without permission in writing from the publisher. Inquiries to the publisher should be directed to the following address: Russell A. Mittermeier, Chair, IUCN SSC Primate Specialist Group, Conservation International, 2011 Crystal Drive, Suite 500, Arlington, VA 22202, USA. Citation (report): Schwitzer, C., Mittermeier, R.A., Rylands, A.B., Chiozza, F., Williamson, E.A., Macfie, E.J., Wallis, J. and Cotton, A. (eds.). 2017. Primates in Peril: The World’s 25 Most Endangered Primates 2016–2018. IUCN SSC Primate Specialist Group (PSG), International Primatological Society (IPS), Conservation International (CI), and Bristol Zoological Society, Arlington, VA. 99 pp. Citation (species): Salmona, J., Patel, E.R., Chikhi, L. and Banks, M.A. 2017. Propithecus perrieri (Lavauden, 1931). In: C. Schwitzer, R.A. Mittermeier, A.B. Rylands, F. Chiozza, E.A. Williamson, E.J. Macfie, J. Wallis and A. Cotton (eds.), Primates in Peril: The World’s 25 Most Endangered Primates 2016–2018, pp. 40-43. IUCN SSC Primate Specialist Group (PSG), International Primatological Society (IPS), Conservation International (CI), and Bristol Zoological Society, Arlington, VA. -

Project Update: August 2002 Habitat Quality and Fragmentation and The
Project Update: August 2002 Habitat Quality and Fragmentation and the Distribution and Abundance of the Tana River Red Colobus Monkey, Procolobus Rufomitratus, in Eastern Kenya Habitat fragmentation constitutes the most significant threat to survival of primates in the world and therefore there is an urgent need for effective conservation measures that protect primates living in fragmented habitats. Few studies have investigated what habitat factors determine the probability that primates will occur in habitat fragments or effectiveness of specific conservation measures established to protect them. I analysed forest habitat and colobus population attributes to determine factors that influenced occurrence and abundance of the critically endangered Tana River red colobus in forest fragments in Kenya. The probability of colobus inhabiting forests was positively related to amount of habitat edge and tree species composition. Most of the variance in the density of colobus monkeys was explained by: basal area of food trees, basal area per food tree, density of food trees, and basal area per tree for all trees. Therefore effective primate conservation in fragmented habitats may require strategies that consider different factors that influence both the occurrence of a species and its abundance. To determine whether the Tana River Primate National Reserve is effective in conserving the Tana River red colobus, and by extension other species, I determined whether colobus population attributes or forest habitat attributes differed between inside and outside the reserve. I found no difference in colobus density or mean group size between forests inside and outside the reserve. Forests outside the reserve had significantly higher total basal area of trees, mean basal area per tree, and total basal area of stumps left by trees cut for human use. -

Influence of Plant and Soil Chemistry on Food Selection, Ranging Patterns, and Biomass of Colobus Guereza in Kakamega Forest, Ke
International Journal of Primatology, Vol. 28, No. 3, June 2007 (C 2007) DOI: 10.1007/s10764-006-9096-2 Influence of Plant and Soil Chemistry on Food Selection, Ranging Patterns, and Biomass of Colobus guereza in Kakamega Forest, Kenya Peter J. Fashing,1,2,3,7 Ellen S. Dierenfeld,4,5 and Christopher B. Mowry6 Received February 22, 2006; accepted May 26, 2006; Published Online May 24, 2007 Nutritional factors are among the most important influences on primate food choice. We examined the influence of macronutrients, minerals, and sec- ondary compounds on leaf choices by members of a foli-frugivorous popula- tion of eastern black-and-white colobus—or guerezas (Colobus guereza)— inhabiting the Kakamega Forest, Kenya. Macronutrients exerted a complex influence on guereza leaf choice at Kakamega. At a broad level, protein con- tent was the primary factor determining whether or not guerezas consumed specific leaf items, with eaten leaves at or above a protein threshold of ca. 14% dry matter. However, a finer grade analysis considering the selection ra- tios of only items eaten revealed that fiber played a much greater role than protein in influencing the rates at which different items were eaten relative to their abundance in the forest. Most minerals did not appear to influence leaf choice, though guerezas did exhibit strong selectivity for leaves rich in zinc. Guerezas avoided most leaves high in secondary compounds, though their top food item (Prunus africana mature leaves) contained some of the high- est condensed tannin concentrations of any leaves in their diet. Kakamega 1Department of Science and Conservation, Pittsburgh Zoo and PPG Aquarium, One Wild Place, Pittsburgh, Pennsylvania 15206. -

The Effects of Habitat Disturbance on the Populations of Geoffroy's Spider Monkeys in the Yucatan Peninsula
The Effects of Habitat Disturbance on the Populations of Geoffroy’s Spider Monkeys in the Yucatan Peninsula PhD thesis Denise Spaan Supervisor: Filippo Aureli Co-supervisor: Gabriel Ramos-Fernández August 2017 Instituto de Neuroetología Universidad Veracruzana 1 For the spider monkeys of the Yucatan Peninsula, and all those dedicated to their conservation. 2 Acknowledgements This thesis turned into the biggest project I have ever attempted and it could not have been completed without the invaluable help and support of countless people and organizations. A huge thank you goes out to my supervisors Drs. Filippo Aureli and Gabriel Ramos- Fernández. Thank you for your guidance, friendship and encouragement, I have learnt so much and truly enjoyed this experience. This thesis would not have been possible without you and I am extremely proud of the results. Additionally, I would like to thank Filippo Aureli for all his help in organizing the logistics of field work. Your constant help and dedication to this project has been inspiring, and kept me pushing forward even when it was not always easy to do so, so thank you very much. I would like to thank Dr. Martha Bonilla for offering me an amazing estancia at the INECOL. Your kind words have encouraged and inspired me throughout the past three years, and have especially helped me to get through the last few months. Thank you! A big thank you to Drs. Colleen Schaffner and Jorge Morales Mavil for all your feedback and ideas over the past three years. Colleen, thank you for helping me to feel at home in Mexico and for all your support! I very much look forward to continue working with all of you in the future! I would like to thank the CONACYT for my PhD scholarship and the Instituto de Neuroetología for logistical, administrative and financial support. -

Tana+River+Red+Colobus.Pdf
Tana River Red Colobus Procolobus rufomitratus (Peters, 1879) Kenya (2002, 2004, 2006, 2008) David N. M. Mbora & Thomas M. Butynski Gallery forests found in the lower Tana River, which means that none of the habitat of the Tana Kenya, appear to be remnants of a previously River red colobus and Tana River mangabey is legally continuous forest that extended from Central Africa to protected. Furthermore, habitat loss outside the East Africa 25,000–30,000 years ago). The forests are TRPNR has been exacerbated by the failure of the Tana part of the East African Coastal Forests Biodiversity Delta Irrigation Project’s (TDIP) rice-growing scheme Hotspot and for this, and other reasons, are of great (under the administration of the Tana and Athi Rivers conservation value. In particular, they are the only Development Authority [TARDA], with financing habitat for two endemic primate species; Tana River from Japan International Cooperation Agency [JICA]) red colobus, Procolobus rufomitratus (Peters, 1879), to protect forest patches on their land. Now TARDA is and Tana River mangabey, Cercocebus galeritus Peters, in the process of expanding its activities in the region 1879. These two species inhabit the forests along a 60- by establishing a 110 km2 sugar cane plantation. In km stretch of the lower Tana River from Nkanjonja to addition, a further 500 km2 of land in and around the Mitapani (01°55’S, 40°05’E). All of these forests are delta are earmarked for the development of sugarcane small, ranging in size from <1 ha to c.500 ha. Six other plantations by Mat International Sugar Limited. -

AFRICAN PRIMATES the Journal of the Africa Section of the IUCN SSC Primate Specialist Group
Volume 9 2014 ISSN 1093-8966 AFRICAN PRIMATES The Journal of the Africa Section of the IUCN SSC Primate Specialist Group Editor-in-Chief: Janette Wallis PSG Chairman: Russell A. Mittermeier PSG Deputy Chair: Anthony B. Rylands Red List Authorities: Sanjay Molur, Christoph Schwitzer, and Liz Williamson African Primates The Journal of the Africa Section of the IUCN SSC Primate Specialist Group ISSN 1093-8966 African Primates Editorial Board IUCN/SSC Primate Specialist Group Janette Wallis – Editor-in-Chief Chairman: Russell A. Mittermeier Deputy Chair: Anthony B. Rylands University of Oklahoma, Norman, OK USA Simon Bearder Vice Chair, Section on Great Apes:Liz Williamson Oxford Brookes University, Oxford, UK Vice-Chair, Section on Small Apes: Benjamin M. Rawson R. Patrick Boundja Regional Vice-Chairs – Neotropics Wildlife Conservation Society, Congo; Univ of Mass, USA Mesoamerica: Liliana Cortés-Ortiz Thomas M. Butynski Andean Countries: Erwin Palacios and Eckhard W. Heymann Sustainability Centre Eastern Africa, Nanyuki, Kenya Brazil and the Guianas: M. Cecília M. Kierulff, Fabiano Rodrigues Phillip Cronje de Melo, and Maurício Talebi Jane Goodall Institute, Mpumalanga, South Africa Regional Vice Chairs – Africa Edem A. Eniang W. Scott McGraw, David N. M. Mbora, and Janette Wallis Biodiversity Preservation Center, Calabar, Nigeria Colin Groves Regional Vice Chairs – Madagascar Christoph Schwitzer and Jonah Ratsimbazafy Australian National University, Canberra, Australia Michael A. Huffman Regional Vice Chairs – Asia Kyoto University, Inuyama, -

Christopher B. Mowry
Christopher B. Mowry Berry College (706) 236-1712 Box 430/Department of Biology [email protected] Mt. Berry, GA 30149 http://sites.berry.edu/cmowry/ EDUCATION 1994 Ph.D. and M.S., Ecology, Emory University Atlanta, Georgia 1982 B.A., Biology, Wake Forest University Winston-Salem, North Carolina PROFESSIONAL EXPERIENCE 2001- present Associate Professor of Biology Berry College 2014 Visiting Professor Organization for Tropical Studies, Palo Verde, Costa Rica 2007- 2013 Chairman, Department of Biology Berry College 2010 Visiting Professor University of Dundee, Scotland 2006 – 2008, Graduate Faculty Member 2013 - 2016 University of Tennessee at Chattanooga 1994 - 2001 Assistant Professor of Biology Berry College 1988 - 1992 Teaching Assistant, Department of Biology Emory University 1988 - 1989 Educator Science and Technology Museum of Atlanta (SciTrek) 1982 - 1988 Research Assistant, Department of Pharmacology Emory University TEACHING EXPERIENCE • Principles of Cell Biology (BIO 111) • Behavioral Ecology (BIO 415) • Principle of Zoology (BIO 202) • Conservation Biology (BIO 405) • Advanced Ecology (BIO 455) • Seminar in Biology (BIO 490) • General Biology (for non-majors) (BIO 103) 8/18 - 1 - Mowry CURRENT RESEARCH PROJECTS • Urban wildlife/Atlanta Coyote Project (www.atlantacoyoteproject.org) • Coyote diets in Yellowstone National Park • Sea turtle conservation on St. Catherines Island, GA • Prevalence of chytrid fungal infections in Georgia amphibians PUBLICATIONS Articles Mowry, C.B., C.M. Keene, S.E. Prisland, B.D. Tyler, A.A. Montgomery, A.P. Mowry, R.A. Martin, S. Stevens, J. Ellwanger, and M.B. Morgan (2017) A survey of Batrachochytrium dendrobatidis occurrence in amphibians of Walker and Floyd Counties, Georgia, USA. Herpetological Review 48(4): 777-779. Ganzhorn, J.U., S.J. -

Cercopithecus
Edinburgh Research Explorer New simian immunodeficiency virus infecting De Brazza's monkeys (Cercopithecus neglectus) Citation for published version: Bibollet-Ruche, F, Bailes, E, Gao, F, Pourrut, X, Barlow, KL, Clewley, JP, Mwenda, JM, Langat, DK, Chege, GK, McClure, HM, Mpoudi-Ngole, E, Delaporte, E, Peeters, M, Shaw, GM, Sharp, PM & Hahn, BH 2004, 'New simian immunodeficiency virus infecting De Brazza's monkeys (Cercopithecus neglectus): Evidence for a Cercopithecus monkey virus clade', Journal of Virology, vol. 78, no. 14, pp. 7748-7762. https://doi.org/10.1128/JVI.78.14.7748-7762.2004 Digital Object Identifier (DOI): 10.1128/JVI.78.14.7748-7762.2004 Link: Link to publication record in Edinburgh Research Explorer Document Version: Publisher's PDF, also known as Version of record Published In: Journal of Virology Publisher Rights Statement: Free in PMC. General rights Copyright for the publications made accessible via the Edinburgh Research Explorer is retained by the author(s) and / or other copyright owners and it is a condition of accessing these publications that users recognise and abide by the legal requirements associated with these rights. Take down policy The University of Edinburgh has made every reasonable effort to ensure that Edinburgh Research Explorer content complies with UK legislation. If you believe that the public display of this file breaches copyright please contact [email protected] providing details, and we will remove access to the work immediately and investigate your claim. Download date: 25. Sep. 2021 JOURNAL OF VIROLOGY, July 2004, p. 7748–7762 Vol. 78, No. 14 0022-538X/04/$08.00ϩ0 DOI: 10.1128/JVI.78.14.7748–7762.2004 Copyright © 2004, American Society for Microbiology. -

Factors Influencing Population and Interspecific Interaction of Three Primates in South Nandi Forest, Kenya
FACTORS INFLUENCING POPULATION AND INTERSPECIFIC INTERACTION OF THREE PRIMATES IN SOUTH NANDI FOREST, KENYA Margaret Mwikali Wambua Thesis submitted to the Graduate School in partial fulfillment for the requirements of a Master of Science in Natural Resources Management Degree of Egerton University. EGERTON UNIVERSITY April, 2017 DECLARATION AND APPROVAL DECLARATION I hereby solemnly declare that the work presented in this dissertation is my original work and that to the best of my knowledge; it has not been presented for a degree inthis University or any other University. Signed…………………………... Date………………………….. Margaret Wambua NM11/3637/13 APPROVAL This thesis has been submitted with our approval as University supervisors Signed………………………………. Date……………………………. Dr. George Eshiamwata Department of Natural Resources EGERTON UNIVERSITY Signed………………………………… Date…………………………….. Gilbert O.Obwoyere (Dr.rer.nat) Department of Natural Resources EGERTON UNIVERSITY ii COPYRIGHT ©2016 Wambua M.W No part of this thesis may be reproduced in part or full, sorted in any retrieval system or transmitted in any form or means (electronic, mechanical, photocopying, recording or otherwise) except for short extracts with acknowledgement, without written permission of the author or Egerton University on behalf of the author. All rights reserved iii DEDICATION This work is dedicated to my brothers and sisters Francis Mutuku, Patrick Mutiso, Hellen Mwelu, Elizabeth Nduku, Angela Mumo, Martha Kalondu and Cecilia Muthio for their financial and moral support during the entire period of my study. It is also posthumously dedicated to my late parents (Annunciata Mutono and Peter Wambua) and sister (Stellamaris Ndunge). This thesis is in memory of my mother who always encouraged me to work hard and to always put God first in everything that I do. -

LEAF MONKEY, Trachypithecus Auratus Sondaicus, in the PANGANDARAN NATURE RESERVE, WEST JAVA, INDONESIA
BEHAVIOURAL ECOLOGY OF THE SILVER LEAF MONKEY, Trachypithecus auratus sondaicus, IN THE PANGANDARAN NATURE RESERVE, WEST JAVA, INDONESIA by KAREN MARGARETHA KOOL A dissertation submitted to the University of New South Wales for the degree of Doctor of Philosophy School of Biological Science University of New South Wales Sydney, New South Wales Australia March, 1989 SR PT02 Form 2 RETENTION THE UNIVERSITY OF NEW SOUTH WALES DECLARATION RELATING TO DISPOSITION OF PROJECT REPORT/THESIS This is to certify that I . .1�9.-.C:eo...... MQ;.r.tQ.J:£'\hP..-. .. .J�l.... being a candidate for the degree of 1)!=>.d:o.r.--.of.-.Y.hSos.Of.hy .. am fully aware of the policy of the University relating to the retention and use of higherdegree project reports and theses, namely that the University retains the copies submitted for examination and is free to allow them to be consulted or borrowed. Subject to the provisions of the Copyright Act, 1968, the University may issue a project report or thesis in whole or in part, in photostat or microfilm or other copying medium. In the light of these provisions I declare that I wish to retain my full privileges of copyright and request that neither the whole nor any portion of my project report/thesis be published by the University Librarian and that the Librarian may not authorise the publication of the whole or any part of it, and that I further declare that thispreservation of my copyright privileges shall lapse from the ... :fit.�t ............ day of ...3.o.n ....... 9..r: { ............... 19.�.<:\.................