Fig. S1: Behavioral Results for the High (HP) and Low Probability (LP) Condition
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
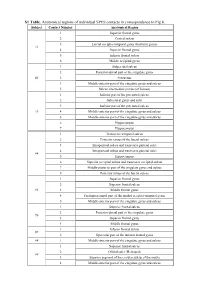
S1 Table. Anatomical Regions of Individual SPES Contacts in Correspondence to Fig 8
S1 Table. Anatomical regions of individual SPES contacts in correspondence to Fig 8. Subject Contact Number Anatomical Region 1 Superior frontal gyrus 2 Central sulcus 3 Lateral occipito-temporal gyrus (fusiform gyrus) #1 4 Superior frontal gyrus 5 Inferior frontal sulcus 6 Middle occipital gyrus 1 Subparietal sulcus 2 Posterior-dorsal part of the cingulate gyrus #2 3 Precuneus 4 Middle-anterior part of the cingulate gyrus and sulcus 5 Sulcus intermedius primus (of Jensen) 1 Inferior part of the precentral sulcus 2 Subcentral gyrus and sulci 3 Inferior part of the precentral sulcus #3 4 Middle-anterior part of the cingulate gyrus and sulcus 5 Middle-anterior part of the cingulate gyrus and sulcus 6 Hippocampus 7 Hippocampus 1 Transverse temporal sulcus 2 Posterior ramus of the lateral sulcus 3 Intraparietal sulcus and transverse parietal sulci 4 Intraparietal sulcus and transverse parietal sulci #4 5 Hippocampus 6 Superior occipital sulcus and transverse occipital sulcus 7 Middle-posterior part of the cingulate gyrus and sulcus 8 Posterior ramus of the lateral sulcus 1 Superior frontal gyrus 2 Superior frontal sulcus #5 3 Middle frontal gyrus 4 Parahippocampal part of the medial occipito-temporal gyrus 5 Middle-anterior part of the cingulate gyrus and sulcus 1 Superior frontal sulcus 2 Posterior-dorsal part of the cingulate gyrus #6 3 Superior frontal gyrus 4 Middle frontal gyrus 1 Inferior frontal sulcus #7 2 Opercular part of the inferior frontal gyrus #8 1 Middle-anterior part of the cingulate gyrus and sulcus 1 Superior frontal sulcus 2 Orbital sulci (H-shaped) #9 3 Superior segment of the circular sulcus of the insula 4 Middle-anterior part of the cingulate gyrus and sulcus . -

Toward a Common Terminology for the Gyri and Sulci of the Human Cerebral Cortex Hans Ten Donkelaar, Nathalie Tzourio-Mazoyer, Jürgen Mai
Toward a Common Terminology for the Gyri and Sulci of the Human Cerebral Cortex Hans ten Donkelaar, Nathalie Tzourio-Mazoyer, Jürgen Mai To cite this version: Hans ten Donkelaar, Nathalie Tzourio-Mazoyer, Jürgen Mai. Toward a Common Terminology for the Gyri and Sulci of the Human Cerebral Cortex. Frontiers in Neuroanatomy, Frontiers, 2018, 12, pp.93. 10.3389/fnana.2018.00093. hal-01929541 HAL Id: hal-01929541 https://hal.archives-ouvertes.fr/hal-01929541 Submitted on 21 Nov 2018 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. REVIEW published: 19 November 2018 doi: 10.3389/fnana.2018.00093 Toward a Common Terminology for the Gyri and Sulci of the Human Cerebral Cortex Hans J. ten Donkelaar 1*†, Nathalie Tzourio-Mazoyer 2† and Jürgen K. Mai 3† 1 Department of Neurology, Donders Center for Medical Neuroscience, Radboud University Medical Center, Nijmegen, Netherlands, 2 IMN Institut des Maladies Neurodégénératives UMR 5293, Université de Bordeaux, Bordeaux, France, 3 Institute for Anatomy, Heinrich Heine University, Düsseldorf, Germany The gyri and sulci of the human brain were defined by pioneers such as Louis-Pierre Gratiolet and Alexander Ecker, and extensified by, among others, Dejerine (1895) and von Economo and Koskinas (1925). -

01 05 Lateral Surface of the Brain-NOTES.Pdf
Lateral Surface of the Brain Medical Neuroscience | Tutorial Notes Lateral Surface of the Brain 1 MAP TO NEUROSCIENCE CORE CONCEPTS NCC1. The brain is the body's most complex organ. LEARNING OBJECTIVES After study of the assigned learning materials, the student will: 1. Demonstrate the four paired lobes of the cerebral cortex and describe the boundaries of each. 2. Sketch the major features of each cerebral lobe, as seen from the lateral view, identifying major gyri and sulci that characterize each lobe. NARRATIVE by Leonard E. WHITE and Nell B. CANT Duke Institute for Brain Sciences Department of Neurobiology Duke University School of Medicine Overview When you view the lateral aspect of a human brain specimen (see Figures A3A and A102), three structures are usually visible: the cerebral hemispheres, the cerebellum, and part of the brainstem (although the brainstem is not visible in the specimen photographed in lateral view for Fig. 1 below). The spinal cord has usually been severed (but we’ll consider the spinal cord later), and the rest of the subdivisions are hidden from lateral view by the hemispheres. The diencephalon and the rest of the brainstem are visible on the medial surface of a brain that has been cut in the midsagittal plane. Parts of all of the subdivisions are also visible from the ventral surface of the whole brain. Over the next several tutorials, you will find video demonstrations (from the brain anatomy lab) and photographs (in the tutorial notes) of these brain surfaces, and sufficient detail in the narrative to appreciate the overall organization of the parts of the brain that are visible from each perspective. -

PDF Hosted at the Radboud Repository of the Radboud University Nijmegen
PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/200480 Please be advised that this information was generated on 2021-10-05 and may be subject to change. REVIEW published: 19 November 2018 doi: 10.3389/fnana.2018.00093 Toward a Common Terminology for the Gyri and Sulci of the Human Cerebral Cortex Hans J. ten Donkelaar 1*†, Nathalie Tzourio-Mazoyer 2† and Jürgen K. Mai 3† 1 Department of Neurology, Donders Center for Medical Neuroscience, Radboud University Medical Center, Nijmegen, Netherlands, 2 IMN Institut des Maladies Neurodégénératives UMR 5293, Université de Bordeaux, Bordeaux, France, 3 Institute for Anatomy, Heinrich Heine University, Düsseldorf, Germany The gyri and sulci of the human brain were defined by pioneers such as Louis-Pierre Gratiolet and Alexander Ecker, and extensified by, among others, Dejerine (1895) and von Economo and Koskinas (1925). Extensive discussions of the cerebral sulci and their variations were presented by Ono et al. (1990), Duvernoy (1992), Tamraz and Comair (2000), and Rhoton (2007). An anatomical parcellation of the spatially normalized single high resolution T1 volume provided by the Montreal Neurological Institute (MNI; Collins, 1994; Collins et al., 1998) was used for the macroscopical labeling of functional studies (Tzourio-Mazoyer et al., 2002; Rolls et al., 2015). In the standard atlas of the human brain by Mai et al. (2016), the terminology from Mai and Paxinos (2012) is used. It contains an extensively analyzed individual brain hemisphere in the MNI- space. -

Supplementary Tables
Supplementary Tables: ROI Atlas Significant table grey matter Test ROI # Brainetome area beta volume EG pre vs post IT 8 'superior frontal gyrus, part 4 (dorsolateral area 6), right', 0.773 17388 11 'superior frontal gyrus, part 6 (medial area 9), left', 0.793 18630 12 'superior frontal gyrus, part 6 (medial area 9), right', 0.806 24543 17 'middle frontal gyrus, part 2 (inferior frontal junction), left', 0.819 22140 35 'inferior frontal gyrus, part 4 (rostral area 45), left', 1.3 10665 67 'paracentral lobule, part 2 (area 4 lower limb), left', 0.86 13662 EG pre vs post ET 20 'middle frontal gyrus, part 3 (area 46), right', 0.934 28188 21 'middle frontal gyrus, part 4 (ventral area 9/46 ), left' 0.812 27864 31 'inferior frontal gyrus, part 2 (inferior frontal sulcus), left', 0.864 11124 35 'inferior frontal gyrus, part 4 (rostral area 45), left', 1 10665 50 'orbital gyrus, part 5 (area 13), right', -1.7 22626 67 'paracentral lobule, part 2 (area 4 lower limb), left', 1.1 13662 180 'cingulate gyrus, part 3 (pregenual area 32), right', 0.9 10665 261 'Cerebellar lobule VIIb, vermis', -1.5 729 IG pre vs post IT 16 middle frontal gyrus, part 1 (dorsal area 9/46), right', -0.8 27567 24 'middle frontal gyrus, part 5 (ventrolateral area 8), right', -0.8 22437 40 'inferior frontal gyrus, part 6 (ventral area 44), right', -0.9 8262 54 'precentral gyrus, part 1 (area 4 head and face), right', -0.9 14175 64 'precentral gyrus, part 2 (caudal dorsolateral area 6), left', -1.3 18819 81 'middle temporal gyrus, part 1 (caudal area 21), left', -1.4 14472 -

Cortical Parcellation Protocol
CORTICAL PARCELLATION PROTOCOL APRIL 5, 2010 © 2010 NEUROMORPHOMETRICS, INC. ALL RIGHTS RESERVED. PRINCIPAL AUTHORS: Jason Tourville, Ph.D. Research Assistant Professor Department of Cognitive and Neural Systems Boston University Ruth Carper, Ph.D. Assistant Research Scientist Center for Human Development University of California, San Diego Georges Salamon, M.D. Research Dept., Radiology David Geffen School of Medicine at UCLA WITH CONTRIBUTIONS FROM MANY OTHERS Neuromorphometrics, Inc. 22 Westminster Street Somerville MA, 02144-1630 Phone/Fax (617) 776-7844 neuromorphometrics.com OVERVIEW The cerebral cortex is divided into 49 macro-anatomically defined regions in each hemisphere that are of broad interest to the neuroimaging community. Region of interest (ROI) boundary definitions were derived from a number of cortical labeling methods currently in use. Protocols from the Laboratory of Neuroimaging at UCLA (LONI; Shattuck et al., 2008), the University of Iowa Mental Health Clinical Research Center (IOWA; Crespo-Facorro et al., 2000; Kim et al., 2000), the Center for Morphometric Analysis at Massachusetts General Hospital (MGH-CMA; Caviness et al., 1996), a collaboration between the Freesurfer group at MGH and Boston University School of Medicine (MGH-Desikan; Desikan et al., 2006), and UC San Diego (Carper & Courchesne, 2000; Carper & Courchesne, 2005; Carper et al., 2002) are specifically referenced in the protocol below. Methods developed at Boston University (Tourville & Guenther, 2003), Brigham and Women’s Hospital (McCarley & Shenton, 2008), Stanford (Allan Reiss lab), the University of Maryland (Buchanan et al., 2004), and the University of Toyoma (Zhou et al., 2007) were also consulted. The development of the protocol was also guided by the Ono, Kubik, and Abernathy (1990), Duvernoy (1999), and Mai, Paxinos, and Voss (Mai et al., 2008) neuroanatomical atlases. -

Not for Reprintlaboratory INVESTIGATION Anterior Peri-Insular Quadrantotomy: a Cadaveric White Matter Dissection Study
Authors: Please review all authors’ names, academicLow-resolution degrees, affiliations, and PDF contributions, as well as the Disclosure, MS# 19-472, read before authors for spelling and accuracy. Not for reprintLABORATORY INVESTIGATION Anterior peri-insular quadrantotomy: a cadaveric white matter dissection study *Pablo Gonzalez-Lopez, MD, PhD,1 Giulia Cossu, MD,2 Etienne Pralong, MD,2 Matias Baldoncini, MD,3 Mahmoud Messerer, MD, MSc,2,4 and Roy Thomas Daniel, MD, MCh2,4 1Department of Neurosurgery, Hospital General Universitario Alicante, Spain; 2Department of Neurosurgery, University Hospital of Lausanne, Switzerland; 3Department of Neurological Surgery, San Fernando Hospital, Buenos Aires, Argentina; and 4Faculty of Medicine and Biology, University of Lausanne, Switzerland OBJECTIVE Anterior quadrant disconnection represents a safe surgical option in well-selected pediatric patients with a large frontal lobe lesion anterior to the motor cortex. The understanding of the anatomy of the white matter tracts con- necting the frontal lobe with the rest of the cerebrum forms the basis of a safe and successful disconnective surgery. The authors explored and illustrated the relevant white matter tracts sectioned during each surgical step using fiber dissection techniques. METHODS Five human cadaveric hemispheres were dissected to illustrate the frontal connections in the 3 planes. The dissections were performed from lateral to medial, medial to lateral, and ventral to dorsal to describe the various tracts sectioned during the 4 steps of this surgery, namely the anterior suprainsular window, intrafrontal disconnection, anterior callosotomy, and frontobasal disconnection. RESULTS At the beginning of each surgical step, the U fibers were cut. During the anterior suprainsular window, the superior longitudinal fasciculus (SLF), the uncinate fasciculus, and the inferior fronto-occipital fasciculus (IFOF) were vi- sualized and sectioned, followed by sectioning of the anterior limb of the internal capsule. -

Normal Cortical Anatomy
Normal Cortical Anatomy MGH Massachusetts General Hospital Harvard Medical School NORMAL CORTICAL ANATOMY • Sagittal • Axial • Coronal • The Central Sulcus NP/MGH Sagittal Neuroanatomy NP/MGH Cingulate sulcus Superior frontal gyrus Marginal ramus of Cingulate sulcus Cingulate gyrus Paracentral lobule Superior parietal lobule Parietooccipital sulcus Cuneus Calcarine sulcus Lingual gyrus Subcallosal gyrus Gyrus rectus Fastigium, fourth ventricle NP/MGH Superior frontal gyrus Cingulate sulcus Precentral gyrus Marginal ramus of Cingulate gyrus Central sulcus Cingulate sulcus Superior parietal lobule Precuneus Parietooccipital sulcus Cuneus Calcarine sulcus Frontomarginal gyrus Lingual gyrus Caudothallamic groove Gyrus rectus NP/MGH Precentral sulcus Central sulcus Superior frontal gyrus Marginal ramus of Corona radiata Cingulate sulcus Superior parietal lobule Precuneus Parietooccipital sulcus Calcarine sulcus Inferior occipital gyrus Lingual gyrus NP/MGH Central sulcus Superior parietal lobule Parietooccipital sulcus Frontopolar gyrus Frontomarginal gyrus Superior occipital gyrus Middle occipital gyrus Medial orbital gyrus Lingual gyrus Posterior orbital gyrus Inferior occipital gyrus Inferior temporal gyrus Temporal horn, lateral ventricle NP/MGH Central sulcus Superior Temporal gyrus Middle Temporal gyrus Inferior Temporal gyrus NP/MGH Central sulcus Superior parietal gyrus Inferior frontal gyrus Frontomarginal gyrus Anterior orbital gyrus Superior occipital gyrus Middle occipital Posterior orbital gyrus gyrus Superior Temporal gyrus Inferior -

Dynamic Mapping of Human Cortical Development During Childhood Through Early Adulthood
Dynamic mapping of human cortical development during childhood through early adulthood Nitin Gogtay*†, Jay N. Giedd*, Leslie Lusk*, Kiralee M. Hayashi‡, Deanna Greenstein*, A. Catherine Vaituzis*, Tom F. Nugent III*, David H. Herman*, Liv S. Clasen*, Arthur W. Toga‡, Judith L. Rapoport*, and Paul M. Thompson‡ *Child Psychiatry Branch, National Institutes of Mental Health, National Institutes of Health, Bethesda, MD 20892; and ‡Laboratory of Neuro Imaging, Department of Neurology, University of California School of Medicine, Los Angeles, CA 90095-1769 Communicated by Leslie G. Ungerleider, National Institutes of Health, Bethesda, MD, April 15, 2004 (received for review January 7, 2004) We report the dynamic anatomical sequence of human cortical temporal cortex, which contains association areas that integrate gray matter development between the age of 4–21 years using information from several sensory modalities, matured last. Fur- quantitative four-dimensional maps and time-lapse sequences. thermore, the maturation of the cortex also appeared to follow Thirteen healthy children for whom anatomic brain MRI scans were the evolutionary sequence in which these regions were created. obtained every 2 years, for 8–10 years, were studied. By using models of the cortical surface and sulcal landmarks and a statistical Methods model for gray matter density, human cortical development could Subjects. Sample demographics are shown in Table 1. All subjects be visualized across the age range in a spatiotemporally detailed were recruited from the community for an ongoing National time-lapse sequence. The resulting time-lapse ‘‘movies’’ reveal that Institute of Mental Health study of human brain development (i) higher-order association cortices mature only after lower-order (20). -

Supplementary Materials
Supplementary Materials. Holt et al. 2009. Schizophrenia Bulletin Supplementary Table 1: Example stimuli. Table S1. Example Stimuli. First sentence Second sentence Neutral Positive Negative (without the critical word) critical word critical word critical word Nancy’s son ended up just He was already a ____ by husband millionaire criminal like his father. age 25. Stephen owned a lot of Everyone knew that he ____ bought loved forged nineteenth century art. paintings of old masters. An unfamiliar man rang He had come to _____ her. register congratulate arrest Lenora's doorbell one day. Mr. Jenners planned to The reason for this was obvious reassuring hidden move his family to New _____ to the children. York. Cheryl's baby cried when She quieted him with pacifier lullaby drug she took him to bed. a ____ that night. Two-sentence descriptions of social situations for each of three experimental conditions (neutral, positive and negative), were constructed. For each pair of sentences, the first sentence was neutral and ambiguous in content. The emotional meaning of the sentence- pair was conferred by a positively-valenced, negatively-valenced or neutral word (the critical word) in the second sentence. Thus, other than one valence-associated or neutral word in the second sentence, the three conditions were identical in word content. The critical words of each condition were matched with respect to mean number of letters, frequency, and abstractness, but differed according to their affective valence and arousal. Norming studies in participants who did not participate in the fMRI study confirmed that the three conditions systematically differed according to their emotional valence (ratings on a 1-7 Likert scale: positive sentence pairs: mean valence rating > 5; negative sentence pairs: mean valence rating < 3; neutral sentence pairs: mean valence rating >3 and < 5) 1. -

Reorganization of the Neurobiology of Language After Sentence Overlearning
Sentence overlearning 1 Reorganization of the neurobiology of language after sentence overlearning 1* 1,2 3 1 4 Jeremy I Skipper , Sarah Aliko , Stephen Brown , Yoon Ju Jo , Serena Lo , Emilia 5 1,6 Molimpakis and Daniel R Lametti 1 E xperimental Psychology, University College London, UK 2 L ondon Interdisciplinary Biosciences Consortium, University College London, UK 3 N atural Sciences, University College London, UK 4 S peech and Language Sciences, University College London, UK 5 W ellcome Centre for Human Neuroimaging, University College London, UK 6 D epartment of Psychology, Acadia University, Nova Scotia, Canada * C orresponding author, [email protected] Supplementary Material Figure Captions Figure S1. Additional novel listening linear mixed-effects model results. General linear test contrasting novel sentences from session one (blues) and two (reds) in the left (LH) and right hemispheres (RH) presented on lateral (top) and medial (bottom) surface views. The colour bar represents z-scores and the images are thresholded at an alpha (α) level of p < .01, corrected for multiple comparisons. Figure S2. Additional overlearned sentence listening linear mixed-effects model results. A) Session one general linear test (GLT) for sentence; B) Session two GLT for sentence; C) Direct contrast of session one sentences (blues) with session two sentences (reds). The top two rows are the left hemisphere (LH) while the bottom two are the right hemispheres (RH) lateral and medial surface views. The colour bar represents z-scores and the images are thresholded at an alpha (α) level of p < .01, corrected for multiple comparisons. Figure S3. Additional overlearned minus novel sentence listening linear mixed-effects model results. -

Mapping Human Cortical Areasin
The Journal of Neuroscience, August 10, 2011 • 31(32):11597–11616 • 11597 Behavioral/Systems/Cognitive Mapping Human Cortical Areas In Vivo Based on Myelin Content as Revealed by T1- and T2-Weighted MRI Matthew F. Glasser and David C. Van Essen Department of Anatomy and Neurobiology, Washington University School of Medicine, St. Louis, Missouri 63110 Noninvasively mapping the layout of cortical areas in humans is a continuing challenge for neuroscience. We present a new method of mapping cortical areas based on myelin content as revealed by T1-weighted (T1w) and T2-weighted (T2w) MRI. The method is general- izable across different 3T scanners and pulse sequences. We use the ratio of T1w/T2w image intensities to eliminate the MR-related image intensity bias and enhance the contrast to noise ratio for myelin. Data from each subject were mapped to the cortical surface and aligned across individuals using surface-based registration. The spatial gradient of the group average myelin map provides an observer- independent measure of sharp transitions in myelin content across the surface—i.e., putative cortical areal borders. We found excellent agreement between the gradients of the myelin maps and the gradients of published probabilistic cytoarchitectonically defined cortical areas that were registered to the same surface-based atlas. For other cortical regions, we used published anatomical and functional information to make putative identifications of dozens of cortical areas or candidate areas. In general, primary and early unimodal association cortices are heavily myelinated and higher, multimodal, association cortices are more lightly myelinated, but there are notable exceptions in the literature that are confirmed by our results.