Chemicals of Low Concern for Human Health Based on an Initial Assessment of Hazards
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

4월 24일(목), 09:00~10:30
포스터 I: 4월 24일(목), 09:00~10:30 공업화학: 4월 24일(목), 09:00 ~ 10:30 좌장: 박정훈(동국대) Cleaning system study with mounting activated species of solution using P공업목-1(118) (강원대)권흥수, 이원규 atmospheric pressure plasma 기공구조 조절 제올라이트 촉매를 이용한 액체연료의 흡열량 향상 (고려대)현동훈, 김중연, 김유리, 전병희, P공업목-2(118) (우수 포스터 발표상 후보) 김성현, (국방과학(연))정병훈, 한정식 (고려대)신동건, 김성현, 이창훈, P공업목-3(118) 열 안정성이 우수한 CO 흡착제용 다공성 유무기 복합 지지체 제조 2 조동현, 정현철 (고려대)김유리, 김성현, 전병희, P공업목-4(119) 분산제의 구조에 따른 Jet A-1 연료의 열산화반응 탄소침적물 감소 연구 김중연, 현동훈 (국방과학(연))한정식, 정병훈 Effect of SAA Pretreatment on Temperature of Enzymatic Hydrolysis Using P공업목-5(119) (경기대)장서윤, 박장한, 김준석 Lignocellulosic Biomass Pretreatment and fractionation of Helianthus tuberosus residue by flow-through P공업목-6(119) (경기대)박용철, 박장한, 김준석 reaction (순천대)오성준 (순천대)양지선, 박권필 P공업목-7(119) Carbon Felt 산처리에 의한 바나듐 레독스 흐름전지에 대한 영향 ((주)CNL Energy)나일채, 이정훈 ((주)ETIS)추천호 메틸카바메이트 및 메탄올을 이용한 디메틸카보네이트 제조를 위한 고활성 P공업목-8(120) (한양대)이재홍, 서영웅 촉매 조사 (우수 포스터 발표상 후보) 연속흐름반응기에서 고농도 NaBH 가수분해 반응의 촉매 종류에 따른 P공업목-9(120) 4 (순천대)오성준, 정현승, 박권필 수소발생연구 (순천대)안명원, 곽동영, 복영빈, 박권필 P공업목-10(120) 해조류 종류별 후코산틴 추출 및 염분세척 ((주) ETIS)추천호, 김영숙 (순천대)이세훈, 박권필, ((주)ETIS)김영숙, P공업목-11(120) 효소연료전지에서 Anode 조건에 따른 OCV 변화 추천호, ((주)CNL Energy)나일채 (순천대)정재현, 정회범, 박권필 P공업목-12(121) PEMFC MEA제법에 의한 성능 및 내구성 향상 ((주)CNL Energy)라일채, 이 호 (순천대)정재현, 신은경, 정회범, 박권필 P공업목-13(121) PEM 수전해 과정에서 전극과 전해질 막의 변화 ((주)CNL Energy)라일채, 이 호 (순천대)정재현, 박권필 P공업목-14(121) 직접 개미산 연료전지(DFAFC)의 구동 조건에 따른 성능 비교 ((주)CNL Energy)나일채 (순천대)정재진, 박권필 P공업목-15(121) 가속화도 4.0의 전극, 막, MEA 가속화 기법 개발 (현대자동차)안병기, 김세훈, 고재준 (순천대)정재진, 박권필 P공업목-16(122) 탄화수소막을 사용한 -

Here the Nonspecific Term Cupreous Metals Is Used for Copper and the Alloys Such As Brass and Bronze Where Copper Predominates
CONSERVAnON OF CUPREOUS METALS Here the nonspecific term cupreous metals is used for copper and the alloys such as brass and bronze where copper predominates. In general it matters little what the specific alloy is, for they are usually treated in the same way. Care needs to be taken only when there is a high percentage of lead or tin, both of which are amphoteric metals and dissolve in alkaline solutions. There are a considerable number of chemical treatments for copper, bronze and brass, and most are not satisfactory for cupreous metals from marine sites. Consult the bibliography at the end of this section for further information. In a marine environment the two most commonly encountered copper corrosion products are cuprous chloride and cuprous sulfide. However, the mineral alterations in the copper alloys are more complex than those of just copper. Once any cupreous object is recovered and exposed to the air, it continues to corrode by a process referred to as bronze disease. Cuprous cblorides in the presence of moisture and oxygen are hydrolyzed to form hydrochloric acid and basic cupric chloride. The hydrochloric acid in turn attacks thc uncorroded metal to form more cuprous chloride. The reaction continues until no metal remains. Any conservation of chloride contaminated cupreous objects requires that I) the cuprous chlorides be removed, 2) the cuprous chlorides be converted to harmless cuprous oxide, or 3) the chemical action of the chlorides be prevented. Cbemical Cleaning Tecbniques I. ACIDS All acid treatments require a solid metal core. All acids can and will strip off corrosion layers down to bare metal and many will even etch the barc metal. -

Preparing to Manufacture Hydrogen Peroxide
PREPARING TO MANUFACTURE HYDROGEN PEROXIDE Part of the Hydrogen Peroxide Propulsion Guide The early laboratory preparation of hydrogen peroxide was based on the technique that Thenard used during the initial preparation of hydrogen peroxide. In this technique, barium nitrate, purified by recrystallization, was decomposed by heating in air in a porcelain retort. The resulting oxide was further oxidized by heating in a stream of oxygen to a dull red heat. The barium peroxide which formed was then dampened, ground, and dissolved in hydrochloric acid (nitric acid was used in Thenard’s initial experiments). A slight excess of sulfuric acid was then added to precipitate barium sulfate and regenerate hydrochloric acid. The procedure of barium peroxide solution and sulfate precipitation was repeated several times in the same solution to increase the peroxide concentration (concentrations of up to 33 percent by weight hydrogen peroxide could be achieved in this manner). The concentrated solution containing water, hydrogen peroxide, and hydrochloric acid, along with accumulated impurities, was cooled with ice and saturated with barium peroxide; iron and manganese impurities in the solution were then precipitated out as phosphates. The hydrochloric acid was removed by the addition of silver sulfate and the sulfate ion was removed by the subsequent addition of barium oxide. Further concentration was accomplished by vacuum distillation until “no further density increase occurs.” Thenard reported that 100 w/o hydrogen peroxide (on the basis of density data and the measurement of the volume of oxygen released) could be obtained by this technique. The first record of commercial production of hydrogen peroxide appeared in the 1865 to 1875 period. -

Hydrite General Product Flyer
PRODUCT OFFERING forms, trade names and a variety of certifi cations. Please call us about your specifi c chemical requirements. PRODUCTS A-Z Acetic Acid Calcium Chloride Calcium Dioctyl Phthalate Glycol Ether DPM Reduction Chemicals Acetone Hydroxide (Lime) Calcium Dipotassium Phosphate Glycol Ether EB Liquid Inorganic Salts Aluminum Brite Dips Hypochlorite Calcium (DKP) Glycol Ether EE Magnesium Bisulfi te Aluminum Chlorhydrate Phosphates Dipropylene Glycol Glycol Ether EE-AC Magnesium Chloride Aluminum Sulfate (Alum) Carboxymethyl Cellulose Disodium Phosphate Glycol Ether EM Magnesium Hydroxide Ammonium Bicarbonate Caustic Potash Dodecylbenzenesulfonic Glycol Ether EP Magnesium Oxide Ammonium Bifl uor ide Caustic Soda Acid (DDBSA) Glycol Ether PM Magnesium Phosphate Ammonium Bisulfi te Chelants Dye Fixatives Glycol Ether PM-AC Magnesium Sulfate Ammonium Chloride Chlorine Epoxy Resins HAN, Heavy Aromatic Magnesium Sulfi te Ammonium Hydroxide Chromic Acid Ethyl Acetate Naphtha Metal Finishing Products (Aqua Ammonia) Citric Acid Ethyl Alcohol Heat Transfer Fluids Methanol Ammonium Persulfate Copper Carbonate Ethylene Diamine Tetra Heptane Methyl Amyl Ketone Ammonium Phosphates Copper Cyanide Acetic Acid (EDTA) Hexane Methyl Ethyl Ketone Ammonium Sulfate Copper Sulfate Ethylene Dichloride Hexylene Glycol Methyl Isobutyl Ketone Ammonium Sulfi te Cyclohexane Ethylene Glycol HTH Methylene Chloride Mineral Anhydrous Ammonia Cyclohexanone Felt & Wire Cleaners Hydrochloric Acid Fillers Anodizing Chemicals Dairy Cleaners Ferric Chloride Hydrofluoric Acid -

Most Common Substances in Chemicals for Different Industries
Table 14 Most common substances by industry Figures for 2009. Substances contained in more than 10 products. A substance can be omitted for secrecy reasons. Industry CAS-no Substance Number of products A01 Agriculture 7732-18-5 Water 458 6484-52-2 Nitric acid ammonium salt 77 57-55-6 1,2-Propanediol 75 64742-65-0 Distillates (petroleum), solvent-dewaxed heavy 74 paraffinic 63148-62-9 Siloxanes and Silicones, di-Me 60 57-13-6 Urea 54 67-63-0 2-Propanol 54 11138-66-2 Xanthan gum 53 7783-20-2 Sulfuric acid diammonium salt 53 7722-76-1 Phosphoric acid, monoammonium salt 52 7487-88-9 Sulfuric acid magnesium salt (1:1) 51 7447-40-7 Potassium chloride 46 56-81-5 1,2,3-Propanetriol 44 7778-80-5 Sulfuric acid dipotassium salt 43 1310-73-2 Sodium hydroxide 41 7647-14-5 Sodium chloride 41 2634-33-5 1,2-Benzisothiazol-3(2H)-one 38 1310-58-3 Potassium hydroxide 33 7757-79-1 Nitric acid potassium salt 33 7785-87-7 Manganese sulfate 32 7664-38-2 Phosphoric acid 32 72623-87-1 Lubricating oils, petroleum, C20-50, 31 hydrotreated neutral oil-based 64741-89-5 Distillates (petroleum), solvent-refined light 29 paraffinic 77-92-9 1,2,3-Propanetricarboxylic acid, 2-hydroxy- 28 8061-51-6 Lignosulfonic acid, sodium salt 28 7778-18-9 Sulfuric acid, calcium salt (1:1) 27 64-17-5 Ethanol 27 14807-96-6 Talc 26 64742-94-5 Solvent naphtha (petroleum), heavy arom. 25 68649-42-3 Phosphorodithioic acid, O,O-di-C1-14-alkyl 24 esters, zinc salts (2:1) 64-02-8 Glycine, N,N'-1,2-ethanediylbis[N- 24 (carboxymethyl)-, tetrasodium salt 64-19-7 Acetic acid 23 99-76-3 Benzoic acid, -
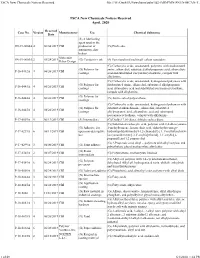
TSCA New Chemicals Notices Received File:///W:/Oneepa/Newchems/Pubs/5D2-IMD/PMN-SNUN-MCAN-T
TSCA New Chemicals Notices Received file:///W:/OneEPA/Newchems/pubs/5d2-IMD/PMN-SNUN-MCAN-T... TSCA New Chemicals Notices Received April, 2020 Received Case No. Version Manufacturer Use Chemical Substance Date (S) A lubricating agent used in the SN-19-0004A 4 06/04/2019 CBI production of (G) Pitch coke automotive disc brakes Molecular SN-19-0005A 2 05/28/2019 (G) Conductive ink (S) Functionalized multiwall carbon nanotubes Rebar Design (G) Carboxylic acids, unsaturated, polymers with disubstituted (G) Polymer for amine, alkanediol, substituted alkylpropanoic acid, alkanedioic P-16-0442A 4 06/26/2019 CBI coatings acid and substituted isocyanatocycloalkane, compds with alkylamine (G) Carboxylic acids, unsaturated, hydrogenated polymers with (G) Polymer for disubstituted amine, alkanediol, substituted alkylpropanoic P-16-0443A 4 06/26/2019 CBI coatings acid, alkanedioic acid and substituted isocyanatocycloalkane, compds with alkylamine (G) Polymer for P-16-0444A 4 06/26/2019 CBI (G) Amine salted polyurethane coatings (G) Carboxylic acids, unsaturated, hydrogenated polymers with (G) Polymer for substituted alkanediamine, alkanediol, substituted P-16-0445A 4 06/26/2019 CBI coatings alkylpropanoic acid, alkanedioic acid and substituted isocyanatocycloalkane, compds with alkylamine P-17-0007A 5 06/13/2019 CBI (S) Intermediate (G) Dialkyl 7,10-dioxa, dithiahexadeca diene (G) Substituted carboxylic acid, polymer with 2,4-diisocyanato- (G) Adhesive for 1-methylbenzene, hexanedioic acid, alpha-hydro-omega- P-17-0239A 6 06/11/2019 CBI open non-descriptive -

Bronze Disease: Understanding, Curing, and Preventative Treatment Jason Sanchez Associate, American Numismatic Society Member, Crescent City Coin Club
Bronze Disease: Understanding, Curing, and Preventative Treatment Jason Sanchez Associate, American Numismatic Society Member, Crescent City Coin Club Dr. Ken Harl, Ph.D Tulane University History Department Fellow and Trustee, American Numismatic Society Agenda Bronze Disease: Understanding, Curing and Preventative Treatment ¨What is Bronze Disease? ¨Do I have a Patina, or Bronze Disease? ¨If I have Bronze Disease, what do I do? ¨Long-term care: serious storage concerns 1 What is Bronze? Before we find out about Bronze Disease, first, the more important question: what is Bronze? ¨ An alloy of Copper (CU) of more than 90% purity in conjunction with other metals (usually tin, antimony and zinc) ¨ One of the first metals used by man – “Bronze Age” man dated from c. 3,000 BCE in Asia Minor, but the use of bronze is noted in some parts of Asia (Chiang Mai, Thailand and Sanxingdui, China) by c. 4,500 BCE ¨ American Pre-Columbian use of Bronze may have begun by 1000 CE ¨ Bronze was one of the first metals that was used for coinage ¨ Bronze coinage by far outstrips silver or gold as a constituent of the money supply for most countries up to the modern age 2 What is Bronze Disease? ¨Bronze “disease” is a condition in which the coin produces acid (normally hydrochloric or hydrosulfuric acid) internally, and begins to disintegrate ¨The exterior usually exhibits green or brown “growths” that cover the pitting that acids will create ¨Because these growths were originally believed to be caused by a bacteria, the condition became known as bronze “disease” -

SAFETY DATA SHEET Sodium Persulfate
SAFETY DATA SHEET Sodium Persulfate SDS # : 7775-27-1 Revision date: 2021-02-17 Format: NA Version 1.06 1. PRODUCT AND COMPANY IDENTIFICATION Product Identifier Product Name Sodium Persulfate CAS-No 7775-27-1 Synonyms Sodium Peroxydisulfate; Disodium Peroxydisulfate; Peroxydisulfuric acid, disodium salt; Peroxydisulfuric acid, sodium salt. Recommended use of the chemical and restrictions on use Recommended Use: Polymerization initiator; Etchant and cleaner for printed circuit boards; Hair bleaching formulations; Secondary oil recovery; Oxidizing agent for a variety of organic reactions. Restrictions on Use No uses to be advised against were identified. Manufacturer/Supplier PeroxyChem LLC 2005 Market Street Suite 3200 Philadelphia, PA 19103 Phone: +1 267/ 422-2400 (General Information) E-Mail: [email protected] Emergency telephone numbers For leak, fire, spill or accident emergencies, call: 1 800 / 424 9300 (CHEMTREC - U.S.A.) 1 703 / 527 3887 (CHEMTREC - Collect - All Other Countries) +1 303/ 389-1409 (Medical - U.S. - Call Collect) Page 1 / 10 Sodium Persulfate SDS # : 7775-27-1 Revision date: 2021-02-17 Version 1.06 2. HAZARDS IDENTIFICATION Classification OSHA Regulatory Status This material is considered hazardous by the OSHA Hazard Communication Standard (29 CFR 1910.1200) Acute toxicity - Oral Category 4 Skin corrosion/irritation Category 2 Serious eye damage/eye irritation Category 2B Respiratory sensitization Category 1 Skin sensitization Category 1 Specific target organ toxicity (single exposure) Category 3 Oxidizing Solids -

Monitoring the Conservation Treatment of Corroded Cupreous Artefacts: the Use of Electrochemistry and Synchrotron Radiation Based Spectroelectrochemistry
FACULTY OF SCIENCE Department of Analytical Chemistry Monitoring the conservation treatment of corroded cupreous artefacts: The use of electrochemistry and synchrotron radiation based spectroelectrochemistry Thesis submitted in fulfilment of the requirements for the degree of Doctor in Science, Chemistry by Karen Leyssens Promoter: Prof. Dr. A. Adriaens Co-promoter: Dr. C. Degrigny Ghent 2006 Woord van Dank Men kan misschien wel onderzoek voeren op z’n eentj e, maar men geraakt zoveel verder met de hulp van anderen. Daarom zou ik langs deze weg graag mijn collega’s, vrienden en familie willen bedanken voor hun voortdurende steun tijdens het voltooien van dit doctoraat. Prof. Dr. A. Adriaens wens ik te bedanken voor de kans dei ze me gaf om te doctoreren, voor haar vertrouwen en de begeleiding gedurende deze 4 jaar. In het bijzonder de contacten en internationale samenwerkingen die zij tot stand bracht waren van enorme waarde voor dit werk. In this respect, I have to start with thanking Dr. C. Degrigny for suggesting several subjects of interest in the area of conservation science and even more for the discussions and support in the years thereafter. I certainly have to thank Prof. Dr. M. Dowsett from the University of Warwick for all the effort and time he invested in the design of the in situ cell and the related software, his enthusiasm during each of the measuring sessions and the hours of discussion for interpretating the XAS results. The second part of this work would not have been possible without him. Of course I should not forget all the other people involved in the synchrotron sessions for their dedication to make a succes out of each measurement. -

19650003847.Pdf
NATIONAL BUREAU OF STANDARDS REPO RT 8595 PRELIMINARY REPORT ON A SURVEY OF THERMODYNAMIC PROPERTIES OF THE COMPOUNDS OF THE ELEMENTS CHNOPS i N65 2 CACC£S$IO NUMB£:R) CTHRU) ~ ___f£ / I- (PAGES) (CdOEI ::; ~ CIU - d -P?02c2/ :33 (NASA CR OR TMX OR AD NUMBER) (CATEQORY) Progress Report for the Period 1 August to 31 October 1964 to National Aeronautics and Space Administration GPO PRICE $ _____ OTS PRICE(S) $ 1 November 1964 Hard copy (He) of· cJV r Microfiche (MF) __....;... : .:::.j~I?J~_ <@> U.S. DEPARTMENT OF COMMERCE NATIONAL BUREAU OF STANDARDS THE NATIONAL BUREAU OF STANDARD S The ational Bureau of Standards is a principal focal point in the Federal Government for assuring maximum application of the physical and engineering sciences to the advancement of technology in industry and commerce. It responsibilities include development and main tena nce of the national stand· ards of measurement, and the provisions of means for making measurements consi tent with those standard ; determination of physical constants and properties of materials; development of methods for testing materials, mechanisms, and structures, and making such tests as may be nece sary, particu larl: for ~overn ment agencies; cooperation in the establi hment of standard practices for incorpora tion in codes and specifications; advisory service to government agencies on scientific and technical problems; invention and development of device to serve special needs of the Government: assi tance to indus! r). business. and consumers in the development and acceptance of commercial tandard and simplified trade practice recommendations; administration of programs in cooperation with United tates bu iness groups and standards organizations for the development of international standard of practice; and maintenance of a clearinghouse for the collection and dis emination of scientific, tech nical. -

Safety Assessment of Simple Carbonate Salts As Used in Cosmetics
Safety Assessment of Simple Carbonate Salts as Used in Cosmetics Status: Tentative Report for Public Comment Release Date: June 16, 2016 Panel Date: September 26-27, 2016 All interested persons are provided 60 days from the above date to comment on this safety assessment and to identify additional published data that should be included or provide unpublished data which can be made public and included. Information may be submitted without identifying the source or the trade name of the cosmetic product containing the ingredient. All unpublished data submitted to CIR will be discussed in open meetings, will be available at the CIR office for review by any interested party and may be cited in a peer-reviewed scientific journal. Please submit data, comments, or requests to the CIR Director, Dr. Lillian J. Gill. The 2016 Cosmetic Ingredient Review Expert Panel members are: Chair, Wilma F. Bergfeld, M.D., F.A.C.P.; Donald V. Belsito, M.D.; Ronald A. Hill, Ph.D.; Curtis D. Klaassen, Ph.D.; Daniel C. Liebler, Ph.D.; James G. Marks, Jr., M.D.; Ronald C. Shank, Ph.D.; Thomas J. Slaga, Ph.D.; and Paul W. Snyder, D.V.M., Ph.D. The CIR Director is Lillian J. Gill, D.P.A. This report was prepared by Wilbur Johnson, Jr., M.S., Senior Scientific Analyst and Bart Heldreth, Ph.D., Chemist. © Cosmetic Ingredient Review 1620 L STREET, NW, SUITE 1200 ◊ WASHINGTON, DC 20036-4702 ◊ PH 202.331.0651 ◊ FAX 202.331.0088 ◊ [email protected] ABSTRACT: The Cosmetic Ingredient Review (CIR) Expert Panel (Panel) reviewed the safety of 6 simple carbonate salts which function as absorbents, bulking agents, opacifying agents, pH adjusters, buffering agents, abrasives, and oral care agents in cosmetic products. -

Air Permit Application for Dry Sorbent and Activated Carbon Injection Units 5R4 and Sr6
AL) CJFZX AIR PERMIT APPLICATION FOR DRY SORBENT AND ACTIVATED CARBON INJECTION UNITS 5R4 AND SR6 SCHILLER STATION PORTSMOUTH, NEW HAMPSHIRE PREPARED FOR: Public Service Company of New Hampshire Manchester, New Hampshire PREPARED BY: GZA GeoEnvironmental, Inc. Manchester, New Hampshire February 2014 File No. 04.0029995.00 Copyright © 2014 GZA GeoEnvironmental, Inc. Public Service PSNH Energy Park of New Hampshire 780 North Commercial Street, Manchester, NH 03101 A Northeast lJiilities Company Pul)lic Service Company of New Hampshire P.O. Box 330 Manchester, NH 03105-0330 February 27, 2014 (603) 634-2851 [email protected] D32795 1ECEIVEO 7J HAMPSHIRE Mr. Craig Wright, Acting Director , William H. Smagula, P.E. Air Resources Division EB 2 / 2014 Vice President - Generation NH Dept. of Environmental Services 29 Hazen Drive, P0 Box 95 AIR OURCES Di VISION Concord, NH 03302-0095 Re: Public Service Company of New Hampshire Schiller Station - Temporary Permit Application for Installation of Dry Sorbent and Activated Carbon Injection Emission Control Technology Dear Mr. Wright: Public Service Company of New Hampshire (PSNH) submits the enclosed temporary permit application for the installation of dry sorbent (DSI) and activated carbon injection (ACI) emission control technologies on Schiller Station Units SR4 and SR6. The installation of the DSL'ACI systems will be used in conjunction with existing control technologies to meet the requirements of 40 CFR 63, Subpart UUUUU and National Emission Standards for Hazardous Air Pollutant: Coal and Oil Fired Electric Utility Steam Generating Units (Mercury and Air Toxics Standard-MATS). The proposed DSI and ACI systems are intended to reduce the emissions of acid gases (hydrogen chloride and hydrogen fluoride) and mercury, and are anticipated to also reduce emissions of sulfur dioxide and sulfuric acid mist.