Bats As Bushmeat: a Global Review S Imon M Ickleburgh,Kerry W Aylen and P Aul R Acey
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Recent Bat Survey Reveals Bukit Barisan Selatan Landscape As A
A Recent Bat Survey Reveals Bukit Barisan Selatan Landscape as a Chiropteran Diversity Hotspot in Sumatra Author(s): Joe Chun-Chia Huang, Elly Lestari Jazdzyk, Meyner Nusalawo, Ibnu Maryanto, Maharadatunkamsi, Sigit Wiantoro, and Tigga Kingston Source: Acta Chiropterologica, 16(2):413-449. Published By: Museum and Institute of Zoology, Polish Academy of Sciences DOI: http://dx.doi.org/10.3161/150811014X687369 URL: http://www.bioone.org/doi/full/10.3161/150811014X687369 BioOne (www.bioone.org) is a nonprofit, online aggregation of core research in the biological, ecological, and environmental sciences. BioOne provides a sustainable online platform for over 170 journals and books published by nonprofit societies, associations, museums, institutions, and presses. Your use of this PDF, the BioOne Web site, and all posted and associated content indicates your acceptance of BioOne’s Terms of Use, available at www.bioone.org/page/terms_of_use. Usage of BioOne content is strictly limited to personal, educational, and non-commercial use. Commercial inquiries or rights and permissions requests should be directed to the individual publisher as copyright holder. BioOne sees sustainable scholarly publishing as an inherently collaborative enterprise connecting authors, nonprofit publishers, academic institutions, research libraries, and research funders in the common goal of maximizing access to critical research. Acta Chiropterologica, 16(2): 413–449, 2014 PL ISSN 1508-1109 © Museum and Institute of Zoology PAS doi: 10.3161/150811014X687369 A recent -

Chiroptera: Pteropodidae)
Chapter 6 Phylogenetic Relationships of Harpyionycterine Megabats (Chiroptera: Pteropodidae) NORBERTO P. GIANNINI1,2, FRANCISCA CUNHA ALMEIDA1,3, AND NANCY B. SIMMONS1 ABSTRACT After almost 70 years of stability following publication of Andersen’s (1912) monograph on the group, the systematics of megachiropteran bats (Chiroptera: Pteropodidae) was thrown into flux with the advent of molecular phylogenetics in the 1980s—a state where it has remained ever since. One particularly problematic group has been the Austromalayan Harpyionycterinae, currently thought to include Dobsonia and Harpyionycteris, and probably also Aproteles.Inthis contribution we revisit the systematics of harpyionycterines. We examine historical hypotheses of relationships including the suggestion by O. Thomas (1896) that the rousettine Boneia bidens may be related to Harpyionycteris, and report the results of a series of phylogenetic analyses based on new as well as previously published sequence data from the genes RAG1, RAG2, vWF, c-mos, cytb, 12S, tVal, 16S,andND2. Despite a striking lack of morphological synapomorphies, results of our combined analyses indicate that Boneia groups with Aproteles, Dobsonia, and Harpyionycteris in a well-supported, expanded Harpyionycterinae. While monophyly of this group is well supported, topological changes within this clade across analyses of different data partitions indicate conflicting phylogenetic signals in the mitochondrial partition. The position of the harpyionycterine clade within the megachiropteran tree remains somewhat uncertain. Nevertheless, biogeographic patterns (vicariance-dispersal events) within Harpyionycterinae appear clear and can be directly linked to major biogeographic boundaries of the Austromalayan region. The new phylogeny of Harpionycterinae also provides a new framework for interpreting aspects of dental evolution in pteropodids (e.g., reduction in the incisor dentition) and allows prediction of roosting habits for Harpyionycteris, whose habits are unknown. -
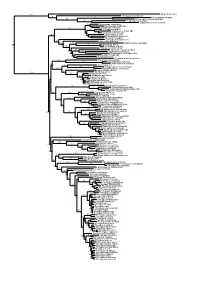
Figs1 ML Tree.Pdf
100 Megaderma lyra Rhinopoma hardwickei 71 100 Rhinolophus creaghi 100 Rhinolophus ferrumequinum 100 Hipposideros armiger Hipposideros commersoni 99 Megaerops ecaudatus 85 Megaerops niphanae 100 Megaerops kusnotoi 100 Cynopterus sphinx 98 Cynopterus horsfieldii 69 Cynopterus brachyotis 94 50 Ptenochirus minor 86 Ptenochirus wetmorei Ptenochirus jagori Dyacopterus spadiceus 99 Sphaerias blanfordi 99 97 Balionycteris maculata 100 Aethalops alecto 99 Aethalops aequalis Thoopterus nigrescens 97 Alionycteris paucidentata 33 99 Haplonycteris fischeri 29 Otopteropus cartilagonodus Latidens salimalii 43 88 Penthetor lucasi Chironax melanocephalus 90 Syconycteris australis 100 Macroglossus minimus 34 Macroglossus sobrinus 92 Boneia bidens 100 Harpyionycteris whiteheadi 69 Harpyionycteris celebensis Aproteles bulmerae 51 Dobsonia minor 100 100 80 Dobsonia inermis Dobsonia praedatrix 99 96 14 Dobsonia viridis Dobsonia peronii 47 Dobsonia pannietensis 56 Dobsonia moluccensis 29 Dobsonia anderseni 100 Scotonycteris zenkeri 100 Casinycteris ophiodon 87 Casinycteris campomaanensis Casinycteris argynnis 99 100 Eonycteris spelaea 100 Eonycteris major Eonycteris robusta 100 100 Rousettus amplexicaudatus 94 Rousettus spinalatus 99 Rousettus leschenaultii 100 Rousettus aegyptiacus 77 Rousettus madagascariensis 87 Rousettus obliviosus Stenonycteris lanosus 100 Megaloglossus woermanni 100 91 Megaloglossus azagnyi 22 Myonycteris angolensis 100 87 Myonycteris torquata 61 Myonycteris brachycephala 33 41 Myonycteris leptodon Myonycteris relicta 68 Plerotes anchietae -

Ecological Assessments in the B+WISER Sites
Ecological Assessments in the B+WISER Sites (Northern Sierra Madre Natural Park, Upper Marikina-Kaliwa Forest Reserve, Bago River Watershed and Forest Reserve, Naujan Lake National Park and Subwatersheds, Mt. Kitanglad Range Natural Park and Mt. Apo Natural Park) Philippines Biodiversity & Watersheds Improved for Stronger Economy & Ecosystem Resilience (B+WISER) 23 March 2015 This publication was produced for review by the United States Agency for International Development. It was prepared by Chemonics International Inc. The Biodiversity and Watersheds Improved for Stronger Economy and Ecosystem Resilience Program is funded by the USAID, Contract No. AID-492-C-13-00002 and implemented by Chemonics International in association with: Fauna and Flora International (FFI) Haribon Foundation World Agroforestry Center (ICRAF) The author’s views expressed in this publication do not necessarily reflect the views of the United States Agency for International Development or the United States Government. Ecological Assessments in the B+WISER Sites Philippines Biodiversity and Watersheds Improved for Stronger Economy and Ecosystem Resilience (B+WISER) Program Implemented with: Department of Environment and Natural Resources Other National Government Agencies Local Government Units and Agencies Supported by: United States Agency for International Development Contract No.: AID-492-C-13-00002 Managed by: Chemonics International Inc. in partnership with Fauna and Flora International (FFI) Haribon Foundation World Agroforestry Center (ICRAF) 23 March -

Priority-Setting for Philippine Bats Using Practical Approach to Guide Effective Species Conservation and Policy-Making in the Anthropocene
Published by Associazione Teriologica Italiana Volume 30 (1): 74–83, 2019 Hystrix, the Italian Journal of Mammalogy Available online at: http://www.italian-journal-of-mammalogy.it doi:10.4404/hystrix–00172-2019 Research Article Priority-setting for Philippine bats using practical approach to guide effective species conservation and policy-making in the Anthropocene Krizler Cejuela Tanalgo1,2,3,4,∗, Alice Catherine Hughes1,3 1Landscape Ecology Group, Centre for Integrative Conservation, Xishuangbanna Tropical Botanical Garden, Chinese Academy of Sciences, Menglun, Mengla, Yunnan Province 666303, People’s Republic of China 2International College, University of Chinese Academy of Sciences, Beijing 100049, People’s Republic of China 3Southeast Asia Biodiversity Research Institute, Chinese Academy of Sciences, Menglun, Mengla, Yunnan Province 666303, People’s Republic of China 4Department of Biological Sciences, College of Arts and Sciences, University of Southern Mindanao, Kabacan 9407, North Cotabato, the Republic of the Philippines Keywords: Abstract asian tropics endemism National level approaches to the development and implementation of effective conservation policy forest loss and practice are often challenged by limited capacity and resources. Developing relevant and hunting achievable priorities at the national level is a crucial step for effective conservation. The Philip- islands pine archipelago includes over 7000 islands and is one of only two countries considered both a oil palm global biodiversity hotspot and a megadiversity country. Yet, few studies have conducted over- arching synthesis for threats and conservation priorities of any species group. As bats make up a Article history: significant proportion of mammalian diversity in the Philippines and fulfil vital roles to maintain Received: 01/19/2019 ecosystem health and services we focus on assessing the threats and priorities to their conserva- Accepted: 24/06/2019 tion across the Philippines. -

Bat Count 2003
BAT COUNT 2003 Working to promote the long term, sustainable conservation of globally threatened flying foxes in the Philippines, by developing baseline population information, increasing public awareness, and training students and protected area managers in field monitoring techniques. 1 A Terminal Report Submitted by Tammy Mildenstein1, Apolinario B. Cariño2, and Samuel Stier1 1Fish and Wildlife Biology, University of Montana, USA 2Silliman University and Mt. Talinis – Twin Lakes Federation of People’s Organizations, Diputado Extension, Sibulan, Negros Oriental, Philippines Photo by: Juan Pablo Moreiras 2 EXECUTIVE SUMMARY Large flying foxes in insular Southeast Asia are the most threatened of the Old World fruit bats due to deforestation, unregulated hunting, and little conservation commitment from local governments. Despite the fact they are globally endangered and play essential ecological roles in forest regeneration as seed dispersers and pollinators, there have been only a few studies on these bats that provide information useful to their conservation management. Our project aims to promote the conservation of large flying foxes in the Philippines by providing protected area managers with the training and the baseline information necessary to design and implement a long-term management plan for flying foxes. We focused our efforts on the globally endangered Philippine endemics, Acerodon jubatus and Acerodon leucotis, and the bats that commonly roost with them, Pteropus hypomelanus, P. vampyrus lanensis, and P. pumilus which are thought to be declining in the Philippines. Local participation is an integral part of our project. We conducted the first national training workshop on flying fox population counts and conservation at the Subic Bay area. -

Index of Handbook of the Mammals of the World. Vol. 9. Bats
Index of Handbook of the Mammals of the World. Vol. 9. Bats A agnella, Kerivoula 901 Anchieta’s Bat 814 aquilus, Glischropus 763 Aba Leaf-nosed Bat 247 aladdin, Pipistrellus pipistrellus 771 Anchieta’s Broad-faced Fruit Bat 94 aquilus, Platyrrhinus 567 Aba Roundleaf Bat 247 alascensis, Myotis lucifugus 927 Anchieta’s Pipistrelle 814 Arabian Barbastelle 861 abae, Hipposideros 247 alaschanicus, Hypsugo 810 anchietae, Plerotes 94 Arabian Horseshoe Bat 296 abae, Rhinolophus fumigatus 290 Alashanian Pipistrelle 810 ancricola, Myotis 957 Arabian Mouse-tailed Bat 164, 170, 176 abbotti, Myotis hasseltii 970 alba, Ectophylla 466, 480, 569 Andaman Horseshoe Bat 314 Arabian Pipistrelle 810 abditum, Megaderma spasma 191 albatus, Myopterus daubentonii 663 Andaman Intermediate Horseshoe Arabian Trident Bat 229 Abo Bat 725, 832 Alberico’s Broad-nosed Bat 565 Bat 321 Arabian Trident Leaf-nosed Bat 229 Abo Butterfly Bat 725, 832 albericoi, Platyrrhinus 565 andamanensis, Rhinolophus 321 arabica, Asellia 229 abramus, Pipistrellus 777 albescens, Myotis 940 Andean Fruit Bat 547 arabicus, Hypsugo 810 abrasus, Cynomops 604, 640 albicollis, Megaerops 64 Andersen’s Bare-backed Fruit Bat 109 arabicus, Rousettus aegyptiacus 87 Abruzzi’s Wrinkle-lipped Bat 645 albipinnis, Taphozous longimanus 353 Andersen’s Flying Fox 158 arabium, Rhinopoma cystops 176 Abyssinian Horseshoe Bat 290 albiventer, Nyctimene 36, 118 Andersen’s Fruit-eating Bat 578 Arafura Large-footed Bat 969 Acerodon albiventris, Noctilio 405, 411 Andersen’s Leaf-nosed Bat 254 Arata Yellow-shouldered Bat 543 Sulawesi 134 albofuscus, Scotoecus 762 Andersen’s Little Fruit-eating Bat 578 Arata-Thomas Yellow-shouldered Talaud 134 alboguttata, Glauconycteris 833 Andersen’s Naked-backed Fruit Bat 109 Bat 543 Acerodon 134 albus, Diclidurus 339, 367 Andersen’s Roundleaf Bat 254 aratathomasi, Sturnira 543 Acerodon mackloti (see A. -

A Checklist of the Mammals of South-East Asia
A Checklist of the Mammals of South-east Asia A Checklist of the Mammals of South-east Asia PHOLIDOTA Pangolin (Manidae) 1 Sunda Pangolin (Manis javanica) 2 Chinese Pangolin (Manis pentadactyla) INSECTIVORA Gymnures (Erinaceidae) 3 Moonrat (Echinosorex gymnurus) 4 Short-tailed Gymnure (Hylomys suillus) 5 Chinese Gymnure (Hylomys sinensis) 6 Large-eared Gymnure (Hylomys megalotis) Moles (Talpidae) 7 Slender Shrew-mole (Uropsilus gracilis) 8 Kloss's Mole (Euroscaptor klossi) 9 Large Chinese Mole (Euroscaptor grandis) 10 Long-nosed Chinese Mole (Euroscaptor longirostris) 11 Small-toothed Mole (Euroscaptor parvidens) 12 Blyth's Mole (Parascaptor leucura) 13 Long-tailed Mole (Scaptonyx fuscicauda) Shrews (Soricidae) 14 Lesser Stripe-backed Shrew (Sorex bedfordiae) 15 Myanmar Short-tailed Shrew (Blarinella wardi) 16 Indochinese Short-tailed Shrew (Blarinella griselda) 17 Hodgson's Brown-toothed Shrew (Episoriculus caudatus) 18 Bailey's Brown-toothed Shrew (Episoriculus baileyi) 19 Long-taied Brown-toothed Shrew (Episoriculus macrurus) 20 Lowe's Brown-toothed Shrew (Chodsigoa parca) 21 Van Sung's Shrew (Chodsigoa caovansunga) 22 Mole Shrew (Anourosorex squamipes) 23 Himalayan Water Shrew (Chimarrogale himalayica) 24 Styan's Water Shrew (Chimarrogale styani) Page 1 of 17 Database: Gehan de Silva Wijeyeratne, www.jetwingeco.com A Checklist of the Mammals of South-east Asia 25 Malayan Water Shrew (Chimarrogale hantu) 26 Web-footed Water Shrew (Nectogale elegans) 27 House Shrew (Suncus murinus) 28 Pygmy White-toothed Shrew (Suncus etruscus) 29 South-east -

Investigating the Role of Bats in Emerging Zoonoses
12 ISSN 1810-1119 FAO ANIMAL PRODUCTION AND HEALTH manual INVESTIGATING THE ROLE OF BATS IN EMERGING ZOONOSES Balancing ecology, conservation and public health interest Cover photographs: Left: © Jon Epstein. EcoHealth Alliance Center: © Jon Epstein. EcoHealth Alliance Right: © Samuel Castro. Bureau of Animal Industry Philippines 12 FAO ANIMAL PRODUCTION AND HEALTH manual INVESTIGATING THE ROLE OF BATS IN EMERGING ZOONOSES Balancing ecology, conservation and public health interest Edited by Scott H. Newman, Hume Field, Jon Epstein and Carol de Jong FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 2011 Recommended Citation Food and Agriculture Organisation of the United Nations. 2011. Investigating the role of bats in emerging zoonoses: Balancing ecology, conservation and public health interests. Edited by S.H. Newman, H.E. Field, C.E. de Jong and J.H. Epstein. FAO Animal Production and Health Manual No. 12. Rome. The designations employed and the presentation of material in this information product do not imply the expression of any opinion whatsoever on the part of the Food and Agriculture Organization of the United Nations (FAO) concerning the legal or development status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. The mention of specific companies or products of manufacturers, whether or not these have been patented, does not imply that these have been endorsed or recommended by FAO in preference to others of a similar nature that are not mentioned. The views expressed in this information product are those of the author(s) and do not necessarily reflect the views of FAO. -

Taxonomy and Natural History of the Southeast Asian Fruit-Bat Genus Dyacopterus
Journal of Mammalogy, 88(2):302–318, 2007 TAXONOMY AND NATURAL HISTORY OF THE SOUTHEAST ASIAN FRUIT-BAT GENUS DYACOPTERUS KRISTOFER M. HELGEN,* DIETER KOCK,RAI KRISTIE SALVE C. GOMEZ,NINA R. INGLE, AND MARTUA H. SINAGA Division of Mammals, National Museum of Natural History (NHB 390, MRC 108), Smithsonian Institution, P.O. Box 37012, Washington, D.C. 20013-7012, USA (KMH) Forschungsinstitut Senckenberg, Senckenberganlage 25, Frankfurt, D-60325, Germany (DK) Philippine Eagle Foundation, VAL Learning Village, Ruby Street, Marfori Heights, Davao City, 8000, Philippines (RKSCG) Department of Natural Resources, Cornell University, Ithaca, NY 14853, USA (NRI) Division of Mammals, Field Museum of Natural History, Chicago, IL 60605, USA (NRI) Museum Zoologicum Bogoriense, Jl. Raya Cibinong Km 46, Cibinong 16911, Indonesia (MHS) The pteropodid genus Dyacopterus Andersen, 1912, comprises several medium-sized fruit-bat species endemic to forested areas of Sundaland and the Philippines. Specimens of Dyacopterus are sparsely represented in collections of world museums, which has hindered resolution of species limits within the genus. Based on our studies of most available museum material, we review the infrageneric taxonomy of Dyacopterus using craniometric and other comparisons. In the past, 2 species have been described—D. spadiceus (Thomas, 1890), described from Borneo and later recorded from the Malay Peninsula, and D. brooksi Thomas, 1920, described from Sumatra. These 2 nominal taxa are often recognized as species or conspecific subspecies representing these respective populations. Our examinations instead suggest that both previously described species of Dyacopterus co-occur on the Sunda Shelf—the smaller-skulled D. spadiceus in peninsular Malaysia, Sumatra, and Borneo, and the larger-skulled D. -

Habitat Selection of Endangered and Endemic Large Flying-Foxes in Subic
Biological Conservation 126 (2005) 93–102 www.elsevier.com/locate/biocon Habitat selection of endangered and endemic large Xying-foxes in Subic Bay, Philippines Tammy L. Mildenstein a,¤, Sam C. Stier a, C.E. Nuevo-Diego b, L. Scott Mills c a c/o US Peace Corps, Patio Madrigal Compound, 2775 Roxas Blvd., Pasay City, Metro Manila, Philippines b 9872 Isarog St. Umali Subdivision, Los Baños, Laguna 4031, Philippines c Wildlife Biology Program, School of Forestry, University of Montana, Missoula, MT 59812-0596, USA Received 6 June 2004 Available online 5 July 2005 Abstract Large Xying-foxes in insular Southeast Asia are the most threatened of the Old World fruit bats due to high levels of deforestation and hunting and eVectively little local conservation commitment. The forest at Subic Bay, Philippines, supports a rare, large colony of vulnerable Philippine giant fruit bats (Pteropus vampyrus lanensis) and endangered and endemic golden-crowned Xying-foxes (Acerodon jubatus). These large Xying-foxes are optimal for conservation focus, because in addition to being keystone, Xagship, and umbrella species, the bats are important to Subic Bay’s economy and its indigenous cultures. Habitat selection information stream- lines management’s eVorts to protect and conserve these popular but threatened animals. We used radio telemetry to describe the bats’ nighttime use of habitat on two ecological scales: vegetation and microhabitat. The fruit bats used the entire 14,000 ha study area, including all of Subic Bay Watershed Reserve, as well as neighboring forests just outside the protected area boundaries. Their recorded foraging locations ranged between 0.4 and 12 km from the roost. -

PARASITIC on MEGACHIROPTERAN BATS X
Pacific Insects Monograph 28: 213-243 20 June 1971 REVIEW OF THE STREBLIDAE (Diptera) PARASITIC ON MEGACHIROPTERAN BATS x By T. C. Maa2 Abstract. Of the 16 streblid species previously recorded as parasites of the Megachiroptera, only 6 are here considered to be correctly so associated. Five of these 6 species are re-assigned to a new genus and only 1 is retained in the genus Brachytarsina (^Nycteribosca). These 2 genera are each divided into 2 subgenera and their host relationships, distributional patterns and evolutionary trends are discussed. Earlier records of the species are critically reviewed and are incorporated with new data which are based on some 650 specimens. The new taxa described are Megastrebla, n. gen. (type N. gigantea Speiser); Aoroura, n. subgen, (type N. nigriceps Jobling); Psilacris, n. subgen, (type N. longiarista Jobling); M. (A) limbooliati, n. sp. (Malaya, Borneo); M. (M.) gigantea kaluzvawae, n. ssp. (Fergusson I.); M. (M) gigantea salomonis, n. ssp. (Solomon Is.); M. (M) parvior papuae, n. ssp. (New Guinea). Streblid batflies are rarely found on the suborder Megachiroptera, composed of the single family Pteropodidae, whose members are generally referred to as fruit bats. Only 16 species have been recorded on these bats. A closer examination of the pub lished records clearly indicates that 10 of these 16 species (see Appendix II) should not be considered true parasites of the Megachiroptera; available data support the con cept that no streblids normally breed simultaneously on both the Megachiroptera and Microchiroptera, and among the 39 genera of the former suborder, only those which usually roost in partially illuminated caves and rock-crevices serve as normal breeding hosts of Streblidae.