Genetic Defect in Phospholipase Cd1 Protects Mice from Obesity By
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supplemental Figure 1. Vimentin
Double mutant specific genes Transcript gene_assignment Gene Symbol RefSeq FDR Fold- FDR Fold- FDR Fold- ID (single vs. Change (double Change (double Change wt) (single vs. wt) (double vs. single) (double vs. wt) vs. wt) vs. single) 10485013 BC085239 // 1110051M20Rik // RIKEN cDNA 1110051M20 gene // 2 E1 // 228356 /// NM 1110051M20Ri BC085239 0.164013 -1.38517 0.0345128 -2.24228 0.154535 -1.61877 k 10358717 NM_197990 // 1700025G04Rik // RIKEN cDNA 1700025G04 gene // 1 G2 // 69399 /// BC 1700025G04Rik NM_197990 0.142593 -1.37878 0.0212926 -3.13385 0.093068 -2.27291 10358713 NM_197990 // 1700025G04Rik // RIKEN cDNA 1700025G04 gene // 1 G2 // 69399 1700025G04Rik NM_197990 0.0655213 -1.71563 0.0222468 -2.32498 0.166843 -1.35517 10481312 NM_027283 // 1700026L06Rik // RIKEN cDNA 1700026L06 gene // 2 A3 // 69987 /// EN 1700026L06Rik NM_027283 0.0503754 -1.46385 0.0140999 -2.19537 0.0825609 -1.49972 10351465 BC150846 // 1700084C01Rik // RIKEN cDNA 1700084C01 gene // 1 H3 // 78465 /// NM_ 1700084C01Rik BC150846 0.107391 -1.5916 0.0385418 -2.05801 0.295457 -1.29305 10569654 AK007416 // 1810010D01Rik // RIKEN cDNA 1810010D01 gene // 7 F5 // 381935 /// XR 1810010D01Rik AK007416 0.145576 1.69432 0.0476957 2.51662 0.288571 1.48533 10508883 NM_001083916 // 1810019J16Rik // RIKEN cDNA 1810019J16 gene // 4 D2.3 // 69073 / 1810019J16Rik NM_001083916 0.0533206 1.57139 0.0145433 2.56417 0.0836674 1.63179 10585282 ENSMUST00000050829 // 2010007H06Rik // RIKEN cDNA 2010007H06 gene // --- // 6984 2010007H06Rik ENSMUST00000050829 0.129914 -1.71998 0.0434862 -2.51672 -

(12) Patent Application Publication (10) Pub. No.: US 2003/0082511 A1 Brown Et Al
US 20030082511A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2003/0082511 A1 Brown et al. (43) Pub. Date: May 1, 2003 (54) IDENTIFICATION OF MODULATORY Publication Classification MOLECULES USING INDUCIBLE PROMOTERS (51) Int. Cl." ............................... C12O 1/00; C12O 1/68 (52) U.S. Cl. ..................................................... 435/4; 435/6 (76) Inventors: Steven J. Brown, San Diego, CA (US); Damien J. Dunnington, San Diego, CA (US); Imran Clark, San Diego, CA (57) ABSTRACT (US) Correspondence Address: Methods for identifying an ion channel modulator, a target David B. Waller & Associates membrane receptor modulator molecule, and other modula 5677 Oberlin Drive tory molecules are disclosed, as well as cells and vectors for Suit 214 use in those methods. A polynucleotide encoding target is San Diego, CA 92121 (US) provided in a cell under control of an inducible promoter, and candidate modulatory molecules are contacted with the (21) Appl. No.: 09/965,201 cell after induction of the promoter to ascertain whether a change in a measurable physiological parameter occurs as a (22) Filed: Sep. 25, 2001 result of the candidate modulatory molecule. Patent Application Publication May 1, 2003 Sheet 1 of 8 US 2003/0082511 A1 KCNC1 cDNA F.G. 1 Patent Application Publication May 1, 2003 Sheet 2 of 8 US 2003/0082511 A1 49 - -9 G C EH H EH N t R M h so as se W M M MP N FIG.2 Patent Application Publication May 1, 2003 Sheet 3 of 8 US 2003/0082511 A1 FG. 3 Patent Application Publication May 1, 2003 Sheet 4 of 8 US 2003/0082511 A1 KCNC1 ITREXCHO KC 150 mM KC 2000000 so 100 mM induced Uninduced Steady state O 100 200 300 400 500 600 700 Time (seconds) FIG. -

Supplementary Table 2
Supplementary Table 2. Differentially Expressed Genes following Sham treatment relative to Untreated Controls Fold Change Accession Name Symbol 3 h 12 h NM_013121 CD28 antigen Cd28 12.82 BG665360 FMS-like tyrosine kinase 1 Flt1 9.63 NM_012701 Adrenergic receptor, beta 1 Adrb1 8.24 0.46 U20796 Nuclear receptor subfamily 1, group D, member 2 Nr1d2 7.22 NM_017116 Calpain 2 Capn2 6.41 BE097282 Guanine nucleotide binding protein, alpha 12 Gna12 6.21 NM_053328 Basic helix-loop-helix domain containing, class B2 Bhlhb2 5.79 NM_053831 Guanylate cyclase 2f Gucy2f 5.71 AW251703 Tumor necrosis factor receptor superfamily, member 12a Tnfrsf12a 5.57 NM_021691 Twist homolog 2 (Drosophila) Twist2 5.42 NM_133550 Fc receptor, IgE, low affinity II, alpha polypeptide Fcer2a 4.93 NM_031120 Signal sequence receptor, gamma Ssr3 4.84 NM_053544 Secreted frizzled-related protein 4 Sfrp4 4.73 NM_053910 Pleckstrin homology, Sec7 and coiled/coil domains 1 Pscd1 4.69 BE113233 Suppressor of cytokine signaling 2 Socs2 4.68 NM_053949 Potassium voltage-gated channel, subfamily H (eag- Kcnh2 4.60 related), member 2 NM_017305 Glutamate cysteine ligase, modifier subunit Gclm 4.59 NM_017309 Protein phospatase 3, regulatory subunit B, alpha Ppp3r1 4.54 isoform,type 1 NM_012765 5-hydroxytryptamine (serotonin) receptor 2C Htr2c 4.46 NM_017218 V-erb-b2 erythroblastic leukemia viral oncogene homolog Erbb3 4.42 3 (avian) AW918369 Zinc finger protein 191 Zfp191 4.38 NM_031034 Guanine nucleotide binding protein, alpha 12 Gna12 4.38 NM_017020 Interleukin 6 receptor Il6r 4.37 AJ002942 -

Supplementary Material Contents
Supplementary Material Contents Immune modulating proteins identified from exosomal samples.....................................................................2 Figure S1: Overlap between exosomal and soluble proteomes.................................................................................... 4 Bacterial strains:..............................................................................................................................................4 Figure S2: Variability between subjects of effects of exosomes on BL21-lux growth.................................................... 5 Figure S3: Early effects of exosomes on growth of BL21 E. coli .................................................................................... 5 Figure S4: Exosomal Lysis............................................................................................................................................ 6 Figure S5: Effect of pH on exosomal action.................................................................................................................. 7 Figure S6: Effect of exosomes on growth of UPEC (pH = 6.5) suspended in exosome-depleted urine supernatant ....... 8 Effective exosomal concentration....................................................................................................................8 Figure S7: Sample constitution for luminometry experiments..................................................................................... 8 Figure S8: Determining effective concentration ......................................................................................................... -

Human Urinary Exosomes As Innate Immune Effectors
BASIC RESEARCH www.jasn.org Human Urinary Exosomes as Innate Immune Effectors † † ‡ Thomas F. Hiemstra,* Philip D. Charles, Tannia Gracia, Svenja S. Hester,§ † ‡ | Laurent Gatto, Rafia Al-Lamki,* R. Andres Floto,* Ya Su, Jeremy N. Skepper, † ‡ Kathryn S. Lilley, and Fiona E. Karet Frankl *Department of Medicine, †Cambridge Centre for Proteome Research and Cambridge Systems Biology Centre, Department of Biochemistry, ‡Department of Medical Genetics, and |Multi-Imaging Centre, Department of Anatomy, University of Cambridge, Cambridge, United Kingdom; and §Sir William Dunn School of Pathology, University of Oxford, Oxford, United Kingdom ABSTRACT Exosomes are small extracellular vesicles, approximately 50 nm in diameter, derived from the endocytic pathway and released by a variety of cell types. Recent data indicate a spectrum of exosomal functions, including RNA transfer, antigen presentation, modulation of apoptosis, and shedding of obsolete protein. Exosomes derived from all nephron segments are also present in human urine, where their function is unknown. Although one report suggested in vitro uptake of exosomes by renal cortical collecting duct cells, most studies of human urinary exosomes have focused on biomarker discovery rather than exosome function. Here, we report results from in-depth proteomic analyses and EM showing that normal human urinary exosomes are significantly enriched for innate immune proteins that include antimicrobial proteins and peptides and bacterial and viral receptors. Urinary exosomes, but not the prevalent soluble urinary protein uromodulin (Tamm–Horsfall protein), potently inhibited growth of pathogenic and commensal Escherichia coli and induced bacterial lysis. Bacterial killing depended on exosome structural integrity and occurred optimally at the acidic pH typical of urine from omnivorous humans. -

Potent Lipolytic Activity of Lactoferrin in Mature Adipocytes
Biosci. Biotechnol. Biochem., 77 (3), 566–571, 2013 Potent Lipolytic Activity of Lactoferrin in Mature Adipocytes y Tomoji ONO,1;2; Chikako FUJISAKI,1 Yasuharu ISHIHARA,1 Keiko IKOMA,1;2 Satoru MORISHITA,1;3 Michiaki MURAKOSHI,1;4 Keikichi SUGIYAMA,1;5 Hisanori KATO,3 Kazuo MIYASHITA,6 Toshihide YOSHIDA,4;7 and Hoyoku NISHINO4;5 1Research and Development Headquarters, Lion Corporation, 100 Tajima, Odawara, Kanagawa 256-0811, Japan 2Department of Supramolecular Biology, Graduate School of Nanobioscience, Yokohama City University, 3-9 Fukuura, Kanazawa-ku, Yokohama, Kanagawa 236-0004, Japan 3Food for Life, Organization for Interdisciplinary Research Projects, The University of Tokyo, 1-1-1 Yayoi, Bunkyo-ku, Tokyo 113-8657, Japan 4Kyoto Prefectural University of Medicine, Kawaramachi-Hirokoji, Kamigyou-ku, Kyoto 602-8566, Japan 5Research Organization of Science and Engineering, Ritsumeikan University, 1-1-1 Nojihigashi, Kusatsu, Shiga 525-8577, Japan 6Department of Marine Bioresources Chemistry, Faculty of Fisheries Sciences, Hokkaido University, 3-1-1 Minatocho, Hakodate, Hokkaido 041-8611, Japan 7Kyoto City Hospital, 1-2 Higashi-takada-cho, Mibu, Nakagyou-ku, Kyoto 604-8845, Japan Received October 22, 2012; Accepted November 26, 2012; Online Publication, March 7, 2013 [doi:10.1271/bbb.120817] Lactoferrin (LF) is a multifunctional glycoprotein resistance, high blood pressure, and dyslipidemia. To found in mammalian milk. We have shown in a previous prevent progression of metabolic syndrome, lifestyle clinical study that enteric-coated bovine LF tablets habits must be improved to achieve a balance between decreased visceral fat accumulation. To address the energy intake and consumption. In addition, the use of underlying mechanism, we conducted in vitro studies specific food factors as helpful supplements is attracting and revealed the anti-adipogenic action of LF in pre- increasing attention. -

Induction of Therapeutic Tissue Tolerance Foxp3 Expression Is
Downloaded from http://www.jimmunol.org/ by guest on October 2, 2021 is online at: average * The Journal of Immunology , 13 of which you can access for free at: 2012; 189:3947-3956; Prepublished online 17 from submission to initial decision 4 weeks from acceptance to publication September 2012; doi: 10.4049/jimmunol.1200449 http://www.jimmunol.org/content/189/8/3947 Foxp3 Expression Is Required for the Induction of Therapeutic Tissue Tolerance Frederico S. Regateiro, Ye Chen, Adrian R. Kendal, Robert Hilbrands, Elizabeth Adams, Stephen P. Cobbold, Jianbo Ma, Kristian G. Andersen, Alexander G. Betz, Mindy Zhang, Shruti Madhiwalla, Bruce Roberts, Herman Waldmann, Kathleen F. Nolan and Duncan Howie J Immunol cites 35 articles Submit online. Every submission reviewed by practicing scientists ? is published twice each month by Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts http://jimmunol.org/subscription http://www.jimmunol.org/content/suppl/2012/09/17/jimmunol.120044 9.DC1 This article http://www.jimmunol.org/content/189/8/3947.full#ref-list-1 Information about subscribing to The JI No Triage! Fast Publication! Rapid Reviews! 30 days* Why • • • Material References Permissions Email Alerts Subscription Supplementary The Journal of Immunology The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2012 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. This information is current as of October 2, 2021. -

Acetylcholine Induces Yeast to Hyphal Form Transition in Candida Albicans
mic eno s & G B l i a o g l o n g u y F Ali and Karuppayil, Fungal Genom Biol 2018, 8:1 Fungal Genomics & Biology DOI: 10.4172/2165-8056.1000154 ISSN: 2165-8056 Research Open Access Acetylcholine Induces Yeast to Hyphal Form Transition in Candida Albicans Ali A and Karuppayil SM* Former Director, School of Life Sciences, Swami Ramanand Teerth Marathwada University, India *Corresponding author:: Sankunny Mohan Karuppayil, Former Director, School of Life Sciences Swami Ramanand Teerth Marathwada University, India, Tel: +919764386253; E-mail:[email protected] Received date: March 12, 2018; Accepted date: March 28, 2018; Published date: April 04, 2018 Copyright: © 2018 Ali A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Abstract Background: In Candida Albicans, yeast to hyphal form transition can be induced by serum, proline, glucose, and N-acetyl glucosamine. Acetylcholine is a neuromodulator which can stimulate both muscarinic and nicotinic acetylcholine receptors in humans. In this study, we are reporting that acetylcholine can induce yeast to hyphal form transition in C. Albicans. The adenylyl cyclase inhibitor, MDL 12, 330A inhibited this transition indicating the role of cAMP. Muscarinic receptors in C. Albicans did not report yet. We have reported that C. Albicans Rrp9 exhibits identity and similarity with the human muscarinic receptor M1. In humans, activation of muscarinic M1 receptor can produce cAMP through inositol phosphate pathway. The inositol phosphate pathway in C. -

Mechanisms Underlying Memory Deficits Following West Nile Virus Neuroinvasive Disease Michael John Vasek Washington University in St
Washington University in St. Louis Washington University Open Scholarship Arts & Sciences Electronic Theses and Dissertations Arts & Sciences Summer 8-15-2015 Mechanisms Underlying Memory Deficits Following West Nile Virus Neuroinvasive Disease Michael John Vasek Washington University in St. Louis Follow this and additional works at: https://openscholarship.wustl.edu/art_sci_etds Recommended Citation Vasek, Michael John, "Mechanisms Underlying Memory Deficits Following West Nile Virus Neuroinvasive Disease" (2015). Arts & Sciences Electronic Theses and Dissertations. 688. https://openscholarship.wustl.edu/art_sci_etds/688 This Dissertation is brought to you for free and open access by the Arts & Sciences at Washington University Open Scholarship. It has been accepted for inclusion in Arts & Sciences Electronic Theses and Dissertations by an authorized administrator of Washington University Open Scholarship. For more information, please contact [email protected]. WASHINGTON UNIVERSITY IN ST. LOUIS Division of Biology and Biomedical Sciences Neurosciences Dissertation Examination Committee: Robyn Klein, Chair John Cirrito Michael Diamond David Gutmann Keiko Hirose Mechanisms Underlying Memory Deficits Following West Nile Virus Neuroinvasive Disease by Michael John Vasek A dissertation presented to the Graduate School of Arts & Sciences of Washington University in partial fulfillment of the requirements for the degree of Doctor of Philosophy August 2015 St. Louis, Missouri Table of Contents LIST OF FIGURES…………………………………………………....…….. iv LIST OF TABLES …………………………...………………………….……vi ACKNOWLEDGEMENTS …………….………………………………….…vii ABSTRACT………………………………………………………………...….x Chapter 1: Introduction The effects of neuroinflammation on memory and cognition…………….. 1 West Nile virus and the host immune response………..………………..... 1 The complement system of pathogen defense………………………..….. 3 Leukocytes and cytokines in anti-WNV defense…..…………………….... 5 Microglia as immune cells of the CNS…..………………………………..... 5 Neural development………..……………………………………..……….... -

Supplemental Figures 04 12 2017
Jung et al. 1 SUPPLEMENTAL FIGURES 2 3 Supplemental Figure 1. Clinical relevance of natural product methyltransferases (NPMTs) in brain disorders. (A) 4 Table summarizing characteristics of 11 NPMTs using data derived from the TCGA GBM and Rembrandt datasets for 5 relative expression levels and survival. In addition, published studies of the 11 NPMTs are summarized. (B) The 1 Jung et al. 6 expression levels of 10 NPMTs in glioblastoma versus non‐tumor brain are displayed in a heatmap, ranked by 7 significance and expression levels. *, p<0.05; **, p<0.01; ***, p<0.001. 8 2 Jung et al. 9 10 Supplemental Figure 2. Anatomical distribution of methyltransferase and metabolic signatures within 11 glioblastomas. The Ivy GAP dataset was downloaded and interrogated by histological structure for NNMT, NAMPT, 12 DNMT mRNA expression and selected gene expression signatures. The results are displayed on a heatmap. The 13 sample size of each histological region as indicated on the figure. 14 3 Jung et al. 15 16 Supplemental Figure 3. Altered expression of nicotinamide and nicotinate metabolism‐related enzymes in 17 glioblastoma. (A) Heatmap (fold change of expression) of whole 25 enzymes in the KEGG nicotinate and 18 nicotinamide metabolism gene set were analyzed in indicated glioblastoma expression datasets with Oncomine. 4 Jung et al. 19 Color bar intensity indicates percentile of fold change in glioblastoma relative to normal brain. (B) Nicotinamide and 20 nicotinate and methionine salvage pathways are displayed with the relative expression levels in glioblastoma 21 specimens in the TCGA GBM dataset indicated. 22 5 Jung et al. 23 24 Supplementary Figure 4. -

The Endosome Is a Master Regulator of Plasma Membrane Collagen Fibril Assembly
bioRxiv preprint doi: https://doi.org/10.1101/2021.03.25.436925; this version posted March 25, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. The endosome is a master regulator of plasma membrane collagen fibril assembly 1Joan Chang*, 1Adam Pickard, 1Richa Garva, 1Yinhui Lu, 2Donald Gullberg and 1Karl E. Kadler* 1Wellcome Centre for Cell-Matrix Research, Faculty of Biology, Medical and Health, University of Manchester, Michael Smith Building, Oxford Road, Manchester M13 9PT UK, 2Department of Biomedicine and Center for Cancer Biomarkers, Norwegian Center of Excellence, University of Bergen, Norway. * Co-corresponding authors: JC email: [email protected] (orcid.org/0000-0002-7283- 9759); KEK email: [email protected] (orcid.org/0000-0003-4977-4683) Keywords: collagen-I, endocytosis, extracellular matrix, fibril, fibrillogenesis, integrin-a11, trafficking, VPS33b, [abstract] [149 word max] Collagen fibrils are the principal supporting elements in vertebrate tissues. They account for 25% of total protein mass, exhibit a broad range of size and organisation depending on tissue and stage of development, and can be under circadian clock control. Here we show that the remarkable dynamic pleomorphism of collagen fibrils is underpinned by a mechanism that distinguishes between collagen secretion and initiation of fibril assembly, at the plasma membrane. Collagen fibrillogenesis occurring at the plasma membrane requires vacuolar protein sorting (VPS) 33b (which is under circadian clock control), collagen-binding integrin-a11 subunit, and is reduced when endocytosis is inhibited. Fibroblasts lacking VPS33b secrete soluble collagen without assembling fibrils, whereas constitutive over-expression of VPS33b increases fibril number with loss of fibril rhythmicity. -
Downloaded from the Mouse Lysosome Gene Database, Mlgdb
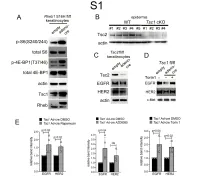
1 Supplemental Figure Legends 2 3 Supplemental Figure S1: Epidermal-specific mTORC1 gain-of-function models show 4 increased mTORC1 activation and down-regulate EGFR and HER2 protein expression in a 5 mTORC1-sensitive manner. (A) Immunoblotting of Rheb1 S16H flox/flox keratinocyte cultures 6 infected with empty or adenoviral cre recombinase for markers of mTORC1 (p-S6, p-4E-BP1) 7 activity. (B) Tsc1 cKO epidermal lysates also show decreased expression of TSC2 by 8 immunoblotting of the same experiment as in Figure 2A. (C) Immunoblotting of Tsc2 flox/flox 9 keratinocyte cultures infected with empty or adenoviral cre recombinase showing decreased EGFR 10 and HER2 protein expression. (D) Expression of EGFR and HER2 was decreased in Tsc1 cre 11 keratinocytes compared to empty controls, and up-regulated in response to Torin1 (1µM, 24 hrs), 12 by immunoblot analyses. Immunoblots are contemporaneous and parallel from the same biological 13 replicate and represent the same experiment as depicted in Figure 7B. (E) Densitometry 14 quantification of representative immunoblot experiments shown in Figures 2E and S1D (r≥3; error 15 bars represent STDEV; p-values by Student’s T-test). 16 17 18 19 20 21 22 23 Supplemental Figure S2: EGFR and HER2 transcription are unchanged with epidermal/ 24 keratinocyte Tsc1 or Rptor loss. Egfr and Her2 mRNA levels in (A) Tsc1 cKO epidermal lysates, 25 (B) Tsc1 cKO keratinocyte lysates and(C) Tsc1 cre keratinocyte lysates are minimally altered 26 compared to their respective controls. (r≥3; error bars represent STDEV; p-values by Student’s T- 27 test).