Cannabinoid Conditioned Reward and Aversion: Behavioral and Neural Processes Jennifer E
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Cannabinoids in the Pathophysiology of Skin Inflammation
molecules Review Cannabinoids in the Pathophysiology of Skin Inflammation Cristian Scheau 1 , Ioana Anca Badarau 1, Livia-Gratiela Mihai 1, Andreea-Elena Scheau 2, Daniel Octavian Costache 3, Carolina Constantin 4,5, Daniela Calina 6 , Constantin Caruntu 1,7,*, Raluca Simona Costache 8,* and Ana Caruntu 9,10 1 Department of Physiology, “Carol Davila” University of Medicine and Pharmacy, 050474 Bucharest, Romania; [email protected] (C.S.); [email protected] (I.A.B.); [email protected] (L.-G.M.) 2 Department of Radiology and Medical Imaging, Fundeni Clinical Institute, 022328 Bucharest, Romania; [email protected] 3 Department of Dermatology, “Carol Davila” Central Military Emergency Hospital, 010825 Bucharest, Romania; [email protected] 4 Immunology Department, ”Victor Babes” National Institute of Pathology, 050096 Bucharest, Romania; [email protected] 5 Department of Pathology, Colentina University Hospital, 020125 Bucharest, Romania 6 Department of Clinical Pharmacy, University of Medicine and Pharmacy of Craiova, 200349 Craiova, Romania; [email protected] 7 Department of Dermatology, “Prof. N. Paulescu” National Institute of Diabetes, Nutrition and Metabolic Diseases, 011233 Bucharest, Romania 8 Gastroenterology and Internal Medicine Clinic, Carol Davila University Central Emergency Military Hospital, Carol Davila University of Medicine and Pharmacy, 050474 Bucharest, Romania 9 Department of Oral and Maxillofacial Surgery, “Carol Davila” Central Military Emergency Hospital, 010825 Bucharest, Romania; [email protected] 10 Faculty of Medicine, “Titu Maiorescu” University, 031593 Bucharest, Romania * Correspondence: [email protected] (C.C.); [email protected] (R.S.C.); Tel.: +40-745-086-978 (C.C.) Academic Editor: Eric J. Downer Received: 30 December 2019; Accepted: 2 February 2020; Published: 4 February 2020 Abstract: Cannabinoids are increasingly-used substances in the treatment of chronic pain, some neuropsychiatric disorders and more recently, skin disorders with an inflammatory component. -

A Dissertation Entitled Uncovering Cannabinoid Signaling in C. Elegans
A Dissertation Entitled Uncovering Cannabinoid Signaling in C. elegans: A New Platform to Study the Effects of Medicinal Cannabis By Mitchell Duane Oakes Submitted to the Graduate Faculty as partial fulfillment of the requirements for the Doctor of Philosophy Degree in Biology ________________________________________ Dr. Richard Komuniecki, Committee Chair _______________________________________ Dr. Bruce Bamber, Committee Member ________________________________________ Dr. Patricia Komuniecki, Committee Member ________________________________________ Dr. Robert Steven, Committee Member ________________________________________ Dr. Ajith Karunarathne, Committee Member ________________________________________ Dr. Jianyang Du, Committee Member ________________________________________ Dr. Amanda Bryant-Friedrich, Dean College of Graduate Studies The University of Toledo August 2018 Copyright 2018, Mitchell Duane Oakes This document is copyrighted material. Under copyright law, no parts of this document may be reproduced without the expressed permission of the author. An Abstract of Uncovering Cannabinoid Signaling in C. elegans: A New Platform to Study the Effects of Medical Cannabis By Mitchell Duane Oakes Submitted to the Graduate Faculty as partial fulfillment of the requirements for the Doctor of Philosophy Degree in Biology The University of Toledo August 2018 Cannabis or marijuana, a popular recreational drug, alters sensory perception and exerts a range of medicinal benefits. The present study demonstrates that C. elegans exposed to -
Marinol Cannabidiol C21H30O2 Trade Name
Cannabinoids are a group of terpenophenolic compounds secreted by Cannabis flowers that provide relief from a wide array of symptoms including, pain, nausea, and inflammation. They operate by imitating the body’s natural endocannabinoids, which activate to maintain internal stability and overall health. When consumed, cannabinoids bind to receptor sites throughout the brain (CB1 receptors) and body (CB2 receptors). Different cannabinoids have different effects based on their binding affinity for each receptor. By targeting specific cannabinoids at these receptors, different types of relief can be achieved. Presently, there are at least 113 different cannabinoids isolated from Cannabis—each exhibiting varied effects. THC Tetrahydrocannabinol C21H30O2 Trade name: Marinol Legal Status: US – Schedule I, Schedule II (as Cesamet), Schedule III (as Marinol) OH CA – Schedule II UK – Class B AU – S8 (controlled) H Psychoactive Tetrahydrocannabinol (THC) is typically the most abundant cannabinoid present in cannabis products on the market today. THC has very high psychoactive characteristics and is associated with the ‘high’ and euphoria experienced when using cannabis products. When smoked or ingested, THC binds to cannabinoid receptors throughout the body and affects memory, O coordination, concentration, pleasure, and time perception. H Medicinal Benefits Analgesic • Anti-nauseant • Appetite Stimulant Reduces Glaucoma Symptoms Sleep Aid • Reduces Anxiety and PTSD Symptoms CBD Cannabidiol C21H30O2 Trade name: Epidiolex OH Legal Status: US – Schedule I CA – Schedule II UK – POM (Perscription only) H AU – S4 (Perscription only) non-psychoactive Cannabidiol (CBD) is a major phytocannabinoid and accounts for up to 40% of the plant’s extract. Due to its lack of psychoactivity and HO non-interference with motor and psychological functions, it is a leading candidate for a wide variety of medical applications. -

Recent Developments in Cannabis Chemistry
Recent Developments in Cannabis Chemistry BY ALEXANDER T. SHULGIN, Ph.D. The marijuana plant Cannabis sativa contains a bewildering Introduction array of organic chemicals. As is true with other botanic species, there are representatives of almost all chemical classes present, including mono- and sesquiterpenes, carbohy- drates, aromatics, and a variety of nitrogenous compounds. Interest in the study of this plant has centered primarily on the resinous fraction, as it is this material that is invested with the pharmacological activity that is peculiar to the plant. This resin is secreted by the female plant as a protective agent during seed ripening, although it can be found as a microscopic exudate through the aerial portions of plants of either sex. The pure resin, hashish or charas, is the most potent fraction of the plant, and has served as the source material for most of the chemical studies. The family of chemicals that has been isolated from this source has been referred to as the cannabinoid group. It is unique amongst psychotropic materials from plants in that there are no alkaloids present. The fraction is totally nitro- gen-free. Rather, the set of compounds can be considered as analogs of the parent compound cannabinol (I), a fusion product of terpene and a substituted resorcinol. Beyond the scope of this present review are such questions as the distribution of these compounds within the plant, the bo- tanic variability resulting from geographic distribution, the diversity of pharmacological action assignable to the several Reprinted from Journal of Psychedelic Drugs, vol. II, no. 1, 197 1. 397 398 Marijuana: Medical Papers distinct compounds present, and the various preparations and customs of administration. -

INTRODUCTION One of the Tasks of a Pharmacologist Is the Classification of Drugs
NEUROPSYCHOPHARMACOLOGIC STUDIES OF MARIJUANA: SOME SYNTHETIC AND NATURAL THC DERIVATIVES IN ANIMALS AND MAN* Edward F. Domino Department of Pharmacology, University of Michigan, Ann Arbor 48104 and Lafayette Clinic, Detroit, Mich. 48207 INTRODUCTION One of the tasks of a pharmacologist is the classification of drugs. Marijuana and its derivatives pose some problems. Legally marijuana has been classified as a narcotic, yet pharmacologically it is quite different. Mankind has known about the Cannabis plant from which marijuana and other products are obtained for more than 4,708 years. A considerable body of data is available to describe its gross effects. Over the centuries it has been called a depressant, euphoriant, ine- briant, intoxicant, hallucinogen, psychotomimetic, sedative-hypnotic-anesthetic, and stimulant. Perhaps more than any other drug it is a social irritant that elicits rather remarkable behavior reactions in both users and nonusers. Moreau’ in 1845, employed it to produce a model psychosis, thus initiating the fashion to study hallucinogens as a means of gaining insights into mental illness. Lewin2 classified Cannabis as one of the “phantastica: hallucinating substances.” One text on hallucinogens3 does not even refer to Cannabis, but another does? There are some aspects of the subjective effects of both LSD-25 and Cannabis that are similar, yet the latter produces sedative effects, no significant sympatho- mimetic actions, and no cross-tolerance to LSD-25. Hollister5 reviewed the re- cent findings on the effects in man of marijuana and its putative active ingredient Ag-THC, stressing that current investigators have, for the most part, confirmed the Cannabis-induced clinical syndromes long known to occur. -

The Use of Cannabinoids in Animals and Therapeutic Implications for Veterinary Medicine: a Review
Veterinarni Medicina, 61, 2016 (3): 111–122 Review Article doi: 10.17221/8762-VETMED The use of cannabinoids in animals and therapeutic implications for veterinary medicine: a review L. Landa1, A. Sulcova2, P. Gbelec3 1Faculty of Medicine, Masaryk University, Brno, Czech Republic 2Central European Institute of Technology, Masaryk University, Brno, Czech Republic 3Veterinary Hospital and Ambulance AA Vet, Prague, Czech Republic ABSTRACT: Cannabinoids/medical marijuana and their possible therapeutic use have received increased atten- tion in human medicine during the last years. This increased attention is also an issue for veterinarians because particularly companion animal owners now show an increased interest in the use of these compounds in veteri- nary medicine. This review sets out to comprehensively summarise well known facts concerning properties of cannabinoids, their mechanisms of action, role of cannabinoid receptors and their classification. It outlines the main pharmacological effects of cannabinoids in laboratory rodents and it also discusses examples of possible beneficial use in other animal species (ferrets, cats, dogs, monkeys) that have been reported in the scientific lit- erature. Finally, the article deals with the prospective use of cannabinoids in veterinary medicine. We have not intended to review the topic of cannabinoids in an exhaustive manner; rather, our aim was to provide both the scientific community and clinical veterinarians with a brief, concise and understandable overview of the use of cannabinoids in veterinary -

The Endocannabinoid System: Critical for the Neurotrophic Action of Psychotropic Drugs
Biomedical Reviews 2010; 21: 31-46. © Bul garian Society for Cell Biology ISSN 1314-1929 THE ENDOCANNABINOID SYSTEM: CRITICAL FOR THE NEUROTROPHIC ACTION OF PSYCHOTROPIC DRUGS Parichehr Hassanzadeh Research Center for Gastroenterology and Liver Diseases, Shahid Beheshti University of Medical Sciences, Tehran, Iran There is growing evidence that neurotrophins besides their well-established actions in regulating the survival, differentiation, and maintenance of the functions of specific populations of neurons, act as the potential mediators of antidepressant responses. Previous studies on the regulation of nerve growth factor (NGF) levels by psychotropic medications are limited in scope and the underlying mechanism(s) remain elusive. In this review, the latest findings on the effects of pharmacologically heterogeneous groups of psychotropic drugs on NGF contents in the brain regions involved in the modulation of emotions are summarized. Moreover, the therapeutic potentials of the endocannabinoid system which is linked to depression and/or antidepressant effects and appears to interact with neurotrophin signalling, are reviewed. New findings demonstrate that endocannabinoid system is involved in the mechanisms of action of certain psychotropic medications including neurokinin receptor antagonists and that these are mediated via the upregulation of brain regional levels of NGF. This provides a better understanding of the pathophysi- ological mechanisms underlying neuropsychiatric disorders, leading to novel drug designs. Biomed Rev 2010; 21: 31-46. Key words: endocannabinoids, NGF, psychotropics, brain INTRODUCTION glucocorticoid activity are involved in the pathophysiology Depression is a serious and widespread mental disorder with of depression (2-4). However, the monoamine-based anti- high relapse rate which is characterized by an array of dis- depressants do not fulfil the expectations in terms of onset turbances in emotional behavior, memory, neurovegetative of action, efficacy, and tolerability. -

Pharmacological Enhancement of Cannabinoid CB1 Receptor Activity Elicits an Antidepressant-Like Response in the Rat Forced Swim Test
European Neuropsychopharmacology 15 (2005) 593 – 599 www.elsevier.com/locate/euroneuro Pharmacological enhancement of cannabinoid CB1 receptor activity elicits an antidepressant-like response in the rat forced swim test Matthew N. Hill, Boris B. Gorzalka* Department of Psychology, University of British Columbia, Vancouver, 2136 West Mall, Canada V6T 1Z4 Received 15 October 2004; received in revised form 1 February 2005; accepted 22 March 2005 Abstract These experiments aimed to assess whether enhanced activity at the cannabinoid CB1 receptor elicits antidepressant-like effects. To examine this we administered 1 and 5 mg/kg doses of the endocannabinoid uptake inhibitor AM404; 5 and 25 Ag/kg doses of HU-210, a potent CB1 receptor agonist; 1, 2.5 and 5 mg/kg of oleamide, which elicits cannabinoidergic actions; 1 and 5 mg/kg doses of AM 251, a selective CB1 receptor antagonist, as well as 10 mg/kg desipramine (a positive antidepressant control) and measured the duration of immobility, during a 5-min test session of the rat Porsolt forced swim test. Results demonstrated that administration of desipramine reduced immobility duration by about 50% and that all of AM404, oleamide and HU-210 administration induced comparable decreases in immobility that were blocked by pretreatment with AM 251. Administration of the antagonist AM 251 alone had no effect on immobility at either dose. These data suggest that enhancement of CB1 receptor signaling results in antidepressant effects in the forced swim test similar to that seen following conventional antidepressant administration. D 2005 Elsevier B.V. and ECNP. All rights reserved. Keywords: Cannabinoid; Antidepressant; Forced swim test; Oleamide; Rat 1. -

The Effects of Cannabinoids on Stress-Coping
Utah State University DigitalCommons@USU All Graduate Theses and Dissertations Graduate Studies 5-2021 The Effects of Cannabinoids on Stress-Coping Behaviors and Neuroendocrinological Measures in Chronic Unpredtictable Stress: A Preclinical Systematic Review and Meta-Analysis Noa Reuveni Utah State University Follow this and additional works at: https://digitalcommons.usu.edu/etd Part of the Psychology Commons Recommended Citation Reuveni, Noa, "The Effects of Cannabinoids on Stress-Coping Behaviors and Neuroendocrinological Measures in Chronic Unpredtictable Stress: A Preclinical Systematic Review and Meta-Analysis" (2021). All Graduate Theses and Dissertations. 8098. https://digitalcommons.usu.edu/etd/8098 This Thesis is brought to you for free and open access by the Graduate Studies at DigitalCommons@USU. It has been accepted for inclusion in All Graduate Theses and Dissertations by an authorized administrator of DigitalCommons@USU. For more information, please contact [email protected]. THE EFFECTS OF CANNABINOIDS ON STRESS-COPING BEHAVIORS AND NEUROENDOCRINOLOGICAL MEASURES IN CHRONIC UNPREDTICTABLE STRESS: A PRECLINICAL SYSTEMATIC REVIEW AND META-ANALYSIS by Noa Reuveni A thesis submitted in partial fulfillment of the requirements for the degree of MASTER OF SCIENCE in Psychology Approved: __________________ __________________ Sara Freeman, Ph.D. Scott Bates, Ph.D. Co-Major Professor Co-Major Professor __________________ __________________ Diana Meter, Ph.D. Sarah Schwartz, Ph.D. Committee Member Committee Member __________________ __________________ Tyler Renshaw, Ph.D. D. Richard Cutler, Ph.D. Committee Member Interim Vice Provost for Graduate Studies UTAH STATE UNIVERSITY Logan, Utah 2021 ii Copyright © Noa Reuveni 2021 All Rights Reserved iii ABSTRACT The Effects of Cannabinoids on Stress-Coping Behaviors and Neuroendocrinological Measures in Chronic Unpredictable Stress: A Preclinical Systematic Review and Meta-Analysis by Noa Reuveni, Master of Science Utah State University, 2021 Major Professors: Dr. -

Influence of the CB1 Cannabinoid Receptors on the Activity of the Monoaminergic System in the Behavioural Tests in Mice
Accepted Manuscript Title: Influence of the CB1 cannabinoid receptors on the activity of the monoaminergic system in the behavioural tests in mice Authors: Ewa Poleszak, Sylwia Wosko,´ Karolina Sławinska,´ Elzbieta˙ Wyska, Aleksandra Szopa, Urszula Doboszewska, Piotr Wlaz,´ Aleksandra Wlaz,´ Jarosław Dudka, Jarosław Szponar, Anna Serefko PII: S0361-9230(19)30298-9 DOI: https://doi.org/10.1016/j.brainresbull.2019.05.021 Reference: BRB 9698 To appear in: Brain Research Bulletin Received date: 16 April 2019 Revised date: 28 May 2019 Accepted date: 29 May 2019 Please cite this article as: Poleszak E, Wosko´ S, Sławinska´ K, Wyska E, Szopa A, Doboszewska U, WlazP,Wla´ z´ A, Dudka J, Szponar J, Serefko A, Influence of the CB1 cannabinoid receptors on the activity of the monoaminergic system in the behavioural tests in mice, Brain Research Bulletin (2019), https://doi.org/10.1016/j.brainresbull.2019.05.021 This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain. Influence of the CB1 cannabinoid receptors on the activity of the monoaminergic system in the behavioural tests in mice Ewa Poleszak 1,*, Sylwia Wośko 1, Karolina Sławińska 1, Elżbieta Wyska -
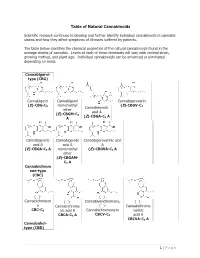
Table of Natural Cannabinoids
Table of Natural Cannabinoids Scientific research continues to develop and further identify individual cannabinoids in cannabis strains and how they affect symptoms of illnesses suffered by patients. The table below identifies the chemical properties of the natural cannabinoids found in the average strains of cannabis. Levels of each of these chemicals will vary with varietal strain, growing method, and plant age. Individual cannabinoids can be enhanced or eliminated depending on need. Cannabigerol- type (CBG) Cannabigerol Cannabigerol Cannabigerovarin (E)-CBG-C monomethyl (E)-CBGV-C 5 Cannabinerolic 3 ether acid A (E)-CBGM-C 5 (Z)-CBGA-C A A 5 Cannabigerolic Cannabigerolic Cannabigerovarinic acid acid A acid A A (E)-CBGA-C5 A monomethyl (E)-CBGVA-C3 A ether (E)-CBGAM- C5 A Cannabichrom ene-type (CBC) (±)- (±)- Cannabichromen (±)- Cannabivarichromene, (±)- e Cannabichrome (±)- Cannabichrome CBC-C5 nic acid A Cannabichromevarin varinic CBCA-C5 A CBCV-C3 acid A CBCVA-C3 A Cannabidiol- type (CBD) 1 | Page (−)-Cannabidiol Cannabidiol Cannabidiol-C4 (−)- Cannabidiorc CBD-C5 momomethyl CBD-C4 Cannabidivarin ol ether CBDV-C3 CBD-C1 CBDM-C5 Cannabidiolic Cannabidivarini acid c acid CBDA-C5 CBDVA-C3 Cannabinodiol- type (CBND) Cannabinodiol Cannabinodivar CBND-C5 in CBND-C3 Tetrahydrocan nabinol-type (THC) 9 9 9 Δ - Δ - Δ - Δ9- Tetrahydrocanna Tetrahydrocan Tetrahydrocannabivarin 9 Tetrahydrocan binol nabinol-C4 Δ -THCV-C3 9 9 nabiorcol Δ -THC-C5 Δ -THC-C4 9 Δ -THCO-C1 9 9 Δ -Tetrahydro- Δ9-Tetrahydro- Δ -Tetrahydro- Δ9-Tetrahydro- cannabinolic -

Cannabinoid Receptor Antagonists Counteract Sensorimotor Gating Deficits in the Phencyclidine Model of Psychosis
Neuropsychopharmacology (2007) 32, 2098–2107 & 2007 Nature Publishing Group All rights reserved 0893-133X/07 $30.00 www.neuropsychopharmacology.org Cannabinoid Receptor Antagonists Counteract Sensorimotor Gating Deficits in the Phencyclidine Model of Psychosis ,1 2,3 4 5 3,6 Martina Ballmaier* , Marco Bortolato , Cristina Rizzetti , Michele Zoli , GianLuigi Gessa , 1,6 4,6 Andreas Heinz and PierFranco Spano 1Department of Psychiatry and Psychotherapy, Charite´ University Medicine, Campus Mitte, Berlin, Germany; 2Department of Pharmacology, 3 4 University of California, Irvine, USA; Department of Neuroscience, Bernard B Brodie, University of Cagliari, Cagliari, Italy; Department of 5 Biomedical Sciences and Biotechnologies, Brescia University Medical School, Brescia, Italy; Department of Biomedical Sciences, University of Modena and Reggio Emilia, Modena, Italy Clinical and laboratory findings suggest that cannabinoids and their receptors are implicated in schizophrenia. The role of cannabinoids in schizophrenia remains however poorly understood, as data are often contradictory. The primary aim of this study was to investigate whether the cannabinoid CB1 receptor antagonists rimonabant and AM251 are able to reverse deficits of sensorimotor gating induced by phencyclidine and to mimic the ‘atypical’ antipsychotic profile of clozapine. The prepulse inhibition (PPI) of the startle reflex was used to measure deficits of sensorimotor gating. PPI-disruptive effects of phencyclidine and their antagonism by rimonabant, AM251, and clozapine were studied in rats. The effects of rimonabant were carefully examined taking into account dose ranges, vehicle, and route of administration. We also examined the ability of rimonabant to reduce the PPI-disruptive effects of dizocilpine and apomorphine. Rimonabant as well as AM251 significantly counteracted the phencyclidine-disruptive model of PPI, comparable to the restoring effect of clozapine; no augmentation effect was observed with rimonabant and clozapine as cotreatment.