And Hippo Signaling-Dependent Plasticity During Lineage
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Primepcr™Assay Validation Report
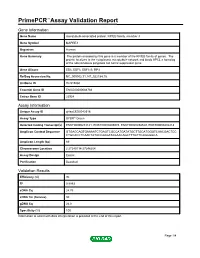
PrimePCR™Assay Validation Report Gene Information Gene Name microtubule-associated protein, RP/EB family, member 3 Gene Symbol MAPRE3 Organism Human Gene Summary The protein encoded by this gene is a member of the RP/EB family of genes. The protein localizes to the cytoplasmic microtubule network and binds APCL a homolog of the adenomatous polyposis coli tumor suppressor gene. Gene Aliases EB3, EBF3, EBF3-S, RP3 RefSeq Accession No. NC_000002.11, NT_022184.15 UniGene ID Hs.515860 Ensembl Gene ID ENSG00000084764 Entrez Gene ID 22924 Assay Information Unique Assay ID qHsaCED0042518 Assay Type SYBR® Green Detected Coding Transcript(s) ENST00000233121, ENST00000405074, ENST00000458529, ENST00000402218 Amplicon Context Sequence GTGACCAGTGAAAATCTGAGTCGCCATGATATGCTTGCATGGGTCAACGACTCC CTGCACCTCAACTATACCAAGATAGAACAGCTTTGTTCAGGGGCA Amplicon Length (bp) 69 Chromosome Location 2:27245114-27246204 Assay Design Exonic Purification Desalted Validation Results Efficiency (%) 90 R2 0.9993 cDNA Cq 24.76 cDNA Tm (Celsius) 80 gDNA Cq 26.8 Specificity (%) 100 Information to assist with data interpretation is provided at the end of this report. Page 1/4 PrimePCR™Assay Validation Report MAPRE3, Human Amplification Plot Amplification of cDNA generated from 25 ng of universal reference RNA Melt Peak Melt curve analysis of above amplification Standard Curve Standard curve generated using 20 million copies of template diluted 10-fold to 20 copies Page 2/4 PrimePCR™Assay Validation Report Products used to generate validation data Real-Time PCR Instrument CFX384 Real-Time PCR Detection System Reverse Transcription Reagent iScript™ Advanced cDNA Synthesis Kit for RT-qPCR Real-Time PCR Supermix SsoAdvanced™ SYBR® Green Supermix Experimental Sample qPCR Human Reference Total RNA Data Interpretation Unique Assay ID This is a unique identifier that can be used to identify the assay in the literature and online. -

AIRE Is a Critical Spindle-Associated Protein in Embryonic Stem Cells Bin Gu1, Jean-Philippe Lambert2, Katie Cockburn1, Anne-Claude Gingras2,3, Janet Rossant1,3*
RESEARCH ARTICLE AIRE is a critical spindle-associated protein in embryonic stem cells Bin Gu1, Jean-Philippe Lambert2, Katie Cockburn1, Anne-Claude Gingras2,3, Janet Rossant1,3* 1Program in Developmental and Stem Cell Biology, Hospital for Sick Children, Toronto, Canada; 2Lunenfeld-Tanenbaum Research Institute at Mount Sinai Hospital, Toronto, Canada; 3Department of Molecular Genetics, University of Toronto, Toronto, Canada Abstract Embryonic stem (ES) cells go though embryo-like cell cycles regulated by specialized molecular mechanisms. However, it is not known whether there are ES cell-specific mechanisms regulating mitotic fidelity. Here we showed that Autoimmune Regulator (Aire), a transcription coordinator involved in immune tolerance processes, is a critical spindle-associated protein in mouse ES(mES) cells. BioID analysis showed that AIRE associates with spindle-associated proteins in mES cells. Loss of function analysis revealed that Aire was important for centrosome number regulation and spindle pole integrity specifically in mES cells. We also identified the c-terminal LESLL motif as a critical motif for AIRE’s mitotic function. Combined maternal and zygotic knockout further revealed Aire’s critical functions for spindle assembly in preimplantation embryos. These results uncovered a previously unappreciated function for Aire and provide new insights into the biology of stem cell proliferation and potential new angles to understand fertility defects in humans carrying Aire mutations. DOI: 10.7554/eLife.28131.001 *For correspondence: janet. [email protected] Introduction Competing interests: The Self-renewal capability, defined as the ability of cells to proliferate while sustaining differentiation authors declare that no potential, is one of the defining features of stem cells (Martello and Smith, 2014). -

Beacon in Developmental Biology
NEWS Beacon in developmental biology n Cite as: CMAJ 2017 October 16;189:E1298-9. doi: 10.1503/cmaj.109-5460 Posted on cmajnews.com on September 27, 2017. he identification of Gurdon, whom some people the genes responsi- may know recently received ble for controlling the Nobel Prize for his work Tcellular fate and embryonic cloning frogs; undertaking development in mice has research that demonstrates made Dr. Janet Rossant a that all of development is beacon in the field of written in our genes, and all developmental biology. of our cells contain DNA that Her work pioneered tech- has that information, yet niques that allowed genes somehow during develop- to be altered within ment, different genes are embryos. It also acceler- turned on and differentiation ated avenues in the fields occurs. That’s what really of stem cell biology and turned me on to the underly- regenerative medicine. ing problem of ‘how do we She was awarded the get from a fertilized egg to an 2015 Canada Gairdner organism?’ John is the per- Wightman Award, pre- son who really turned me on sented annually to a Cana- to that question and he has dian who has demon- been pursuing it ever since strated outstanding and so have I.” leadership in medicine and medical science. Rossant For the diverse audience was recognized “for her who read CMAJ, can you outstanding contributions explain what stem cells to developmental biology are? and for her exceptional “Stem cells are cells that international leadership in have the ability to self- stem cell biology and pol- renew, make endless copies icy-making, and in advanc- of themselves and yet also ing research programs for retain the potential to differ- children’s illnesses.” Courtesy of Dr. -

Annual Report Fy 2018 Human Frontier Science Program Organization
APRIL 2017 APRIL 2018 — MARCH 2019 ANNUAL REPORT FY 2018 HUMAN FRONTIER SCIENCE PROGRAM ORGANIZATION The Human Frontier Science Program Organization (HFSPO) is unique, supporting international collaboration to undertake innovative, risky, basic research at the frontier of the life sciences. Special emphasis is given to the support and training of independent young investigators, beginning at the postdoctoral level. The Program is implemented by an international organisation, supported financially by Australia, Canada, France, Germany, India, Italy, Japan, the Republic of Korea, New Zealand, Norway, Singapore, Switzerland, the United Kingdom of Great Britain and Nothern Ireland, the United States of America, and the European Commission. Since 1990, over 7000 researchers from more than 70 countries have been supported. Of these, 28 HFSP awardees have gone on to receive the Nobel Prize. 2 The following documents are available on the HFSP website www.hfsp.org: Joint Communiqués (Tokyo 1992, Washington 1997, Berlin 2002, Bern 2004, Ottawa 2007, Canberra 2010, Brussels 2013, London 2016): https://www.hfsp.org/about/governance/membership Statutes of the International Human Frontier Science Program Organization: https://www.hfsp.org/about/governance/hfspo-statutes Guidelines for the participation of new members in HFSPO: https://www.hfsp.org/about/governance/membership General reviews of the HFSP (1996, 2001, 2006-2007, 2010, 2018): https://www.hfsp.org/about/strategy/reviews Updated and previous lists of awards, including titles and abstracts: -

Balancing Potency and Differentiation in Mouse Embryo-Derived Stem Cells and in Vivo
Balancing potency and differentiation in mouse embryo-derived stem cells and in vivo By Anne Christine Helness A thesis submitted to Imperial College London for the degree of Doctor of Philosophy Department of Surgery and Oncology Imperial College London September 2011 1 Abstract An inherent challenge for developing organisms is to maintain a critical balance between cell potency and differentiation. This is most obvious in embryonic stem (ES) cells where genetic, epigenetic and cell signalling pathways support ES cell ability to self-renew and to generate all embryonic lineages. Recently, a series of reports revealed how Polycomb-mediated repression might buffer the precocious expression of somatic lineage regulators in ES cells. Notably, these genes carry bivalent chromatin enriched in both repressive (H3K27me3) and permissive (H3K4me2) histone marks. They are targeted by the Polycomb repressive complexes PRC1 (Ring1B) and PRC2, which mediates H3K27me3 and assemble poised RNA polymerase II (RNAP II), conferring silencing of loci primed for future activation (or repression) upon ES cell differentiation. During early development the transition from morula to blastocyst is the starting point for lineage segregation into the inner cell mass (ICM) and trophectoderm (TE). ES cells are derived from the ICM and are pluripotent. By contrast, TE-derived trophoblast stem (TS) cells are multipotent and contribute solely to placenta formation in vivo. To address whether ES cells epigenetic features are unique attributes of pluripotent cells in the early embryo, we compared the epigenetic status of key developmental genes in blastocyst-derived stem cells and in vivo. We provide direct evidence that bivalent histone markings operate in vivo from the eight-cell up to the blastocyst stage. -

EB3 (MAPRE3) (NM 012326) Human Tagged ORF Clone Product Data
OriGene Technologies, Inc. 9620 Medical Center Drive, Ste 200 Rockville, MD 20850, US Phone: +1-888-267-4436 [email protected] EU: [email protected] CN: [email protected] Product datasheet for RC204135 EB3 (MAPRE3) (NM_012326) Human Tagged ORF Clone Product data: Product Type: Expression Plasmids Product Name: EB3 (MAPRE3) (NM_012326) Human Tagged ORF Clone Tag: Myc-DDK Symbol: MAPRE3 Synonyms: EB3; EBF3; EBF3-S; RP3 Vector: pCMV6-Entry (PS100001) E. coli Selection: Kanamycin (25 ug/mL) Cell Selection: Neomycin ORF Nucleotide >RC204135 ORF sequence Sequence: Red=Cloning site Blue=ORF Green=Tags(s) TTTTGTAATACGACTCACTATAGGGCGGCCGGGAATTCGTCGACTGGATCCGGTACCGAGGAGATCTGCC GCCGCGATCGCC ATGGCCGTCAATGTGTACTCCACATCTGTGACCAGTGAAAATCTGAGTCGCCATGATATGCTTGCATGGG TCAACGACTCCCTGCACCTCAACTATACCAAGATAGAACAGCTTTGTTCAGGGGCAGCCTACTGCCAGTT CATGGACATGCTCTTCCCCGGCTGTGTGCACTTGAGGAAAGTGAAGTTCCAGGCCAAACTAGAGCATGAA TACATCCACAACTTCAAGGTGCTGCAAGCAGCTTTCAAGAAGATGGGTGTTGACAAAATCATTCCTGTAG AGAAATTAGTGAAAGGAAAATTCCAAGATAATTTTGAGTTTATTCAGTGGTTTAAGAAATTCTTTGACGC AAACTATGATGGAAAGGATTACAACCCTCTGCTGGCGCGGCAGGGCCAGGACGTAGCGCCACCTCCTAAC CCAGGTGATCAGATCTTCAACAAATCCAAGAAACTCATTGGCACAGCAGTTCCACAGAGGACGTCCCCCA CAGGCCCAAAAAACATGCAGACCTCTGGCCGGCTGAGCAATGTGGCCCCCCCCTGCATTCTCCGGAAGAA TCCTCCATCAGCCCGAAATGGCGGCCATGAGACTGATGCCCAAATTCTTGAACTCAACCAACAGCTGGTG GACTTGAAGCTGACAGTGGATGGGCTGGAGAAGGAACGTGACTTCTACTTCAGCAAACTTCGTGACATCG AGCTCATCTGCCAGGAGCATGAAAGTGAAAACAGCCCTGTTATCTCAGGCATCATTGGCATCCTCTATGC CACAGAGGAAGGATTCGCACCCCCTGAGGACGATGAGATTGAAGAGCATCAACAAGAAGACCAGGACGAG TAC ACGCGTACGCGGCCGCTCGAGCAGAAACTCATCTCAGAAGAGGATCTGGCAGCAAATGATATCCTGGATT -

Supporting Information
Supporting Information Pouryahya et al. SI Text Table S1 presents genes with the highest absolute value of Ricci curvature. We expect these genes to have significant contribution to the network’s robustness. Notably, the top two genes are TP53 (tumor protein 53) and YWHAG gene. TP53, also known as p53, it is a well known tumor suppressor gene known as the "guardian of the genome“ given the essential role it plays in genetic stability and prevention of cancer formation (1, 2). Mutations in this gene play a role in all stages of malignant transformation including tumor initiation, promotion, aggressiveness, and metastasis (3). Mutations of this gene are present in more than 50% of human cancers, making it the most common genetic event in human cancer (4, 5). Namely, p53 mutations play roles in leukemia, breast cancer, CNS cancers, and lung cancers, among many others (6–9). The YWHAG gene encodes the 14-3-3 protein gamma, a member of the 14-3-3 family proteins which are involved in many biological processes including signal transduction regulation, cell cycle pro- gression, apoptosis, cell adhesion and migration (10, 11). Notably, increased expression of 14-3-3 family proteins, including protein gamma, have been observed in a number of human cancers including lung and colorectal cancers, among others, suggesting a potential role as tumor oncogenes (12, 13). Furthermore, there is evidence that loss Fig. S1. The histogram of scalar Ricci curvature of 8240 genes. Most of the genes have negative scalar Ricci curvature (75%). TP53 and YWHAG have notably low of p53 function may result in upregulation of 14-3-3γ in lung cancer Ricci curvatures. -

EMBO Encounters Issue43.Pdf
WINTER 2019/2020 ISSUE 43 Nine group leaders selected Meet the first EMBO Global Investigators PAGE 6 Accelerating scientific publishing EMBO publishing costs Review Commons Making our journals’ platform announced finances public PAGE 3 PAGES 10 – 11 Welcome, Young Investigators! Contract replaces stipend Marking ten years 27 group leaders join the programme EMBO Postdoctoral Fellowships EMBO Molecular Medicine receive an update celebrates anniversary PAGES 4 – 5 PAGE 7 PAGE 13 www.embo.org TABLE OF CONTENTS EMBO NEWS EMBO news Review Commons: accelerating publishing Page 3 EMBO Molecular Medicine turns ten © Marietta Schupp, EMBL Photolab Marietta Schupp, © Page 13 Editorial MBO was founded by scientists for Introducing 27 new Young Investigators scientists. This philosophy remains at Pages 4-5 Ethe heart of our organization until today. EMBO Members are vital in the running of our Meet the first EMBO Global programmes and activities: they screen appli- Accelerating scientific publishing 17 journals on board Investigators cations, interview candidates, decide on fund- Review Commons will manage the transfer of ing, and provide strategic direction. On pages EMBO and ASAPbio announced pre-journal portable review platform the manuscript, reviews, and responses to affili- Page 6 8-9 four members describe why they chose to ate journals. A consortium of seventeen journals New members meet in Heidelberg dedicate their time to an EMBO Committee across six publishers (see box) have joined the Fellowships: from stipends to contracts Pages 14 – 15 and what they took away from the experience. n December 2019, EMBO, in partnership with decide to submit their work to a journal, it will project by committing to use the Review Commons Page 7 When EMBO was created, the focus lay ASAPbio, launched Review Commons, a multi- allow editors to make efficient editorial decisions referee reports for their independent editorial deci- specifically on fostering cross-border inter- Ipublisher partnership which aims to stream- based on existing referee comments. -

Dr. Janet Rossant ANDREW F. HOLMES DEAN of MEDICINE DISTINCTION LECTURES
Dr. Janet Rossant ANDREW F. HOLMES DEAN OF MEDICINE DISTINCTION LECTURES Dr. Janet Rossant, PhD, FRS, FRSC Chief of Research, SickKids Senior Scientist, Developmental & Stem Cell Biology, SickKids Lombard Insurance Chair in Pediatric Research University Professor, Department of Molecular Genetics, Obstetrics and Gynecology, University of Toronto Title Stem cells and early lineage development When Friday, October 18, 2013 11:30 a.m. – 1:00 p.m. Where CP Leblond Amphitheatre Strathcona Anatomy and Dentistry Building 3640 rue University Montreal, QC H3G 1Y6 Dr. Janet Rossant, SickKids Chief of Research and a world-renowned expert in developmental biology, is the definition of a trailblazer. Widely known for her studies of the genes that control embryonic development in the mouse, Dr. Rossant has pioneered techniques for following cell fate and altering genes in embryos. This work continues to resonate in medical genetic research. Her current research focuses on stem cell development and cell differentiation in the developing embryo, important areas for the study of birth defects as well as regenerative medicine. Firmly planted on the front lines of technological change, Dr. Rossant has established SickKids as a global forerunner in genetic research. Janet Rossant was born in Chatham, UK, and trained at the Universities of Oxford and Cambridge, United Kingdom. In 1977, Dr. Rossant moved to Canada and joined the faculty at Brock University. From 1985 to 2005, she was a leading researcher at the Samuel Lunenfeld Research Institute at Mount Sinai Hospital in Toronto. She joined SickKids in 2005 and became Chief of Research of the Research Institute. She is also a University Professor at the University of Toronto. -

In This Table Protein Name, Uniprot Code, Gene Name P-Value
Supplementary Table S1: In this table protein name, uniprot code, gene name p-value and Fold change (FC) for each comparison are shown, for 299 of the 301 significantly regulated proteins found in both comparisons (p-value<0.01, fold change (FC) >+/-0.37) ALS versus control and FTLD-U versus control. Two uncharacterized proteins have been excluded from this list Protein name Uniprot Gene name p value FC FTLD-U p value FC ALS FTLD-U ALS Cytochrome b-c1 complex P14927 UQCRB 1.534E-03 -1.591E+00 6.005E-04 -1.639E+00 subunit 7 NADH dehydrogenase O95182 NDUFA7 4.127E-04 -9.471E-01 3.467E-05 -1.643E+00 [ubiquinone] 1 alpha subcomplex subunit 7 NADH dehydrogenase O43678 NDUFA2 3.230E-04 -9.145E-01 2.113E-04 -1.450E+00 [ubiquinone] 1 alpha subcomplex subunit 2 NADH dehydrogenase O43920 NDUFS5 1.769E-04 -8.829E-01 3.235E-05 -1.007E+00 [ubiquinone] iron-sulfur protein 5 ARF GTPase-activating A0A0C4DGN6 GIT1 1.306E-03 -8.810E-01 1.115E-03 -7.228E-01 protein GIT1 Methylglutaconyl-CoA Q13825 AUH 6.097E-04 -7.666E-01 5.619E-06 -1.178E+00 hydratase, mitochondrial ADP/ATP translocase 1 P12235 SLC25A4 6.068E-03 -6.095E-01 3.595E-04 -1.011E+00 MIC J3QTA6 CHCHD6 1.090E-04 -5.913E-01 2.124E-03 -5.948E-01 MIC J3QTA6 CHCHD6 1.090E-04 -5.913E-01 2.124E-03 -5.948E-01 Protein kinase C and casein Q9BY11 PACSIN1 3.837E-03 -5.863E-01 3.680E-06 -1.824E+00 kinase substrate in neurons protein 1 Tubulin polymerization- O94811 TPPP 6.466E-03 -5.755E-01 6.943E-06 -1.169E+00 promoting protein MIC C9JRZ6 CHCHD3 2.912E-02 -6.187E-01 2.195E-03 -9.781E-01 Mitochondrial 2- -

Web Conference with Dr. Janet Rossant
Web Conference with Dr. Janet Rossant - 00:00 Thank you for taking the time to speak with us, so obviously we'd like to ask you some questions about your work and your research, but can you tell us about yourself and your research, how you got into stem cells in the first place? - 00:16 Okay, so I've been interested in early development for a very long time, sort of all my research career. I started as a graduate student at the University of Cambridge with Richard Gardner who was one of the first people who developed techniques to inject cells into blastocysts to make chimeras, and so I was always interested in how an egg develops into a complex organism, and over the years I've worked on the cell lineage in the early embryo. We've then been able to move in and identify some of the genes and pathways that specifies cell fate in the early embryo, and of course along the way, stem cells come into the picture because it's from the early mammalian embryo, the blastocysts, that you can derive embryonic stem cells, and it's really the work that I and my colleagues did early on in the embryo showing that there was a subset of cells that were pluripotent that gave people the idea that maybe you could capture that pluripotency and derive permanent cell lines from those cells and grow them in cultures. So we've worked on mouse embryonic stem cells, but we also have been able to show that we can derive stem cells from the other two types of the blastocysts, the trophoblast and the primitive endoderm, so in the mouse you can have three stem cell lines from the blastocysts, each of which replicates the properties of those lineages in vivo. -

Pnas11052ackreviewers 5098..5136
Acknowledgment of Reviewers, 2013 The PNAS editors would like to thank all the individuals who dedicated their considerable time and expertise to the journal by serving as reviewers in 2013. Their generous contribution is deeply appreciated. A Harald Ade Takaaki Akaike Heather Allen Ariel Amir Scott Aaronson Karen Adelman Katerina Akassoglou Icarus Allen Ido Amit Stuart Aaronson Zach Adelman Arne Akbar John Allen Angelika Amon Adam Abate Pia Adelroth Erol Akcay Karen Allen Hubert Amrein Abul Abbas David Adelson Mark Akeson Lisa Allen Serge Amselem Tarek Abbas Alan Aderem Anna Akhmanova Nicola Allen Derk Amsen Jonathan Abbatt Neil Adger Shizuo Akira Paul Allen Esther Amstad Shahal Abbo Noam Adir Ramesh Akkina Philip Allen I. Jonathan Amster Patrick Abbot Jess Adkins Klaus Aktories Toby Allen Ronald Amundson Albert Abbott Elizabeth Adkins-Regan Muhammad Alam James Allison Katrin Amunts Geoff Abbott Roee Admon Eric Alani Mead Allison Myron Amusia Larry Abbott Walter Adriani Pietro Alano Isabel Allona Gynheung An Nicholas Abbott Ruedi Aebersold Cedric Alaux Robin Allshire Zhiqiang An Rasha Abdel Rahman Ueli Aebi Maher Alayyoubi Abigail Allwood Ranjit Anand Zalfa Abdel-Malek Martin Aeschlimann Richard Alba Julian Allwood Beau Ances Minori Abe Ruslan Afasizhev Salim Al-Babili Eric Alm David Andelman Kathryn Abel Markus Affolter Salvatore Albani Benjamin Alman John Anderies Asa Abeliovich Dritan Agalliu Silas Alben Steven Almo Gregor Anderluh John Aber David Agard Mark Alber Douglas Almond Bogi Andersen Geoff Abers Aneel Aggarwal Reka Albert Genevieve Almouzni George Andersen Rohan Abeyaratne Anurag Agrawal R. Craig Albertson Noga Alon Gregers Andersen Susan Abmayr Arun Agrawal Roy Alcalay Uri Alon Ken Andersen Ehab Abouheif Paul Agris Antonio Alcami Claudio Alonso Olaf Andersen Soman Abraham H.