Molecular Systematics of Marine Gregarines (Apicomplexa)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

An Intestinal Gregarine of Nothria Conchylega (Polychaeta, Onuphidae)
Journal of Invertebrate Pathology 104 (2010) 172–179 Contents lists available at ScienceDirect Journal of Invertebrate Pathology journal homepage: www.elsevier.com/locate/jip Description of Trichotokara nothriae n. gen. et sp. (Apicomplexa, Lecudinidae) – An intestinal gregarine of Nothria conchylega (Polychaeta, Onuphidae) Sonja Rueckert *, Brian S. Leander Canadian Institute for Advanced Research, Program in Integrated Microbial Biodiversity, Departments of Botany and Zoology, University of British Columbia, #3529 – 6270 University Blvd., Vancouver, BC, Canada V6T 1Z4 article info abstract Article history: The trophozoites of a novel gregarine apicomplexan, Trichotokara nothriae n. gen. et sp., were isolated and Received 12 November 2009 characterized from the intestines of the onuphid tubeworm Nothria conchylega (Polychaeta), collected at Accepted 11 March 2010 20 m depth from the North-eastern Pacific Coast. The trophozoites were 50–155 lm long with a mid-cell Available online 23 March 2010 indentation that formed two prominent bulges (anterior bulge, 14–48 lm wide; posterior bulge, 15– 55 lm wide). Scanning electron microscopy (SEM) demonstrated that approximately 400 densely packed, Keywords: longitudinal epicytic folds (5 folds/lm) inscribe the surface of the trophozoites, and a prominently elon- Alveolata gated mucron (14–60 lm long and 6–12 lm wide) was covered with hair-like projections (mean length, Apicomplexa 1.97 m; mean width, 0.2 m at the base). Although a septum occurred at the junction between the cell Lecudinidae l l Lecudina proper and the mucron in most trophozoites, light microscopy (LM) demonstrated that the cell proper Parasite extended into the core of the mucron as a thin prolongation. -

Phylum Nemertea)
THE BIOLOGY AND SYSTEMATICS OF A NEW SPECIES OF RIBBON WORM, GENUS TUBULANUS (PHYLUM NEMERTEA) By Rebecca Kirk Ritger Submitted to the Faculty of the College of Arts and Sciences of American University in Partial Fulfillment of the Requirements for the Degree of Master of Science In Biology Chair: Dr. Qiristopher'Tudge m Dr.David C r. Jon L. Norenburg Dean of the College of Arts and Sciences JuK4£ __________ Date 2004 American University Washington, D.C. 20016 AMERICAN UNIVERSITY LIBRARY 1 1 0 Reproduced with permission of the copyright owner. Further reproduction prohibited without permission. UMI Number: 1421360 INFORMATION TO USERS The quality of this reproduction is dependent upon the quality of the copy submitted. Broken or indistinct print, colored or poor quality illustrations and photographs, print bleed-through, substandard margins, and improper alignment can adversely affect reproduction. In the unlikely event that the author did not send a complete manuscript and there are missing pages, these will be noted. Also, if unauthorized copyright material had to be removed, a note will indicate the deletion. ® UMI UMI Microform 1421360 Copyright 2004 by ProQuest Information and Learning Company. All rights reserved. This microform edition is protected against unauthorized copying under Title 17, United States Code. ProQuest Information and Learning Company 300 North Zeeb Road P.O. Box 1346 Ann Arbor, Ml 48106-1346 Reproduced with permission of the copyright owner. Further reproduction prohibited without permission. THE BIOLOGY AND SYSTEMATICS OF A NEW SPECIES OF RIBBON WORM, GENUS TUBULANUS (PHYLUM NEMERTEA) By Rebecca Kirk Ritger ABSTRACT Most nemerteans are studied from poorly preserved museum specimens. -
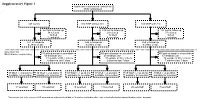
Supplementary Figure 1 Multicenter Randomised Control Trial 2746 Randomised
Supplementary Figure 1 Multicenter randomised control trial 2746 randomised 947 control 910 MNP without zinc 889 MNP with zinc 223 lost to follow up 219 lost to follow up 183 lost to follow up 34 refused 29 refused 37 refused 16 died 12 died 9 died 3 excluded 4 excluded 1 excluded 671 in follow-up 646 in follow-up 659 in follow-up at 24mo of age at 24mo of age at 24mo of age Selection for Microbiome sequencing 516 paired samples unavailable 469 paired samples unavailable 497 paired samples unavailable 69 antibiotic use 63 antibiotic use 67 antibiotic use 31 outside of WLZ criteria 37 outside of WLZ criteria 34 outside of WLZ criteria 6 diarrhea last 7 days 2 diarrhea last 7 days 7 diarrhea last 7 days 39 WLZ > -1 at 24 mo 10 WLZ < -2 at 24mo 58 WLZ > -1 at 24 mo 17 WLZ < -2 at 24mo 48 WLZ > -1 at 24 mo 8 WLZ < -2 at 24mo available for selection available for selection available for selection available for selection available for selection1 available for selection1 14 selected 10 selected 15 selected 14 selected 20 selected1 7 selected1 1 Two subjects (one in the reference WLZ group and one undernourished) had, at 12 months, no diarrhea within 1 day of stool collection but reported diarrhea within 7 days prior. Length, cm kg Weight, Supplementary Figure 2. Length (left) and weight (right) z-scores of children recruited into clinical trial NCT00705445 during the first 24 months of life. Median and quantile values are shown, with medians for participants profiled in current study indicated by red (undernourished) and black (reference WLZ) lines. -

Why the –Omic Future of Apicomplexa Should Include Gregarines Julie Boisard, Isabelle Florent
Why the –omic future of Apicomplexa should include Gregarines Julie Boisard, Isabelle Florent To cite this version: Julie Boisard, Isabelle Florent. Why the –omic future of Apicomplexa should include Gregarines. Biology of the Cell, Wiley, 2020, 10.1111/boc.202000006. hal-02553206 HAL Id: hal-02553206 https://hal.archives-ouvertes.fr/hal-02553206 Submitted on 24 Apr 2020 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Article title: Why the –omic future of Apicomplexa should include Gregarines. Names of authors: Julie BOISARD1,2 and Isabelle FLORENT1 Authors affiliations: 1. Molécules de Communication et Adaptation des Microorganismes (MCAM, UMR 7245), Département Adaptations du Vivant (AVIV), Muséum National d’Histoire Naturelle, CNRS, CP52, 57 rue Cuvier 75231 Paris Cedex 05, France. 2. Structure et instabilité des génomes (STRING UMR 7196 CNRS / INSERM U1154), Département Adaptations du vivant (AVIV), Muséum National d'Histoire Naturelle, CP 26, 57 rue Cuvier 75231 Paris Cedex 05, France. Short Title: Gregarines –omics for Apicomplexa studies -

Tracing the Origin of Planktonic Protists in an Ancient Lake
microorganisms Article Tracing the Origin of Planktonic Protists in an Ancient Lake Nataliia V. Annenkova 1,* , Caterina R. Giner 2,3 and Ramiro Logares 2,* 1 Limnological Institute Siberian Branch of the Russian Academy of Sciences 3, Ulan-Batorskaya St., 664033 Irkutsk, Russia 2 Institute of Marine Sciences (ICM), CSIC, Passeig Marítim de la Barceloneta, 37-49, ES08003 Barcelona, Spain; [email protected] 3 Institute for the Oceans and Fisheries, University of British Columbia, 2202 Main Mall, Vancouver, BC V6T 1Z4, Canada * Correspondence: [email protected] (N.V.A.); [email protected] (R.L.) Received: 26 February 2020; Accepted: 7 April 2020; Published: 9 April 2020 Abstract: Ancient lakes are among the most interesting models for evolution studies because their biodiversity is the result of a complex combination of migration and speciation. Here, we investigate the origin of single celled planktonic eukaryotes from the oldest lake in the world—Lake Baikal (Russia). By using 18S rDNA metabarcoding, we recovered 1414 Operational Taxonomic Units (OTUs) belonging to protists populating surface waters (1–50 m) and representing pico/nano-sized cells. The recovered communities resembled other lacustrine freshwater assemblages found elsewhere, especially the taxonomically unclassified protists. However, our results suggest that a fraction of Baikal protists could belong to glacial relicts and have close relationships with marine/brackish species. Moreover, our results suggest that rapid radiation may have occurred among some protist taxa, partially mirroring what was already shown for multicellular organisms in Lake Baikal. We found 16% of the OTUs belonging to potential species flocks in Stramenopiles, Alveolata, Opisthokonta, Archaeplastida, Rhizaria, and Hacrobia. -

Tubulanus Polymorphus Class: Anopla Order: Paleonemertea an Orange Ribbon Worm Family: Tubulanidae
Phylum: Nemertea Tubulanus polymorphus Class: Anopla Order: Paleonemertea An orange ribbon worm Family: Tubulanidae “Such a worm when seen crawling in long and Palaeonemertea) but with lateral graceful curves over the bottom in clear water transverse grooves (Fig. 2a, b, c). earns for itself a place among the most Head cannot completely withdraw into body (Kozloff 1974). beautiful of all marine invertebrates” (Coe Posterior: No caudal cirrus. 1905) Eyes/Eyespots: None. Mouth: A long slit-like opening (Fig. 2c) Taxonomy: Tubulanus polymorphous was a posterior to the brain, separate from name assigned in unpublished work by proboscis pore (Fig. 2c) and positioned just Renier (1804). The genera Tubulanus and behind transverse furrows (Coe 1901). Carinella were described by Renier (1804) Proboscis: Eversible (phylum Nemertea) and Johnston (1833), respectively, and were and, when not everted, coiled inside synonymized by Bürger in 1904 (Gibson rhynchocoel (cavity). The proboscis in 1995). Melville (1986) and the International Tubulanus polymorphus is short with the Code of Zoological Nomenclature (ICZN) rhynchocoel reaching one third total worm determined that the family name Tubulanidae body length. Proboscis bears no stylets and take precedence over its senior subjective the proboscis pore almost terminal (Fig. 2c). synonym Carinellidae (Ritger and Norenburg Tube/Burrow: As is true for most Tubulanus 2006) and the name Tubulanus polymorphus species, T. polymorphus individuals live in was deemed published and available (ICZN thin parchment tubes that are attached to 1988). Previous names for T. polymorphus rocks or shells and made of hardened include C. polymorpha, C. rubra and C. mucous secretions (Coe 1943). speciosa. Possible Misidentifications Description The genus Tubulanus is slender, soft, Size: A large nemertean, up to three meters extensible without ocelli or cephalic grooves when extended. -

Assessing the Efficiency of Molecular Markers for the Species Identification of Gregarines Isolated from the Mealworm and Super Worm Midgut
Microorganisms 2018, 6, 119 1 of 20 Article Assessing the Efficiency of Molecular Markers for the Species Identification of Gregarines Isolated from the Mealworm and Super Worm Midgut Chiara Nocciolini 1, Claudio Cucini 2, Chiara Leo 2, Valeria Francardi 3, Elena Dreassi 4 and Antonio Carapelli 2,* 1 Polo d’Innovazione di Genomica Genetica e Biologia, 53100 Siena, Italy; [email protected] 2 Department of Life Sciences, University of Siena, 53100 Siena, Italy; [email protected] (C.C.); [email protected] (C.L.) 3 Consiglio per la ricerca in agricoltura e analisi dell’economia agraria – Centro di Difesa e Certificazione CREA-DC) Research Centre for Plant Protection and Certification, via di Lanciola 12/A, 50125 Firenze, Italy; [email protected] 4 Department of Biotechnology, Chemistry and Pharmacy, University of Siena, 53100 Siena, Italy; [email protected] * Correspondence: [email protected]; Tel.: +39-0577-234-410 Received: 28 September 2018; Accepted: 23 November 2018; Published: 27 November 2018 Abstract: Protozoa, of the taxon Gregarinasina, are a heterogeneous group of Apicomplexa that includes ~1600 species. They are parasites of a large variety of both marine and terrestrial invertebrates, mainly annelids, arthropods and mollusks. Unlike coccidians and heamosporidians, gregarines have not proven to have a negative effect on human welfare; thus, they have been poorly investigated. This study focuses on the molecular identification and phylogeny of the gregarine species found in the midgut of two insect species that are considered as an alternative source of animal proteins for the human diet: the mealworm Tenebrio molitor, and the super-worm Zophobas atratus (Coleoptera: Tenebrionidae). -

OREGON ESTUARINE INVERTEBRATES an Illustrated Guide to the Common and Important Invertebrate Animals
OREGON ESTUARINE INVERTEBRATES An Illustrated Guide to the Common and Important Invertebrate Animals By Paul Rudy, Jr. Lynn Hay Rudy Oregon Institute of Marine Biology University of Oregon Charleston, Oregon 97420 Contract No. 79-111 Project Officer Jay F. Watson U.S. Fish and Wildlife Service 500 N.E. Multnomah Street Portland, Oregon 97232 Performed for National Coastal Ecosystems Team Office of Biological Services Fish and Wildlife Service U.S. Department of Interior Washington, D.C. 20240 Table of Contents Introduction CNIDARIA Hydrozoa Aequorea aequorea ................................................................ 6 Obelia longissima .................................................................. 8 Polyorchis penicillatus 10 Tubularia crocea ................................................................. 12 Anthozoa Anthopleura artemisia ................................. 14 Anthopleura elegantissima .................................................. 16 Haliplanella luciae .................................................................. 18 Nematostella vectensis ......................................................... 20 Metridium senile .................................................................... 22 NEMERTEA Amphiporus imparispinosus ................................................ 24 Carinoma mutabilis ................................................................ 26 Cerebratulus californiensis .................................................. 28 Lineus ruber ......................................................................... -

Gregarines (Apicomplexa: Gregarinasina) Are Monoxenous Parasites of Invertebrates
Gregarines (Apicomplexa: Gregarinasina) are monoxenous parasites of invertebrates. Those found in sand flies (Diptera: Psychodidae) and mosquitoes (Diptera: Culicidae) used to be considered a single eugregarine genus Ascogregarina. Our phylogenetic analyses of the gregarine SSU rDNA, including newly obtained sequences of three species from sand flies, showed that mosquito and sand fly gregarines are closely related to neogregarines, and most importantly, they form two disparate monophyletic groups. Based on these molecular features, accompanied by biological differences, we established a new genus Psychodiella for the gregarines from sand flies, reserving the genus Ascogregarina for the mosquito gregarines. In the new genus, two new species Psychodiella sergenti from Phlebotomus sergenti and Psychodiella tobbi from Phlebotomus tobbi were described. They differ in the life cycles (sexual development of Ps. sergenti is triggered by a blood meal intake) and morphology of their life stages, mainly oocysts. The susceptibility of five sand fly species to both gregarines showed their strict host specificity, as they were able to fully develop and complete the life cycle only in their natural hosts. The life cycle of Ps. sergenti was studied in detail using various microscopical methods. Oocysts are attached to the chorion of sand fly eggs. Sporozoites, with a three- layered pellicle and mucron, attach to the 1st instar larval intestine but are never located intracellularly. In the 4th instar larvae, the gregarines occur in the ectoperitrophic space and later in the intestinal lumen. In adults, the parasites appear in the body cavity, and the sexual development of Ps. sergenti takes place only in blood-fed females; gametocysts attach to the accessory glands and oocysts are injected into their lumen. -

A Brief Taxonomy of the Genus Prorocentrum in the Coastal Areas Along Sanya Bay, Hainan Island
R ESEARCH ARTICLE ScienceAsia 44 (2018): 241-246 doi:10.2306 /scienceasia1513-1874.2018.44.241 A brief taxonomy of the genus Prorocentrum in the coastal areas along Sanya Bay, Hainan Island a,b, c a d Anqi Yin ∗, Xinghua Wang ,Yajian Zhang , Hongwu Li a College of Life Sciences and Ecology, Hainan Tropical Ocean University, Sanya, Hainan 572022 China b School of Environmental Science, The University of Shiga Prefecture, Hikone, Shiga 522-8533 Japan c School of Ocean Science and Technology, Hainan Tropical Ocean University, Sanya, Hainan 572022 China d State Key Laboratory of Marine Resource Utilization in South China Sea, Haikou, Hainan 570228 China ∗Corresponding author, e-mail: [email protected] Received 7 May 2018 Accepted 14 Aug 2018 ABSTRACT: This is the first report of the genus Prorocentrum Ehrenberg occurring in the coastal areas along Sanya Bay, Hainan Island. Samples were collected from five coastal stations in 2017, and a brief taxonomy of Prorocentrum species was carried out using an inverted microscope. Five species (P.hoffmannianum, P.lima, P.micans, P.rhathymum, P.sigmoides) were identified, including three toxic species and two red tide-forming species. In particular, P. hoffmannianum and P. lima were both confirmed as the producers of okadaic acid and responsible for diarrhoetic shellfish poisoning events. This may suggest the potential threats of harmful algal blooms in the coastal areas along Sanya Bay that could cause diarrhoetic shellfish poisoning events. KEYWORDS: dinoflagellates, planktonic, benthic, toxic, red tide-forming INTRODUCTION land, is located in a tropical climate region, where shellfish industries, fish farming, and tourism have Dinoflagellates are an important part of the pelagic rapidly developed in the coastal areas during the ecosystem 1, make a large component of primary last decade. -

The Classification of Lower Organisms
The Classification of Lower Organisms Ernst Hkinrich Haickei, in 1874 From Rolschc (1906). By permission of Macrae Smith Company. C f3 The Classification of LOWER ORGANISMS By HERBERT FAULKNER COPELAND \ PACIFIC ^.,^,kfi^..^ BOOKS PALO ALTO, CALIFORNIA Copyright 1956 by Herbert F. Copeland Library of Congress Catalog Card Number 56-7944 Published by PACIFIC BOOKS Palo Alto, California Printed and bound in the United States of America CONTENTS Chapter Page I. Introduction 1 II. An Essay on Nomenclature 6 III. Kingdom Mychota 12 Phylum Archezoa 17 Class 1. Schizophyta 18 Order 1. Schizosporea 18 Order 2. Actinomycetalea 24 Order 3. Caulobacterialea 25 Class 2. Myxoschizomycetes 27 Order 1. Myxobactralea 27 Order 2. Spirochaetalea 28 Class 3. Archiplastidea 29 Order 1. Rhodobacteria 31 Order 2. Sphaerotilalea 33 Order 3. Coccogonea 33 Order 4. Gloiophycea 33 IV. Kingdom Protoctista 37 V. Phylum Rhodophyta 40 Class 1. Bangialea 41 Order Bangiacea 41 Class 2. Heterocarpea 44 Order 1. Cryptospermea 47 Order 2. Sphaerococcoidea 47 Order 3. Gelidialea 49 Order 4. Furccllariea 50 Order 5. Coeloblastea 51 Order 6. Floridea 51 VI. Phylum Phaeophyta 53 Class 1. Heterokonta 55 Order 1. Ochromonadalea 57 Order 2. Silicoflagellata 61 Order 3. Vaucheriacea 63 Order 4. Choanoflagellata 67 Order 5. Hyphochytrialea 69 Class 2. Bacillariacea 69 Order 1. Disciformia 73 Order 2. Diatomea 74 Class 3. Oomycetes 76 Order 1. Saprolegnina 77 Order 2. Peronosporina 80 Order 3. Lagenidialea 81 Class 4. Melanophycea 82 Order 1 . Phaeozoosporea 86 Order 2. Sphacelarialea 86 Order 3. Dictyotea 86 Order 4. Sporochnoidea 87 V ly Chapter Page Orders. Cutlerialea 88 Order 6. -

Molecular Phylogeny and Ultrastructure of Selenidium Serpulae
MolecularBlackwell Publishing Ltd phylogeny and ultrastructure of Selenidium serpulae (Apicomplexa, Archigregarinia) from the calcareous tubeworm Serpula vermicularis (Annelida, Polychaeta, Sabellida) BRIAN S. LEANDER Accepted: 23 November 2006 Leander, B. S. (2007). Molecular phylogeny and ultrastructure of Selenidium serpulae (Apicom- doi:10.1111/j.1463-6409.2007.00272.x plexa, Archigregarinia) from the calcareous tubeworm Serpula vermicularis (Annelida, Polychaeta, Sabellida). — Zoologica Scripta, 36, 213–227. Archigregarines are dynamic single-celled parasites that inhabit the intestinal systems of marine invertebrates, especially suspension feeding and deposit feeding polychaetes. Certain archigregarines in the genus Selenidium have retained several plesiomorphic characters, and improved knowledge of these species is expected to shed considerable light onto the earliest stages in apicomplexan evolution. Although archigregarines are related to some of the most notorious parasites known (e.g., Cryptosporidium and Plasmodium), current knowledge of the group is meagre. In an attempt to improve our understanding of archigregarine diversity and evolution, I have characterised the general ultrastructure and molecular phylogenetic position of Selenidium serpulae (Lankester) Caullery and Mesnil. The parasites were isolated from the intestines of the calcareous tubeworm Serpula vermicularis (Polychaeta) collected in the east- ern Pacific Ocean. The trophozoites (extracellular feeding stages) were spindle-shaped and capable of slow and continuous bending, and coiling, especially when dislodged from the host epithelium. The trophozoite surface was composed of 19–23 longitudinal folds, prominent transverse folds and a robust, trilayered pellicle subtended by a single row of microtubules, each surrounded by an electron transparent sheath. Putative mitochondria were observed, but they were inconspicuous and apparently highly reduced, a condition that is indicative of ana- erobic metabolism.