Degradation of Plectin with Modulation of Cytokeratin 18 In
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Desmin Interacts Directly with Mitochondria
International Journal of Molecular Sciences Article Desmin Interacts Directly with Mitochondria Alexander A. Dayal 1, Natalia V. Medvedeva 1, Tatiana M. Nekrasova 1, Sergey D. Duhalin 1, Alexey K. Surin 1,2 and Alexander A. Minin 1,* 1 Institute of Protein Research of Russian Academy of Sciences, Vavilova st., 34, 119334 Moscow, Russia; [email protected] (A.A.D.); [email protected] (N.V.M.); [email protected] (T.M.N.); [email protected] (S.D.D.); [email protected] (A.K.S.) 2 Pushchino Branch, Shemyakin–Ovchinnikov Institute of Bioorganic Chemistry, Russian Academy of Sciences, Prospekt Nauki 6, Pushchino, 142290 Moscow Region, Russia * Correspondence: [email protected] Received: 14 October 2020; Accepted: 26 October 2020; Published: 30 October 2020 Abstract: Desmin intermediate filaments (IFs) play an important role in maintaining the structural and functional integrity of muscle cells. They connect contractile myofibrils to plasma membrane, nuclei, and mitochondria. Disturbance of their network due to desmin mutations or deficiency leads to an infringement of myofibril organization and to a deterioration of mitochondrial distribution, morphology, and functions. The nature of the interaction of desmin IFs with mitochondria is not clear. To elucidate the possibility that desmin can directly bind to mitochondria, we have undertaken the study of their interaction in vitro. Using desmin mutant Des(Y122L) that forms unit-length filaments (ULFs) but is incapable of forming long filaments and, therefore, could be effectively separated from mitochondria by centrifugation through sucrose gradient, we probed the interaction of recombinant human desmin with mitochondria isolated from rat liver. Our data show that desmin can directly bind to mitochondria, and this binding depends on its N-terminal domain. -

PSPC1 Potentiates IGF1R Expression to Augment Cell Adhesion and Motility
1 Supplementary information 2 PSPC1 potentiates IGF1R expression to augment cell 3 adhesion and motility 4 Hsin-Wei Jen1,2 , De-Leung Gu 2, Yaw-Dong Lang 2 and Yuh-Shan Jou 1,2,* 5 1 Graduate Institute of Life Sciences, National Defense Medical Center, Taipei, Taiwan 6 2 Institute of Biomedical Sciences, Academia Sinica, Taipei, Taiwan 7 * Author to whom correspondence should be addressed 8 Cells 2020, 9, x; doi: FOR PEER REVIEW www.mdpi.com/journal/cells Cells 2020, 9, x FOR PEER REVIEW 2 of 10 9 10 11 Supplementary Figure S1: Expression of IGF1R and integrin in PSPC1-expressing or PSPC1-depleted 12 HCC cells by Western blotting analysis 13 (A) Detection of IGF1R protein levels in three PSPC1-knockdown cells Huh7, HepG2 and Mahlavu. (B) 14 Detection of selected integrin expression in PSPC1-overexpressing or PSPC1-depleted HCC cells by using 15 their total cell lysates immunoblotted with specific integrin antibodies as shown. 16 17 18 Supplementary Figure S2: PSPC1-modulated IGF1R downstream signaling in HCC cells. Cells 2020, 9, x FOR PEER REVIEW 3 of 10 19 (A, B) Immunoblotting of IGF1R expression in PSPC1-overexpressing SK-Hep1 and PLC5 cells 20 treated with IGF1R shRNAs. (C, D) Cell migration and adhesion were measured in PSPC1- 21 knockdown Hep3B cells rescued with exogenous expression of IGF1R. Exogenous expression of 22 IGF1R in PSPC1-knockdown Hep3B cells were then applied for detection of altered AKT/ERK 23 signaling including (E) total PSPC1, IGF1R, AKT, ERK, p-IGF1R, p-AKT(S473), and 24 p-ERK(T202/Y204) as well as altered FAK/Src signaling including (F) total FAK, Src, p-FAK(Y397) 25 and p-Src(Y416) by immunoblotting assay. -

In Situlocalization of Cytoskeletal Elements in the Human Trabecular
Investigative Ophthalmology & Visual Science. Vol. 31. No. 9. September 1990 Cops right £• Association lor Research in Vision and Ophthalmology In Situ Localization of Cytoskeletal Elements in the Human Trabecular Meshwork and Cornea Robert N. Weinreb* and Mark I. Ryderf The authors compared cytoskeletal elements of the in situ human trabccular-mcshwork cell with in situ human corneal cells using indirect immunofluorcsccncc staining for tubulin and intermediate filaments (vimentin, cytokeratin, and desmin) and NBD-phallacidin staining for f-actin using both fixed frozen and unfixed frozen sections from postmortem eyes. Both f-actin and tubulin were found throughout the cell body of trabecular-meshwork cells, keratocytes, corneal endothelium, and corneal epithelium. The f-actin staining pattern was concentrated at the cell periphery of these four cell types. Vimentin stain was intensely localized in focal areas of the trabecular-meshwork cell, keratocytes, and throughout the corneal cndothelium. A general anticytokeratin antibody was intensely localized in corneal epithelium and endothelium. However, PKK-1 anticytokeratin antibody was seen only in superficial layers of corneal epithelium and not in corneal endothelium. The 4.62 anticytokeratin antibody was not observed in either corneal epithelium or endothelium. None of these three cytokera- tin antibodies were seen in trabccular-mcshwork cells or keratocytes. Desmin stain was not noted in any of these cell types. In general, cytoskeletal staining of unfixed frozen sections showed a similar staining pattern for f-actin and tubulin but a more uniform and intense staining pattern for vimentin and cytokcratin compared with fixed frozen material. The authors conclude that these cytoskclctal stains can differentiate human Irabeciilar-meshwork cells from cells of the cornea in situ. -

Human Plectin: Organization of the Gene, Sequence Analysis, and Chromosome Localization (8Q24) CHANG-GONG LIU*, CHRISTIAN MAERCKER*, MARIA J
Proc. Natl. Acad. Sci. USA Vol. 93, pp. 4278-4283, April 1996 Biochemistry Human plectin: Organization of the gene, sequence analysis, and chromosome localization (8q24) CHANG-GONG LIU*, CHRISTIAN MAERCKER*, MARIA J. CASTANONt, RUDOLF HAUPTMANNt, AND GERHARD WICHE* *Institute of Biochemistry and Molecular Cell Biology, University of Vienna-Biocenter, 1030 Vienna, Austria; and tErnst Boehringer Institut, 1121 Vienna, Austria Communicated by Gottfried Schatz, Biozentrum der Universitat Basel, Basel, Switzerland, January 2, 1996 (received for review August 28, 1995) ABSTRACT Plectin, a 500-kDa intermediate filament C-terminal globular domain of the molecule (9). The overex- binding protein, has been proposed to provide mechanical pression of plectin mutant proteins containing this site(s) has strength to cells and tissues by acting as a cross-linking a dramatic dominant negative effect on cells, causing the total element of the cytoskeleton. To set the basis for future studies collapse of cytoplasmic IF networks. Based on the combined on gene regulation, tissue-specific expression, and patholog- information available to date, plectin has been proposed to ical conditions involving this protein, we have cloned the play a key role as a versatile cross-linking element of the human plectin gene, determined its coding sequence, and cytoskeleton. Particularly, because of its strategic localization established its genomic organization. The coding sequence at the cytoskeleton-plasma membrane interface, such as in all contains 32 exons that extend -

Decreased Expression of Profilin 2 in Oral Squamous Cell Carcinoma and Its Clinicopathological Implications
ONCOLOGY REPORTS 26: 813-823, 2011 Decreased expression of profilin 2 in oral squamous cell carcinoma and its clinicopathological implications C.Y. MA1,2, C.P. ZHANG1,2, L.P. ZHONG1,2, H.Y. PAN1,2, W.T. CHEN1,2, L.Z. WANG3, O.W. ANDREW4, T. JI1 and W. HAN1,2 1Department of Oral and Maxillofacial Surgery, Ninth People's Hospital, College of Stomatology; 2Shanghai Key Laboratory of Stomatology and Shanghai Research Institute of Stomatology; 3Department of Oral Pathology, Ninth People's Hospital, College of Stomatology, Shanghai Jiao Tong University School of Medicine, Shanghai 200011, P.R. China; 4Department of Oral and Maxillofacial Surgery, Faculty of Dentistry, National University of Singapore, Singapore 119074, Singapore Received February 8, 2011; Accepted April 11, 2011 DOI: 10.3892/or.2011.1365 Abstract. Profilins are small proteins essential for many clinical and pathological significance. In conclusion, PFN2 normal cellular dynamics and constitute one of the crucial can be utilized as both a potential suppressor marker and a components of actin-based cellular motility. Several recent prognostic protein for OSCC. The function of PFN2 may be to studies have implicated a role for the profilin (PFN) family in regulate the N-WASP/Arp2/3 signaling pathway. cancer pathogenesis and progression. However, their expression and promising functions are largely unknown in oral squamous Introduction cell carcinoma (OSCC). In this study, we analyzed the correlation between PFN1 and PFN2 expression in vitro and Oral squamous cell carcinoma (OSCC) is a significant public in vivo. The protein expression levels were roughly compared health problem with >300,000 new cases being diagnosed between cell lines (HIOEC, HB96) with the employment of annually worldwide (1). -

Crosstalk Between Mitochondria and Cytoskeleton in Cardiac Cells
cells Review Crosstalk between Mitochondria and Cytoskeleton in Cardiac Cells Andrey V. Kuznetsov 1,2,*, Sabzali Javadov 3 , Michael Grimm 1, Raimund Margreiter 4, Michael J. Ausserlechner 2 and Judith Hagenbuchner 5,* 1 Cardiac Surgery Research Laboratory, Department of Cardiac Surgery, Innsbruck Medical University, 6020 Innsbruck, Austria; [email protected] 2 Department of Paediatrics I, Medical University of Innsbruck, 6020 Innsbruck, Austria; [email protected] 3 Department of Physiology, School of Medicine, University of Puerto Rico, San Juan, PR 00936-5067, USA; [email protected] 4 Department of Visceral, Transplant and Thoracic Surgery, Medical University of Innsbruck, 6020 Innsbruck, Austria; [email protected] 5 Department of Paediatrics II, Medical University of Innsbruck, 6020 Innsbruck, Austria * Correspondence: [email protected] (A.V.K.); [email protected] (J.H.); Tel.: +43-512-504-27815 (A.V.K.); +43-512-504-81578 (J.H.) Received: 3 December 2019; Accepted: 13 January 2020; Published: 16 January 2020 Abstract: Elucidation of the mitochondrial regulatory mechanisms for the understanding of muscle bioenergetics and the role of mitochondria is a fundamental problem in cellular physiology and pathophysiology. The cytoskeleton (microtubules, intermediate filaments, microfilaments) plays a central role in the maintenance of mitochondrial shape, location, and motility. In addition, numerous interactions between cytoskeletal proteins and mitochondria can actively participate in the regulation of mitochondrial respiration and oxidative phosphorylation. In cardiac and skeletal muscles, mitochondrial positions are tightly fixed, providing their regular arrangement and numerous interactions with other cellular structures such as sarcoplasmic reticulum and cytoskeleton. -
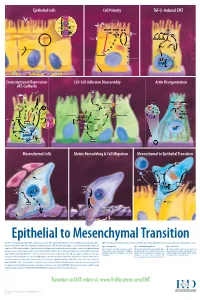
Epithelial to Mesenchymal Transition
Epithelial Cells Cell Polarity TGF-b-Induced EMT MUC-1 O-glycosylation Epithelial Cells ZO-1 Occludin Apical Membrane Tight F-Actin Microvilli Junction Claudin F-Actin p120 β-Catenin Adherens F-Actin Ezrin TGF-β dimer Junction E-Cadherin α-Catenin Plakophilin Crumbs Complex PAR Complex Desmocollin Desmoplakin Desmosome PtdIns(4,5)P2 TGF-β RII TGF-β RI CRB Cdc42Par6 Desmoglein Cytokeratin Pals1 PatJ Tight Junction Plakoglobin aPKC Par3 Domain Smad7 Extracellular PTEN JNK ERK1/2 p38 SARA Smurf1 Cortical Actin Cytoskeleton Space Par3 ZO-1 Adherens Junction PI 3-K Domain Smad-independent Signaling (–) Smad7 Translocation Smad2/3 PtdIns(3,4,5)P3 Smad4 Smad4 NEDD4 Cytokeratin Intermediate Filaments Smad2 Smad4 Smad3 LLGL Proteasome SCRIB DLG Scribble Complex Fibronectin Twist Smad2/3 Vitronectin ZEB 1/2 Microtubule Network Smad4 N-Cadherin Snail Basolateral Membrane CoA, Collagen I Slug CoR MMPs DNA-binding (+) Claudin Desmoplakin Transcription Factor Occludin Cytokeratins E-Cadherin Plakoglobin Integrins β α Nidogen-1/Entactin Perlecan Laminin Collagen IV Transcriptional Repression Cell-Cell Adhesion Disassembly Actin Reorganization of E-Cadherin TGF-β dimer EGF TGF-β RII TGF-β RI IGF FGF Receptor TNF-α Tyrosine Kinase Par6 TNF RI Apical Focal Adhesion Constriction Actin Depolymerization F-Actin Smurf1 Occludin Wnt Frizzled Myosin II Ras RhoA α-Actinin Myosin II ROCK AxinCK1 Dishevelled GSK-3 PI 3-K Src Zyxin MLC Phosphatase APC Proteasome FAK Vinculin RhoA ILK Talin (Inactive) Hakai Talin FAK F-Actin E-Cadherin LIMK Akt Paxillin FAK Stress -

Plakoglobin Is Required for Effective Intermediate Filament Anchorage to Desmosomes Devrim Acehan1, Christopher Petzold1, Iwona Gumper2, David D
ORIGINAL ARTICLE Plakoglobin Is Required for Effective Intermediate Filament Anchorage to Desmosomes Devrim Acehan1, Christopher Petzold1, Iwona Gumper2, David D. Sabatini2, Eliane J. Mu¨ller3, Pamela Cowin2,4 and David L. Stokes1,2,5 Desmosomes are adhesive junctions that provide mechanical coupling between cells. Plakoglobin (PG) is a major component of the intracellular plaque that serves to connect transmembrane elements to the cytoskeleton. We have used electron tomography and immunolabeling to investigate the consequences of PG knockout on the molecular architecture of the intracellular plaque in cultured keratinocytes. Although knockout keratinocytes form substantial numbers of desmosome-like junctions and have a relatively normal intercellular distribution of desmosomal cadherins, their cytoplasmic plaques are sparse and anchoring of intermediate filaments is defective. In the knockout, b-catenin appears to substitute for PG in the clustering of cadherins, but is unable to recruit normal levels of plakophilin-1 and desmoplakin to the plaque. By comparing tomograms of wild type and knockout desmosomes, we have assigned particular densities to desmoplakin and described their interaction with intermediate filaments. Desmoplakin molecules are more extended in wild type than knockout desmosomes, as if intermediate filament connections produced tension within the plaque. On the basis of our observations, we propose a particular assembly sequence, beginning with cadherin clustering within the plasma membrane, followed by recruitment of plakophilin and desmoplakin to the plaque, and ending with anchoring of intermediate filaments, which represents the key to adhesive strength. Journal of Investigative Dermatology (2008) 128, 2665–2675; doi:10.1038/jid.2008.141; published online 22 May 2008 INTRODUCTION dense plaque that is further from the membrane and that Desmosomes are large macromolecular complexes that mediates the binding of intermediate filaments. -

Transiently Structured Head Domains Control Intermediate Filament Assembly
Transiently structured head domains control intermediate filament assembly Xiaoming Zhoua, Yi Lina,1, Masato Katoa,b,c, Eiichiro Morid, Glen Liszczaka, Lillian Sutherlanda, Vasiliy O. Sysoeva, Dylan T. Murraye, Robert Tyckoc, and Steven L. McKnighta,2 aDepartment of Biochemistry, University of Texas Southwestern Medical Center, Dallas, TX 75390; bInstitute for Quantum Life Science, National Institutes for Quantum and Radiological Science and Technology, 263-8555 Chiba, Japan; cLaboratory of Chemical Physics, National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda, MD 20892-0520; dDepartment of Future Basic Medicine, Nara Medical University, 840 Shijo-cho, Kashihara, Nara, Japan; and eDepartment of Chemistry, University of California, Davis, CA 95616 Contributed by Steven L. McKnight, January 2, 2021 (sent for review October 30, 2020; reviewed by Lynette Cegelski, Tatyana Polenova, and Natasha Snider) Low complexity (LC) head domains 92 and 108 residues in length are, IF head domains might facilitate filament assembly in a manner respectively, required for assembly of neurofilament light (NFL) and analogous to LC domain function by RNA-binding proteins in the desmin intermediate filaments (IFs). As studied in isolation, these IF assembly of RNA granules. head domains interconvert between states of conformational disor- IFs are defined by centrally located α-helical segments 300 to der and labile, β-strand–enriched polymers. Solid-state NMR (ss-NMR) 350 residues in length. These central, α-helical segments are spectroscopic studies of NFL and desmin head domain polymers re- flanked on either end by head and tail domains thought to be veal spectral patterns consistent with structural order. -

Cytoskeletal Linkers: New Maps for Old Destinations Megan K
R864 Dispatch Cytoskeletal linkers: New MAPs for old destinations Megan K. Houseweart*† and Don W. Cleveland*†‡§ A new isoform of the actin–neurofilament linker protein as ‘bullous pemphigoid antigen’ (BPAG). These proteins BPAG has been found that binds to and stabilizes are large α-helical coiled-coil molecules which have axonal microtubules. This and other newly identified binding domains for one or more of the three cytoskele- microtubule-associated proteins are likely to be just the tal components (Figure 1). For example, the widely tip of an iceberg of multifunctional proteins that expressed, > 500 kD protein plectin has been shown to stabilize and crosslink cytoskeletal filament networks. associate with microtubules, intermediate filaments (glial fibrillary acidic protein, vimentin, keratins, all Addresses: *Ludwig Institute for Cancer Research, †Program in Biomedical Sciences, ‡Division of Cellular and Molecular Medicine and three neurofilament subunit proteins), actin, myosin and §Department of Neuroscience, University of California at San Diego, itself [3]. Given the widespread distribution and multi- La Jolla, California 92093, USA. ple interactions that are characteristic of these proteins, E-mail: [email protected] it is not surprising that a number of human and mouse Current Biology 1999, 9:R864–R866 diseases have been attributed to aberrant or missing cross-linking proteins [4]. 0960-9822/99/$ – see front matter © 1999 Elsevier Science Ltd. All rights reserved. This is the case for mice lacking the locus encoding the numerous isoforms of the essential ~280 kDa linker The cytoplasm of most eukaryotic cells contains a dynamic protein BPAG. Two neuronal isoforms of BPAG both have filamentous protein scaffold composed of 25 nm micro- a carboxy-terminal intermediate-filament-binding domain tubules, 4 nm actin filaments and 10 nm intermediate fila- and also an amino-terminal actin-binding region (Figure 1). -
HCC and Cancer Mutated Genes Summarized in the Literature Gene Symbol Gene Name References*
HCC and cancer mutated genes summarized in the literature Gene symbol Gene name References* A2M Alpha-2-macroglobulin (4) ABL1 c-abl oncogene 1, receptor tyrosine kinase (4,5,22) ACBD7 Acyl-Coenzyme A binding domain containing 7 (23) ACTL6A Actin-like 6A (4,5) ACTL6B Actin-like 6B (4) ACVR1B Activin A receptor, type IB (21,22) ACVR2A Activin A receptor, type IIA (4,21) ADAM10 ADAM metallopeptidase domain 10 (5) ADAMTS9 ADAM metallopeptidase with thrombospondin type 1 motif, 9 (4) ADCY2 Adenylate cyclase 2 (brain) (26) AJUBA Ajuba LIM protein (21) AKAP9 A kinase (PRKA) anchor protein (yotiao) 9 (4) Akt AKT serine/threonine kinase (28) AKT1 v-akt murine thymoma viral oncogene homolog 1 (5,21,22) AKT2 v-akt murine thymoma viral oncogene homolog 2 (4) ALB Albumin (4) ALK Anaplastic lymphoma receptor tyrosine kinase (22) AMPH Amphiphysin (24) ANK3 Ankyrin 3, node of Ranvier (ankyrin G) (4) ANKRD12 Ankyrin repeat domain 12 (4) ANO1 Anoctamin 1, calcium activated chloride channel (4) APC Adenomatous polyposis coli (4,5,21,22,25,28) APOB Apolipoprotein B [including Ag(x) antigen] (4) AR Androgen receptor (5,21-23) ARAP1 ArfGAP with RhoGAP domain, ankyrin repeat and PH domain 1 (4) ARHGAP35 Rho GTPase activating protein 35 (21) ARID1A AT rich interactive domain 1A (SWI-like) (4,5,21,22,24,25,27,28) ARID1B AT rich interactive domain 1B (SWI1-like) (4,5,22) ARID2 AT rich interactive domain 2 (ARID, RFX-like) (4,5,22,24,25,27,28) ARID4A AT rich interactive domain 4A (RBP1-like) (28) ARID5B AT rich interactive domain 5B (MRF1-like) (21) ASPM Asp (abnormal -

Differential Gene Expression in Oligodendrocyte Progenitor Cells, Oligodendrocytes and Type II Astrocytes
Tohoku J. Exp. Med., 2011,Differential 223, 161-176 Gene Expression in OPCs, Oligodendrocytes and Type II Astrocytes 161 Differential Gene Expression in Oligodendrocyte Progenitor Cells, Oligodendrocytes and Type II Astrocytes Jian-Guo Hu,1,2,* Yan-Xia Wang,3,* Jian-Sheng Zhou,2 Chang-Jie Chen,4 Feng-Chao Wang,1 Xing-Wu Li1 and He-Zuo Lü1,2 1Department of Clinical Laboratory Science, The First Affiliated Hospital of Bengbu Medical College, Bengbu, P.R. China 2Anhui Key Laboratory of Tissue Transplantation, Bengbu Medical College, Bengbu, P.R. China 3Department of Neurobiology, Shanghai Jiaotong University School of Medicine, Shanghai, P.R. China 4Department of Laboratory Medicine, Bengbu Medical College, Bengbu, P.R. China Oligodendrocyte precursor cells (OPCs) are bipotential progenitor cells that can differentiate into myelin-forming oligodendrocytes or functionally undetermined type II astrocytes. Transplantation of OPCs is an attractive therapy for demyelinating diseases. However, due to their bipotential differentiation potential, the majority of OPCs differentiate into astrocytes at transplanted sites. It is therefore important to understand the molecular mechanisms that regulate the transition from OPCs to oligodendrocytes or astrocytes. In this study, we isolated OPCs from the spinal cords of rat embryos (16 days old) and induced them to differentiate into oligodendrocytes or type II astrocytes in the absence or presence of 10% fetal bovine serum, respectively. RNAs were extracted from each cell population and hybridized to GeneChip with 28,700 rat genes. Using the criterion of fold change > 4 in the expression level, we identified 83 genes that were up-regulated and 89 genes that were down-regulated in oligodendrocytes, and 92 genes that were up-regulated and 86 that were down-regulated in type II astrocytes compared with OPCs.