Moths Postfire to Feb16 2021 by Family
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Self-Repair and Self-Cleaning of the Lepidopteran Proboscis
Clemson University TigerPrints All Dissertations Dissertations 8-2019 Self-Repair and Self-Cleaning of the Lepidopteran Proboscis Suellen Floyd Pometto Clemson University, [email protected] Follow this and additional works at: https://tigerprints.clemson.edu/all_dissertations Recommended Citation Pometto, Suellen Floyd, "Self-Repair and Self-Cleaning of the Lepidopteran Proboscis" (2019). All Dissertations. 2452. https://tigerprints.clemson.edu/all_dissertations/2452 This Dissertation is brought to you for free and open access by the Dissertations at TigerPrints. It has been accepted for inclusion in All Dissertations by an authorized administrator of TigerPrints. For more information, please contact [email protected]. SELF-REPAIR AND SELF-CLEANING OF THE LEPIDOPTERAN PROBOSCIS A Dissertation Presented to the Graduate School of Clemson University In Partial Fulfillment of the Requirements for the Degree Doctor of Philosophy ENTOMOLOGY by Suellen Floyd Pometto August 2019 Accepted by: Dr. Peter H. Adler, Major Advisor and Committee Co-Chair Dr. Eric Benson, Committee Co-Chair Dr. Richard Blob Dr. Patrick Gerard i ABSTRACT The proboscis of butterflies and moths is a key innovation contributing to the high diversity of the order Lepidoptera. In addition to taking nectar from angiosperm sources, many species take up fluids from overripe or sound fruit, plant sap, animal dung, and moist soil. The proboscis is assembled after eclosion of the adult from the pupa by linking together two elongate galeae to form one tube with a single food canal. How do lepidopterans maintain the integrity and function of the proboscis while foraging from various substrates? The research questions included whether lepidopteran species are capable of total self- repair, how widespread the capability of self-repair is within the order, and whether the repaired proboscis is functional. -

Entomology of the Aucklands and Other Islands South of New Zealand: Lepidoptera, Ex Cluding Non-Crambine Pyralidae
Pacific Insects Monograph 27: 55-172 10 November 1971 ENTOMOLOGY OF THE AUCKLANDS AND OTHER ISLANDS SOUTH OF NEW ZEALAND: LEPIDOPTERA, EX CLUDING NON-CRAMBINE PYRALIDAE By J. S. Dugdale1 CONTENTS Introduction 55 Acknowledgements 58 Faunal Composition and Relationships 58 Faunal List 59 Key to Families 68 1. Arctiidae 71 2. Carposinidae 73 Coleophoridae 76 Cosmopterygidae 77 3. Crambinae (pt Pyralidae) 77 4. Elachistidae 79 5. Geometridae 89 Hyponomeutidae 115 6. Nepticulidae 115 7. Noctuidae 117 8. Oecophoridae 131 9. Psychidae 137 10. Pterophoridae 145 11. Tineidae... 148 12. Tortricidae 156 References 169 Note 172 Abstract: This paper deals with all Lepidoptera, excluding the non-crambine Pyralidae, of Auckland, Campbell, Antipodes and Snares Is. The native resident fauna of these islands consists of 42 species of which 21 (50%) are endemic, in 27 genera, of which 3 (11%) are endemic, in 12 families. The endemic fauna is characterised by brachyptery (66%), body size under 10 mm (72%) and concealed, or strictly ground- dwelling larval life. All species can be related to mainland forms; there is a distinctive pre-Pleistocene element as well as some instances of possible Pleistocene introductions, as suggested by the presence of pairs of species, one member of which is endemic but fully winged. A graph and tables are given showing the composition of the fauna, its distribution, habits, and presumed derivations. Host plants or host niches are discussed. An additional 7 species are considered to be non-resident waifs. The taxonomic part includes keys to families (applicable only to the subantarctic fauna), and to genera and species. -

NON-REGULATED PESTS (Non-Actionable)
Import Health Standard Commodity Sub-class: Fresh Fruit/Vegetables Grape, Vitis vinifera from Australia ISSUED Issued pursuant to Section 22 of the Biosecurity Act 1993 Date Issued: 20 December 2000 1 NEW ZEALAND NATIONAL PLANT PROTECTION ORGANISATION The official contact point in New Zealand for overseas NPPOs is the Ministry for Primary Industries (MPI). All communication pertaining to this import health standard should be addressed to: Manager, Import and Export Plants Ministry for Primary Industries PO Box 2526 Wellington NEW ZEALAND Fax: 64-4-894 0662 E-mail: [email protected] http://www.mpi.govt.nz 2 GENERAL CONDITIONS FOR ALL PLANT PRODUCTS All plants and plant products are PROHIBITED entry into New Zealand, unless an import health standard has been issued in accordance with Section 22 of the Biosecurity Act 1993. Should prohibited plants or plant products be intercepted by MPI, the importer will be offered the option of reshipment or destruction of the consignment. The national plant protection organisation of the exporting country is requested to inform MPI of any change in its address. The national plant protection organisation of the exporting country is required to inform MPI of any newly recorded organisms which may infest/infect any commodity approved for export to New Zealand. Pursuant to the Hazardous Substances and New Organisms Act 1996, proposals for the deliberate introduction of new organisms (including genetically modified organisms) as defined by the Act should be referred to: IHS Fresh Fruit/Vegetables. Grape, Vitis vinifera from Australia. (Biosecurity Act 1993) ISSUED: 20 December 2000 Page 1 of 16 Environmental Protection Authority Private Bag 63002 Wellington 6140 NEW ZEALAND Or [email protected],nz Note: In order to meet the Environmental Protection Authority requirements the scientific name (i.e. -

Castlemaine Naturalist April 2012 Vol
Castlemaine Naturalist April 2012 Vol. 37.3 #397 Monthly newsletter of the Castlemaine Field Naturalists Club Inc. Plume moth Stangeia xerodes Maldon Photo – Noel Young Carpets in the Mt Alexander Shire. By Chris Timewell Using information from the third volume of the Moths of Victoria, moth species from the subfamily Sterrhinae (commonly known as Waves) that potentially occur in the Mount Alexander shire were addressed in a Castlemaine Naturalist article in late 2011. This present article addresses the moths from the subfamily Larentiinae potentially occurring in the Mount Alexander Shire, also based on information from the third volume of the Moths of Victoria. The Larentiinae are also known as Carpets, due to the patterns on their wings. They are small to medium sized moths. They often have transverse markings on their wings, and are usually inconspicuous when they hold their wings flat against the surface on which they are resting. There are approximately 140 species known to occur within Victoria. Using the distribution maps and other accompanying information provided on the CD that comes with volume 3 of the Moths of Victoria, the two tables below list the Carpet moths that are either known to occur or potentially occur in the Mt Alexander Shire. Victorian species that are unlikely to occur here are not listed. In summary, from the ~140 Carpet moth species from 24 different genus that are known to occur in Victoria, at least 17 species from seven genus have been confirmed as occurring in the Mt Alexander Shire (Table 1). Another 45 species from 15 of the 24 genus are predicted to potentially occur in the shire (Table 2), with the remaining ~80 species unlikely to occur here ever. -

Insecta: Lepidoptera) SHILAP Revista De Lepidopterología, Vol
SHILAP Revista de Lepidopterología ISSN: 0300-5267 [email protected] Sociedad Hispano-Luso-Americana de Lepidopterología España Vives Moreno, A.; Gastón, J. Contribución al conocimiento de los Microlepidoptera de España, con la descripción de una especie nueva (Insecta: Lepidoptera) SHILAP Revista de Lepidopterología, vol. 45, núm. 178, junio, 2017, pp. 317-342 Sociedad Hispano-Luso-Americana de Lepidopterología Madrid, España Disponible en: http://www.redalyc.org/articulo.oa?id=45551614016 Cómo citar el artículo Número completo Sistema de Información Científica Más información del artículo Red de Revistas Científicas de América Latina, el Caribe, España y Portugal Página de la revista en redalyc.org Proyecto académico sin fines de lucro, desarrollado bajo la iniciativa de acceso abierto SHILAP Revta. lepid., 45 (178) junio 2017: 317-342 eISSN: 2340-4078 ISSN: 0300-5267 Contribución al conocimiento de los Microlepidoptera de España, con la descripción de una especie nueva (Insecta: Lepidoptera) A. Vives Moreno & J. Gastón Resumen Se describe una especie nueva Oinophila blayi Vives & Gastón, sp. n. Se registran dos géneros Niphonympha Meyrick, 1914, Sardzea Amsel, 1961 y catorce especies nuevas para España: Niphonympha dealbatella Zeller, 1847, Tinagma balteolella (Fischer von Rösslerstamm, [1841] 1834), Alloclita francoeuriae Walsingham, 1905 (Islas Ca- narias), Epicallima bruandella (Ragonot, 1889), Agonopterix astrantiae (Heinemann, 1870), Agonopterix kuznetzovi Lvovsky, 1983, Depressaria halophilella Chrétien, 1908, Depressaria cinderella Corley, 2002, Metzneria santoline- lla (Amsel, 1936), Phtheochroa sinecarina Huemer, 1990 (Islas Canarias), Sardzea diviselloides Amsel, 1961, Pem- pelia coremetella (Amsel, 1949), Epischnia albella Amsel, 1954 (Islas Canarias) y Metasia cyrnealis Schawerda, 1926. Se citan como nuevas para las Islas Canarias Eucosma cana (Haworth, 1811) y Cydia blackmoreana (Wal- singham, 1903). -

197 Section 9 Sunflower (Helianthus
SECTION 9 SUNFLOWER (HELIANTHUS ANNUUS L.) 1. Taxonomy of the Genus Helianthus, Natural Habitat and Origins of the Cultivated Sunflower A. Taxonomy of the genus Helianthus The sunflower belongs to the genus Helianthus in the Composite family (Asterales order), which includes species with very diverse morphologies (herbs, shrubs, lianas, etc.). The genus Helianthus belongs to the Heliantheae tribe. This includes approximately 50 species originating in North and Central America. The basis for the botanical classification of the genus Helianthus was proposed by Heiser et al. (1969) and refined subsequently using new phenological, cladistic and biosystematic methods, (Robinson, 1979; Anashchenko, 1974, 1979; Schilling and Heiser, 1981) or molecular markers (Sossey-Alaoui et al., 1998). This approach splits Helianthus into four sections: Helianthus, Agrestes, Ciliares and Atrorubens. This classification is set out in Table 1.18. Section Helianthus This section comprises 12 species, including H. annuus, the cultivated sunflower. These species, which are diploid (2n = 34), are interfertile and annual in almost all cases. For the majority, the natural distribution is central and western North America. They are generally well adapted to dry or even arid areas and sandy soils. The widespread H. annuus L. species includes (Heiser et al., 1969) plants cultivated for seed or fodder referred to as H. annuus var. macrocarpus (D.C), or cultivated for ornament (H. annuus subsp. annuus), and uncultivated wild and weedy plants (H. annuus subsp. lenticularis, H. annuus subsp. Texanus, etc.). Leaves of these species are usually alternate, ovoid and with a long petiole. Flower heads, or capitula, consist of tubular and ligulate florets, which may be deep purple, red or yellow. -

The Mcguire Center for Lepidoptera and Biodiversity
Supplemental Information All specimens used within this study are housed in: the McGuire Center for Lepidoptera and Biodiversity (MGCL) at the Florida Museum of Natural History, Gainesville, USA (FLMNH); the University of Maryland, College Park, USA (UMD); the Muséum national d’Histoire naturelle in Paris, France (MNHN); and the Australian National Insect Collection in Canberra, Australia (ANIC). Methods DNA extraction protocol of dried museum specimens (detailed instructions) Prior to tissue sampling, dried (pinned or papered) specimens were assigned MGCL barcodes, photographed, and their labels digitized. Abdomens were then removed using sterile forceps, cleaned with 100% ethanol between each sample, and the remaining specimens were returned to their respective trays within the MGCL collections. Abdomens were placed in 1.5 mL microcentrifuge tubes with the apex of the abdomen in the conical end of the tube. For larger abdomens, 5 mL microcentrifuge tubes or larger were utilized. A solution of proteinase K (Qiagen Cat #19133) and genomic lysis buffer (OmniPrep Genomic DNA Extraction Kit) in a 1:50 ratio was added to each abdomen containing tube, sufficient to cover the abdomen (typically either 300 µL or 500 µL) - similar to the concept used in Hundsdoerfer & Kitching (1). Ratios of 1:10 and 1:25 were utilized for low quality or rare specimens. Low quality specimens were defined as having little visible tissue inside of the abdomen, mold/fungi growth, or smell of bacterial decay. Samples were incubated overnight (12-18 hours) in a dry air oven at 56°C. Importantly, we also adjusted the ratio depending on the tissue type, i.e., increasing the ratio for particularly large or egg-containing abdomens. -
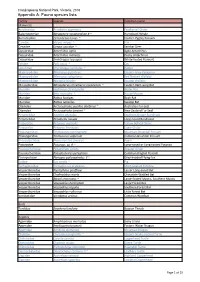
Report-VIC-Croajingolong National Park-Appendix A
Croajingolong National Park, Victoria, 2016 Appendix A: Fauna species lists Family Species Common name Mammals Acrobatidae Acrobates pygmaeus Feathertail Glider Balaenopteriae Megaptera novaeangliae # ~ Humpback Whale Burramyidae Cercartetus nanus ~ Eastern Pygmy Possum Canidae Vulpes vulpes ^ Fox Cervidae Cervus unicolor ^ Sambar Deer Dasyuridae Antechinus agilis Agile Antechinus Dasyuridae Antechinus mimetes Dusky Antechinus Dasyuridae Sminthopsis leucopus White-footed Dunnart Felidae Felis catus ^ Cat Leporidae Oryctolagus cuniculus ^ Rabbit Macropodidae Macropus giganteus Eastern Grey Kangaroo Macropodidae Macropus rufogriseus Red Necked Wallaby Macropodidae Wallabia bicolor Swamp Wallaby Miniopteridae Miniopterus schreibersii oceanensis ~ Eastern Bent-wing Bat Muridae Hydromys chrysogaster Water Rat Muridae Mus musculus ^ House Mouse Muridae Rattus fuscipes Bush Rat Muridae Rattus lutreolus Swamp Rat Otariidae Arctocephalus pusillus doriferus ~ Australian Fur-seal Otariidae Arctocephalus forsteri ~ New Zealand Fur Seal Peramelidae Isoodon obesulus Southern Brown Bandicoot Peramelidae Perameles nasuta Long-nosed Bandicoot Petauridae Petaurus australis Yellow Bellied Glider Petauridae Petaurus breviceps Sugar Glider Phalangeridae Trichosurus cunninghami Mountain Brushtail Possum Phalangeridae Trichosurus vulpecula Common Brushtail Possum Phascolarctidae Phascolarctos cinereus Koala Potoroidae Potorous sp. # ~ Long-nosed or Long-footed Potoroo Pseudocheiridae Petauroides volans Greater Glider Pseudocheiridae Pseudocheirus peregrinus -

Phylogeny and Evolution of Lepidoptera
EN62CH15-Mitter ARI 5 November 2016 12:1 I Review in Advance first posted online V E W E on November 16, 2016. (Changes may R S still occur before final publication online and in print.) I E N C N A D V A Phylogeny and Evolution of Lepidoptera Charles Mitter,1,∗ Donald R. Davis,2 and Michael P. Cummings3 1Department of Entomology, University of Maryland, College Park, Maryland 20742; email: [email protected] 2Department of Entomology, National Museum of Natural History, Smithsonian Institution, Washington, DC 20560 3Laboratory of Molecular Evolution, Center for Bioinformatics and Computational Biology, University of Maryland, College Park, Maryland 20742 Annu. Rev. Entomol. 2017. 62:265–83 Keywords Annu. Rev. Entomol. 2017.62. Downloaded from www.annualreviews.org The Annual Review of Entomology is online at Hexapoda, insect, systematics, classification, butterfly, moth, molecular ento.annualreviews.org systematics This article’s doi: Access provided by University of Maryland - College Park on 11/20/16. For personal use only. 10.1146/annurev-ento-031616-035125 Abstract Copyright c 2017 by Annual Reviews. Until recently, deep-level phylogeny in Lepidoptera, the largest single ra- All rights reserved diation of plant-feeding insects, was very poorly understood. Over the past ∗ Corresponding author two decades, building on a preceding era of morphological cladistic stud- ies, molecular data have yielded robust initial estimates of relationships both within and among the ∼43 superfamilies, with unsolved problems now yield- ing to much larger data sets from high-throughput sequencing. Here we summarize progress on lepidopteran phylogeny since 1975, emphasizing the superfamily level, and discuss some resulting advances in our understanding of lepidopteran evolution. -

ARTHROPODA Subphylum Hexapoda Protura, Springtails, Diplura, and Insects
NINE Phylum ARTHROPODA SUBPHYLUM HEXAPODA Protura, springtails, Diplura, and insects ROD P. MACFARLANE, PETER A. MADDISON, IAN G. ANDREW, JOCELYN A. BERRY, PETER M. JOHNS, ROBERT J. B. HOARE, MARIE-CLAUDE LARIVIÈRE, PENELOPE GREENSLADE, ROSA C. HENDERSON, COURTenaY N. SMITHERS, RicarDO L. PALMA, JOHN B. WARD, ROBERT L. C. PILGRIM, DaVID R. TOWNS, IAN McLELLAN, DAVID A. J. TEULON, TERRY R. HITCHINGS, VICTOR F. EASTOP, NICHOLAS A. MARTIN, MURRAY J. FLETCHER, MARLON A. W. STUFKENS, PAMELA J. DALE, Daniel BURCKHARDT, THOMAS R. BUCKLEY, STEVEN A. TREWICK defining feature of the Hexapoda, as the name suggests, is six legs. Also, the body comprises a head, thorax, and abdomen. The number A of abdominal segments varies, however; there are only six in the Collembola (springtails), 9–12 in the Protura, and 10 in the Diplura, whereas in all other hexapods there are strictly 11. Insects are now regarded as comprising only those hexapods with 11 abdominal segments. Whereas crustaceans are the dominant group of arthropods in the sea, hexapods prevail on land, in numbers and biomass. Altogether, the Hexapoda constitutes the most diverse group of animals – the estimated number of described species worldwide is just over 900,000, with the beetles (order Coleoptera) comprising more than a third of these. Today, the Hexapoda is considered to contain four classes – the Insecta, and the Protura, Collembola, and Diplura. The latter three classes were formerly allied with the insect orders Archaeognatha (jumping bristletails) and Thysanura (silverfish) as the insect subclass Apterygota (‘wingless’). The Apterygota is now regarded as an artificial assemblage (Bitsch & Bitsch 2000). -

The Lepidoptera Rapa Island
J. F. GATES CLA, The Lepidoptera Rapa Island SMITHSONIAN CONTRIBUTIONS TO ZOOLOGY • 1971 NUMBER 56 .-24 f O si % r 17401 •% -390O i 112100) 0 is -•^ i BLAKE*w 1PLATEALP I5 i I >k =(M&2l2Jo SMITHSONIAN CONTRIBUTIONS TO ZOOLOGY NUMBER 56 j. F. Gates Clarke The Lepidoptera of Rapa Island SMITHSONIAN INSTITUTION PRESS CITY OF WASHINGTON 1971 SERIAL PUBLICATIONS OF THE SMITHSONIAN INSTITUTION The emphasis upon publications as a means of diffusing knowledge was expressed by the first Secretary of the Smithsonian Institution. In his formal plan for the Insti- tution, Joseph Henry articulated a program that included the following statement: "It is proposed to publish a series of reports, giving an account of the new discoveries in science, and of the changes made from year to year in all branches of knowledge not strictly professional." This keynote of basic research has been adhered to over the years in the issuance of thousands of titles in serial publications under the Smithsonian imprint, commencing with Smithsonian Contributions to Knowledge in 1848 and continuing with the following active series: Smithsonian Annals of Flight Smithsonian Contributions to Anthropology Smithsonian Contributions to Astrophysics Smithsonian Contributions to Botany Smithsonian Contributions to the Earth Sciences Smithsonian Contributions to Paleobiology Smithsonian Contributions to Zoology Smithsonian Studies in History and Technology In these series, the Institution publishes original articles and monographs dealing with the research and collections of its several museums and offices and of professional colleagues at other institutions of learning. These papers report newly acquired facts, synoptic interpretations of data, or original theory in specialized fields. -

Bosco Palazzi
SHILAP Revista de Lepidopterología ISSN: 0300-5267 ISSN: 2340-4078 [email protected] Sociedad Hispano-Luso-Americana de Lepidopterología España Bella, S; Parenzan, P.; Russo, P. Diversity of the Macrolepidoptera from a “Bosco Palazzi” area in a woodland of Quercus trojana Webb., in southeastern Murgia (Apulia region, Italy) (Insecta: Lepidoptera) SHILAP Revista de Lepidopterología, vol. 46, no. 182, 2018, April-June, pp. 315-345 Sociedad Hispano-Luso-Americana de Lepidopterología España Available in: https://www.redalyc.org/articulo.oa?id=45559600012 How to cite Complete issue Scientific Information System Redalyc More information about this article Network of Scientific Journals from Latin America and the Caribbean, Spain and Journal's webpage in redalyc.org Portugal Project academic non-profit, developed under the open access initiative SHILAP Revta. lepid., 46 (182) junio 2018: 315-345 eISSN: 2340-4078 ISSN: 0300-5267 Diversity of the Macrolepidoptera from a “Bosco Palazzi” area in a woodland of Quercus trojana Webb., in southeastern Murgia (Apulia region, Italy) (Insecta: Lepidoptera) S. Bella, P. Parenzan & P. Russo Abstract This study summarises the known records of the Macrolepidoptera species of the “Bosco Palazzi” area near the municipality of Putignano (Apulia region) in the Murgia mountains in southern Italy. The list of species is based on historical bibliographic data along with new material collected by other entomologists in the last few decades. A total of 207 species belonging to the families Cossidae (3 species), Drepanidae (4 species), Lasiocampidae (7 species), Limacodidae (1 species), Saturniidae (2 species), Sphingidae (5 species), Brahmaeidae (1 species), Geometridae (55 species), Notodontidae (5 species), Nolidae (3 species), Euteliidae (1 species), Noctuidae (96 species), and Erebidae (24 species) were identified.