Melanoregulin Regulates a Shedding Mechanism That Drives
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Defining Functional Interactions During Biogenesis of Epithelial Junctions
ARTICLE Received 11 Dec 2015 | Accepted 13 Oct 2016 | Published 6 Dec 2016 | Updated 5 Jan 2017 DOI: 10.1038/ncomms13542 OPEN Defining functional interactions during biogenesis of epithelial junctions J.C. Erasmus1,*, S. Bruche1,*,w, L. Pizarro1,2,*, N. Maimari1,3,*, T. Poggioli1,w, C. Tomlinson4,J.Lees5, I. Zalivina1,w, A. Wheeler1,w, A. Alberts6, A. Russo2 & V.M.M. Braga1 In spite of extensive recent progress, a comprehensive understanding of how actin cytoskeleton remodelling supports stable junctions remains to be established. Here we design a platform that integrates actin functions with optimized phenotypic clustering and identify new cytoskeletal proteins, their functional hierarchy and pathways that modulate E-cadherin adhesion. Depletion of EEF1A, an actin bundling protein, increases E-cadherin levels at junctions without a corresponding reinforcement of cell–cell contacts. This unexpected result reflects a more dynamic and mobile junctional actin in EEF1A-depleted cells. A partner for EEF1A in cadherin contact maintenance is the formin DIAPH2, which interacts with EEF1A. In contrast, depletion of either the endocytic regulator TRIP10 or the Rho GTPase activator VAV2 reduces E-cadherin levels at junctions. TRIP10 binds to and requires VAV2 function for its junctional localization. Overall, we present new conceptual insights on junction stabilization, which integrate known and novel pathways with impact for epithelial morphogenesis, homeostasis and diseases. 1 National Heart and Lung Institute, Faculty of Medicine, Imperial College London, London SW7 2AZ, UK. 2 Computing Department, Imperial College London, London SW7 2AZ, UK. 3 Bioengineering Department, Faculty of Engineering, Imperial College London, London SW7 2AZ, UK. 4 Department of Surgery & Cancer, Faculty of Medicine, Imperial College London, London SW7 2AZ, UK. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -
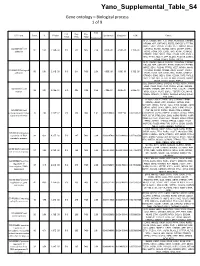
Supplemental Tables4.Pdf
Yano_Supplemental_Table_S4 Gene ontology – Biological process 1 of 9 Fold List Pop Pop GO Term Count % PValue Bonferroni Benjamini FDR Genes Total Hits Total Enrichment DLC1, CADM1, NELL2, CLSTN1, PCDHGA8, CTNNB1, NRCAM, APP, CNTNAP2, FERT2, RAPGEF1, PTPRM, MPDZ, SDK1, PCDH9, PTPRS, VEZT, NRXN1, MYH9, GO:0007155~cell CTNNA2, NCAM1, NCAM2, DDR1, LSAMP, CNTN1, 50 5.61 2.14E-08 510 311 7436 2.34 4.50E-05 4.50E-05 3.70E-05 adhesion ROR2, VCAN, DST, LIMS1, TNC, ASTN1, CTNND2, CTNND1, CDH2, NEO1, CDH4, CD24A, FAT3, PVRL3, TRO, TTYH1, MLLT4, LPP, NLGN1, PCDH19, LAMA1, ITGA9, CDH13, CDON, PSPC1 DLC1, CADM1, NELL2, CLSTN1, PCDHGA8, CTNNB1, NRCAM, APP, CNTNAP2, FERT2, RAPGEF1, PTPRM, MPDZ, SDK1, PCDH9, PTPRS, VEZT, NRXN1, MYH9, GO:0022610~biological CTNNA2, NCAM1, NCAM2, DDR1, LSAMP, CNTN1, 50 5.61 2.14E-08 510 311 7436 2.34 4.50E-05 4.50E-05 3.70E-05 adhesion ROR2, VCAN, DST, LIMS1, TNC, ASTN1, CTNND2, CTNND1, CDH2, NEO1, CDH4, CD24A, FAT3, PVRL3, TRO, TTYH1, MLLT4, LPP, NLGN1, PCDH19, LAMA1, ITGA9, CDH13, CDON, PSPC1 DCC, ENAH, PLXNA2, CAPZA2, ATP5B, ASTN1, PAX6, ZEB2, CDH2, CDH4, GLI3, CD24A, EPHB1, NRCAM, GO:0006928~cell CTTNBP2, EDNRB, APP, PTK2, ETV1, CLASP2, STRBP, 36 4.04 3.46E-07 510 205 7436 2.56 7.28E-04 3.64E-04 5.98E-04 motion NRG1, DCLK1, PLAT, SGPL1, TGFBR1, EVL, MYH9, YWHAE, NCKAP1, CTNNA2, SEMA6A, EPHA4, NDEL1, FYN, LRP6 PLXNA2, ADCY5, PAX6, GLI3, CTNNB1, LPHN2, EDNRB, LPHN3, APP, CSNK2A1, GPR45, NRG1, RAPGEF1, WWOX, SGPL1, TLE4, SPEN, NCAM1, DDR1, GRB10, GRM3, GNAQ, HIPK1, GNB1, HIPK2, PYGO1, GO:0007166~cell RNF138, ROR2, CNTN1, -

Serum Albumin OS=Homo Sapiens
Protein Name Cluster of Glial fibrillary acidic protein OS=Homo sapiens GN=GFAP PE=1 SV=1 (P14136) Serum albumin OS=Homo sapiens GN=ALB PE=1 SV=2 Cluster of Isoform 3 of Plectin OS=Homo sapiens GN=PLEC (Q15149-3) Cluster of Hemoglobin subunit beta OS=Homo sapiens GN=HBB PE=1 SV=2 (P68871) Vimentin OS=Homo sapiens GN=VIM PE=1 SV=4 Cluster of Tubulin beta-3 chain OS=Homo sapiens GN=TUBB3 PE=1 SV=2 (Q13509) Cluster of Actin, cytoplasmic 1 OS=Homo sapiens GN=ACTB PE=1 SV=1 (P60709) Cluster of Tubulin alpha-1B chain OS=Homo sapiens GN=TUBA1B PE=1 SV=1 (P68363) Cluster of Isoform 2 of Spectrin alpha chain, non-erythrocytic 1 OS=Homo sapiens GN=SPTAN1 (Q13813-2) Hemoglobin subunit alpha OS=Homo sapiens GN=HBA1 PE=1 SV=2 Cluster of Spectrin beta chain, non-erythrocytic 1 OS=Homo sapiens GN=SPTBN1 PE=1 SV=2 (Q01082) Cluster of Pyruvate kinase isozymes M1/M2 OS=Homo sapiens GN=PKM PE=1 SV=4 (P14618) Glyceraldehyde-3-phosphate dehydrogenase OS=Homo sapiens GN=GAPDH PE=1 SV=3 Clathrin heavy chain 1 OS=Homo sapiens GN=CLTC PE=1 SV=5 Filamin-A OS=Homo sapiens GN=FLNA PE=1 SV=4 Cytoplasmic dynein 1 heavy chain 1 OS=Homo sapiens GN=DYNC1H1 PE=1 SV=5 Cluster of ATPase, Na+/K+ transporting, alpha 2 (+) polypeptide OS=Homo sapiens GN=ATP1A2 PE=3 SV=1 (B1AKY9) Fibrinogen beta chain OS=Homo sapiens GN=FGB PE=1 SV=2 Fibrinogen alpha chain OS=Homo sapiens GN=FGA PE=1 SV=2 Dihydropyrimidinase-related protein 2 OS=Homo sapiens GN=DPYSL2 PE=1 SV=1 Cluster of Alpha-actinin-1 OS=Homo sapiens GN=ACTN1 PE=1 SV=2 (P12814) 60 kDa heat shock protein, mitochondrial OS=Homo -

MYO5A Rabbit Pab
Leader in Biomolecular Solutions for Life Science MYO5A Rabbit pAb Catalog No.: A9830 Basic Information Background Catalog No. This gene is one of three myosin V heavy-chain genes, belonging to the myosin gene A9830 superfamily. Myosin V is a class of actin-based motor proteins involved in cytoplasmic vesicle transport and anchorage, spindle-pole alignment and mRNA translocation. The Observed MW protein encoded by this gene is abundant in melanocytes and nerve cells. Mutations in 240kDa this gene cause Griscelli syndrome type-1 (GS1), Griscelli syndrome type-3 (GS3) and neuroectodermal melanolysosomal disease, or Elejalde disease. Multiple alternatively Calculated MW spliced transcript variants encoding different isoforms have been reported, but the full- 212kDa/215kDa/218kDa length nature of some variants has not been determined. Category Primary antibody Applications WB,IHC,IF Cross-Reactivity Human, Mouse, Rat Recommended Dilutions Immunogen Information WB 1:500 - 1:2000 Gene ID Swiss Prot 4644 Q9Y4I1 IHC 1:50 - 1:100 Immunogen 1:50 - 1:100 IF Recombinant fusion protein containing a sequence corresponding to amino acids 950-1150 of human MYO5A (NP_000250.3). Synonyms MYO5A;GS1;MYH12;MYO5;MYR12;myosin VA Contact Product Information www.abclonal.com Source Isotype Purification Rabbit IgG Affinity purification Storage Store at -20℃. Avoid freeze / thaw cycles. Buffer: PBS with 0.02% sodium azide,50% glycerol,pH7.3. Validation Data Western blot analysis of extracts of various cell lines, using MYO5A antibody (A9830) at 1:1000 dilution. Secondary antibody: HRP Goat Anti-Rabbit IgG (H+L) (AS014) at 1:10000 dilution. Lysates/proteins: 25ug per lane. Blocking buffer: 3% nonfat dry milk in TBST. -

Bone Morphogenetic Protein Signaling Suppresses Wound-Induced Skin Repair by Inhibiting Keratinocyte Proliferation and Migration Christopher J
ORIGINAL ARTICLE Bone Morphogenetic Protein Signaling Suppresses Wound-Induced Skin Repair by Inhibiting Keratinocyte Proliferation and Migration Christopher J. Lewis1,2,AndreiN.Mardaryev1, Krzysztof Poterlowicz1, Tatyana Y. Sharova3, Ahmar Aziz3, David T. Sharpe2, Natalia V. Botchkareva1 and Andrey A. Sharov3 Bone morphogenetic protein (BMP) signaling plays a key role in the control of skin development and postnatal remodeling by regulating keratinocyte proliferation, differentiation, and apoptosis. To study the role of BMPs in wound-induced epidermal repair, we used transgenic mice overexpressing the BMP downstream component Smad1 under the control of a K14 promoter as an in vivo model, as well as ex vivo and in vitro assays. K14- caSmad1 mice (transgenic mice overexpressing a constitutively active form of Smad1 under K14 promoter) exhibited retarded wound healing associated with significant inhibition of proliferation and increased apoptosis in healing wound epithelium. Furthermore, microarray and quantitative real-time reverse-transcriptase–PCR (qRT– PCR) analyses revealed decreased expression of a number of cytoskeletal/cell motility–associated genes including wound-associated keratins (Krt16, Krt17)andMyosin VA (Myo5a), in the epidermis of K14-caSmad1 mice versus wild-type (WT) controls during wound healing. BMP treatment significantly inhibited keratinocyte migration ex vivo, and primary keratinocytes of K14-caSmad1 mice showed retarded migration compared with WT controls. Finally, small interfering RNA (siRNA)–mediated silencing of BMPR-1B in primary mouse keratinocytes accelerated cell migration and was associated with increased expression of Krt16, Krt17, and Myo5a compared with controls. Thus, this study demonstrates that BMPs inhibit keratinocyte proliferation, cytoskeletal organization, and migration in regenerating skin epithelium during wound healing, and raises a possibility for using BMP antagonists for the management of chronic wounds. -

Myth4-FERM Myosin Based Filopodia Initiation
MyTH4-FERM Myosin based filopodia initiation A DISSERTATION SUBMITTED TO THE FACULTY OF THE GRADUATE SCHOOL OF THE UNIVERSITY OF MINNESOTA BY Ashley L. Arthur IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY Margaret A. Titus, PhD ADVISOR July 2020 © Ashley L Arthur 2020 ACKNOWLEDGEMENTS I would first and foremost like to advisor my mentor, Dr. Margaret Titus, for her unassailable commitment to training, enthusiasm for science and her sup- port of my career. Meg has been superb advisor, has made me a better scientist and communicator and I genuinely enjoyed working for her. I am grateful for the long list of positive experiences and opportunities I gained while working in the Titus lab. I would like to thank all of my past and present lab mates. Thank you to Hilary Bauer, Sinzi Cornea and Zoe Henrot for welcoming me into the lab when I started and especially to Karl Petersen who share his imaging and analysis ex- pertise. I am so grateful for the help and from my PLA project teammate Livia Songster, you brought such great energy to the project and to the lab. Thanks Casey Eddington, Annika Schroeder for their support, encouragement and help reading and discussing many aspect of this work. Thanks to the University of MN undergraduate students who joined my on research projects over the years espe- cially to Himanshu Jain. I would like to thank Jordan Beach at Loyola, Guillermo Marques, Mark Sanders, for their help with imaging. I thank Ashim Rai for his as- sistance with motor purification and motility assays. -

Strong Correlation Between Air-Liquid Interface Cultures and in Vivo Transcriptomics of Nasal Brush Biopsy
Am J Physiol Lung Cell Mol Physiol 318: L1056–L1062, 2020. First published April 1, 2020; doi:10.1152/ajplung.00050.2020. RAPID REPORT Translational Physiology Strong correlation between air-liquid interface cultures and in vivo transcriptomics of nasal brush biopsy X Baishakhi Ghosh,1 Bongsoo Park,1 Debarshi Bhowmik,2 Kristine Nishida,3 Molly Lauver,3 Nirupama Putcha,3 Peisong Gao,4 Murugappan Ramanathan, Jr.,5 Nadia Hansel,3 Shyam Biswal,1 and Venkataramana K. Sidhaye1,3 1Department of Environmental Health and Engineering, Johns Hopkins Bloomberg School of Public Health, Baltimore, Maryland; 2Department of Biology, Johns Hopkins University, Baltimore, Maryland; 3Department of Pulmonary and Critical Care Medicine, Johns Hopkins School of Medicine, Baltimore, Maryland; 4Division of Allergy and Clinical Immunology, Johns Hopkins School of Medicine, Baltimore, Maryland; and 5Department of Otolaryngology-Head and Neck Surgery, Johns Hopkins School of Medicine, Baltimore, Maryland Submitted 18 February 2020; accepted in final form 24 March 2020 Ghosh B, Park B, Bhowmik D, Nishida K, Lauver M, Putcha N, RNA sequencing to determine the correlation between ALI- Gao P, Ramanathan M Jr, Hansel N, Biswal S, Sidhaye VK. cultured epithelial cells and epithelial cells obtained from nasal Strong correlation between air-liquid interface cultures and in vivo brushing and identify differences that may arise as a result of transcriptomics of nasal brush biopsy. Am J Physiol Lung Cell Mol redifferentiation. Physiol 318: L1056–L1062, 2020. First published April 1, 2020; doi:10.1152/ajplung.00050.2020.—Air-liquid interface (ALI) cultures METHODS are ex vivo models that are used extensively to study the epithelium of patients with chronic respiratory diseases. -

Molecular Signatures Differentiate Immune States in Type 1 Diabetes Families
Page 1 of 65 Diabetes Molecular signatures differentiate immune states in Type 1 diabetes families Yi-Guang Chen1, Susanne M. Cabrera1, Shuang Jia1, Mary L. Kaldunski1, Joanna Kramer1, Sami Cheong2, Rhonda Geoffrey1, Mark F. Roethle1, Jeffrey E. Woodliff3, Carla J. Greenbaum4, Xujing Wang5, and Martin J. Hessner1 1The Max McGee National Research Center for Juvenile Diabetes, Children's Research Institute of Children's Hospital of Wisconsin, and Department of Pediatrics at the Medical College of Wisconsin Milwaukee, WI 53226, USA. 2The Department of Mathematical Sciences, University of Wisconsin-Milwaukee, Milwaukee, WI 53211, USA. 3Flow Cytometry & Cell Separation Facility, Bindley Bioscience Center, Purdue University, West Lafayette, IN 47907, USA. 4Diabetes Research Program, Benaroya Research Institute, Seattle, WA, 98101, USA. 5Systems Biology Center, the National Heart, Lung, and Blood Institute, the National Institutes of Health, Bethesda, MD 20824, USA. Corresponding author: Martin J. Hessner, Ph.D., The Department of Pediatrics, The Medical College of Wisconsin, Milwaukee, WI 53226, USA Tel: 011-1-414-955-4496; Fax: 011-1-414-955-6663; E-mail: [email protected]. Running title: Innate Inflammation in T1D Families Word count: 3999 Number of Tables: 1 Number of Figures: 7 1 For Peer Review Only Diabetes Publish Ahead of Print, published online April 23, 2014 Diabetes Page 2 of 65 ABSTRACT Mechanisms associated with Type 1 diabetes (T1D) development remain incompletely defined. Employing a sensitive array-based bioassay where patient plasma is used to induce transcriptional responses in healthy leukocytes, we previously reported disease-specific, partially IL-1 dependent, signatures associated with pre and recent onset (RO) T1D relative to unrelated healthy controls (uHC). -

View/Download
Stoica G, Martini-Stoica H. Commentary: Pleiotropic Neuropathological and Biochemical Alterations Neuromedicine associated with Myo5a Mutation in a Rat Model. J Neurol Neuromed (2018) 3(6): 13-16 www.jneurology.com www.jneurology.com Journal of Neurology && NeuromedicineNeuromedicine Commentary Open Access Commentary: Pleiotropic Neuropathological and Biochemical Alterations associated with Myo5a Mutation in a Rat Model George Stoica1*, Heidi Martini-Stoica2 1Texas A&M University, College Station, TX,USA 2Baylor College of Medicine, Houston, TX, USA Article Info Molecular motors have a broad range of functions in the nervous system, including brain wiring, neuronal survival, neuronal Article Notes plasticity, higher brain function, and control of central nervous Received: November 9, 2018 1 Accepted: December 24, 2018 system and peripheral nervous system development . In Landrock et al., a rat model with neurodegeneration, movement disorder, and early *Correspondence: Dr. George Stoica, DVM, MSc., Ph.D., Professor Emeritus, death is associated with loss of function of the molecular motor protein Texas A&M University, College Station, Texas, USA; E-mail: Myosin5a (Myo5a)2. Myo5a, highly expressed in the brain and skin and [email protected]. the predominant neuronal type, is an actin-based motor protein, which © 2018 Stoica G. This article is distributed under the terms of is part of a large group of unconventional myosins. Myosin5a plays a the Creative Commons Attribution 4.0 International License major role in cytoskeletal dynamics in the nervous system, potentially mediating movements of organelles and proteins in neurons3, 4. However, little is known about the precise role of Myo5a in neuronal function Myo5a loss of functionand survival.2. -

Mirna Regulons Associated with Synaptic Function
miRNA Regulons Associated with Synaptic Function Maria Paschou1, Maria D. Paraskevopoulou2, Ioannis S. Vlachos2, Pelagia Koukouraki1, Artemis G. Hatzigeorgiou2,3, Epaminondas Doxakis1* 1 Basic Neurosciences Division, Biomedical Research Foundation of the Academy of Athens, Athens, Greece, 2 Institute of Molecular Oncology, Biomedical Sciences Research Center ‘‘Alexander Fleming’’ Vari, Greece, 3 Department of Computer and Communication Engineering, University of Thessaly, Volos, Greece Abstract Differential RNA localization and local protein synthesis regulate synapse function and plasticity in neurons. MicroRNAs are a conserved class of regulatory RNAs that control mRNA stability and translation in tissues. They are abundant in the brain but the extent into which they are involved in synaptic mRNA regulation is poorly known. Herein, a computational analysis of the coding and 39UTR regions of 242 presynaptic and 304 postsynaptic proteins revealed that 91% of them are predicted to be microRNA targets. Analysis of the longest 39UTR isoform of synaptic transcripts showed that presynaptic mRNAs have significantly longer 39UTR than control and postsynaptic mRNAs. In contrast, the shortest 39UTR isoform of postsynaptic mRNAs is significantly shorter than control and presynaptic mRNAs, indicating they avert microRNA regulation under specific conditions. Examination of microRNA binding site density of synaptic 39UTRs revealed that they are twice as dense as the rest of protein-coding transcripts and that approximately 50% of synaptic transcripts are predicted to have more than five different microRNA sites. An interaction map exploring the association of microRNAs and their targets revealed that a small set of ten microRNAs is predicted to regulate 77% and 80% of presynaptic and postsynaptic transcripts, respectively. -

Genetics and Human Traits
Help Me Understand Genetics Genetics and Human Traits Reprinted from MedlinePlus Genetics U.S. National Library of Medicine National Institutes of Health Department of Health & Human Services Table of Contents 1 Are fingerprints determined by genetics? 1 2 Is eye color determined by genetics? 3 3 Is intelligence determined by genetics? 5 4 Is handedness determined by genetics? 7 5 Is the probability of having twins determined by genetics? 9 6 Is hair texture determined by genetics? 11 7 Is hair color determined by genetics? 13 8 Is height determined by genetics? 16 9 Are moles determined by genetics? 18 10 Are facial dimples determined by genetics? 20 11 Is athletic performance determined by genetics? 21 12 Is longevity determined by genetics? 23 13 Is temperament determined by genetics? 26 Reprinted from MedlinePlus Genetics (https://medlineplus.gov/genetics/) i Genetics and Human Traits 1 Are fingerprints determined by genetics? Each person’s fingerprints are unique, which is why they have long been used as a way to identify individuals. Surprisingly little is known about the factors that influence a person’s fingerprint patterns. Like many other complex traits, studies suggest that both genetic and environmental factors play a role. A person’s fingerprints are based on the patterns of skin ridges (called dermatoglyphs) on the pads of the fingers. These ridges are also present on the toes, the palms of the hands, and the soles of the feet. Although the basic whorl, arch, and loop patterns may be similar, the details of the patterns are specific to each individual. Dermatoglyphs develop before birth and remain the same throughout life.