HIPK4 Is Essential for Murine Spermiogenesis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Expression and Localization of Myosin VI in Developing Mouse Spermatids
Histochem Cell Biol DOI 10.1007/s00418-017-1579-z ORIGINAL PAPER Expression and localization of myosin VI in developing mouse spermatids Przemysław Zakrzewski1 · Robert Lenartowski2 · Maria Jolanta Re˛dowicz3 · Kathryn G. Miller4 · Marta Lenartowska1 Accepted: 4 May 2017 © The Author(s) 2017. This article is an open access publication Abstract Myosin VI (MVI) is a versatile actin-based and Sertoli cell actin hoops. Since this is the frst report of motor protein that has been implicated in a variety of dif- MVI expression and localization during mouse spermio- ferent cellular processes, including endo- and exocytic genesis and MVI partners in developing sperm have not yet vesicle traffcking, Golgi morphology, and actin structure been identifed, we discuss some probable roles for MVI stabilization. A role for MVI in crucial actin-based pro- in this process. During early stages, MVI is hypothesized cesses involved in sperm maturation was demonstrated in to play a role in Golgi morphology and function as well Drosophila. Because of the prominence and importance of as in actin dynamics regulation important for attachment actin structures in mammalian spermiogenesis, we inves- of developing acrosome to the nuclear envelope. Next, tigated whether MVI was associated with actin-mediated the protein might also play anchoring roles to help gener- maturation events in mammals. Both immunofuorescence ate forces needed for spermatid head elongation. Moreo- and ultrastructural analyses using immunogold labeling ver, association of MVI with actin that accumulates in showed that MVI was strongly linked with key structures the Sertoli cell ectoplasmic specialization and other actin involved in sperm development and maturation. -

Te2, Part Iii
TERMINOLOGIA EMBRYOLOGICA Second Edition International Embryological Terminology FIPAT The Federative International Programme for Anatomical Terminology A programme of the International Federation of Associations of Anatomists (IFAA) TE2, PART III Contents Caput V: Organogenesis Chapter 5: Organogenesis (continued) Systema respiratorium Respiratory system Systema urinarium Urinary system Systemata genitalia Genital systems Coeloma Coelom Glandulae endocrinae Endocrine glands Systema cardiovasculare Cardiovascular system Systema lymphoideum Lymphoid system Bibliographic Reference Citation: FIPAT. Terminologia Embryologica. 2nd ed. FIPAT.library.dal.ca. Federative International Programme for Anatomical Terminology, February 2017 Published pending approval by the General Assembly at the next Congress of IFAA (2019) Creative Commons License: The publication of Terminologia Embryologica is under a Creative Commons Attribution-NoDerivatives 4.0 International (CC BY-ND 4.0) license The individual terms in this terminology are within the public domain. Statements about terms being part of this international standard terminology should use the above bibliographic reference to cite this terminology. The unaltered PDF files of this terminology may be freely copied and distributed by users. IFAA member societies are authorized to publish translations of this terminology. Authors of other works that might be considered derivative should write to the Chair of FIPAT for permission to publish a derivative work. Caput V: ORGANOGENESIS Chapter 5: ORGANOGENESIS -

Myosin Myth4-FERM Structures Highlight Important Principles of Convergent Evolution
Myosin MyTH4-FERM structures highlight important principles of convergent evolution Vicente José Planelles-Herreroa,b, Florian Blanca,c, Serena Sirigua, Helena Sirkiaa, Jeffrey Clausea, Yannick Souriguesa, Daniel O. Johnsrudd, Beatrice Amiguesa, Marco Cecchinic, Susan P. Gilberte, Anne Houdussea,1,2, and Margaret A. Titusd,1,2 aStructural Motility, Institut Curie, CNRS, UMR 144, PSL Research University, F-75005 Paris, France; bUPMC Université de Paris 6, Institut de Formation Doctorale, Sorbonne Universités, 75252 Paris Cedex 05, France; cLaboratoire d’Ingénierie des Fonctions Moléculaires, Institut de Science et d’Ingénierie Supramoléculaires, UMR 7006 CNRS, Université de Strasbourg, F-67083 Strasbourg Cedex, France; dDepartment of Genetics, Cell Biology and Development, University of Minnesota, Minneapolis, MN 55455; and eDepartment of Biological Sciences, Rensselaer Polytechnic Institute, Troy, NY 12180 Edited by James A. Spudich, Stanford University School of Medicine, Stanford, CA, and approved March 31, 2016 (received for review January 15, 2016) Myosins containing MyTH4-FERM (myosin tail homology 4-band (Fig. 1). These MF myosins are widespread and likely quite an- 4.1, ezrin, radixin, moesin, or MF) domains in their tails are found cient because they are found in many different branches of the in a wide range of phylogenetically divergent organisms, such as phylogenetic tree (5, 6), including Opisthokonts (which includes humans and the social amoeba Dictyostelium (Dd). Interestingly, Metazoa, unicellular Holozoa, and Fungi), Amoebozoa, and the evolutionarily distant MF myosins have similar roles in the exten- SAR (Stramenopiles, Alveolates, and Rhizaria) (Fig. 1 A and B). sion of actin-filled membrane protrusions such as filopodia and Over the course of hundreds of millions years of parallel evolution bind to microtubules (MT), suggesting that the core functions of the MF myosins have acquired or maintained roles in the formation these MF myosins have been highly conserved over evolution. -

Non-Syndromic Monogenic Male Infertility
Acta Biomed 2019; Vol. 90, Supplement 10: 62-67 DOI: 10.23750/abm.v90i10-S.8762 © Mattioli 1885 Review Non-syndromic monogenic male infertility Giulia Guerri1, Tiziana Maniscalchi2, Shila Barati2, Gian Maria Busetto3, Francesco Del Giudice3, Ettore De Berardinis3, Rossella Cannarella4, Aldo Eugenio Calogero4, Matteo Bertelli2 1 MAGI’s Lab, Rovereto (TN), Italy; 2 MAGI Euregio, Bolzano, Italy; 3 Department of Urology, University of Rome La Sapien- za, Policlinico Umberto I, Rome, Italy; 4 Department of Clinical and Experimental Medicine, University of Catania, Catania, Italy Summary. Infertility is a widespread clinical problem affecting 8-12% of couples worldwide. Of these, about 30% are diagnosed with idiopathic infertility since no causative factor is found. Overall 40-50% of cases are due to male reproductive defects. Numerical or structural chromosome abnormalities have long been associ- ated with male infertility. Monogenic mutations have only recently been addressed in the pathogenesis of this condition. Mutations of specific genes involved in meiosis, mitosis or spermiohistogenesis result in spermato- genic failure, leading to the following anomalies: insufficient (oligozoospermia) or no (azoospermia) sperm production, limited progressive and/or total sperm motility (asthenozoospermia), altered sperm morphology (teratozoospermia), or combinations thereof. Androgen insensitivity, causing hormonal and sexual impair- ment in males with normal karyotype, also affects male fertility. The genetic causes of non-syndromic mono- genic of male infertility are summarized in this article and a gene panel is proposed. (www.actabiomedica.it) Key words: male infertility, oligozoospermia, azoospermia, asthenozoospermia, teratozoospermia, spermato- genic failure, androgen insensitivity syndrome Introduction development. Genetic causes of male infertility are outlined in Table 1. -

Defining Functional Interactions During Biogenesis of Epithelial Junctions
ARTICLE Received 11 Dec 2015 | Accepted 13 Oct 2016 | Published 6 Dec 2016 | Updated 5 Jan 2017 DOI: 10.1038/ncomms13542 OPEN Defining functional interactions during biogenesis of epithelial junctions J.C. Erasmus1,*, S. Bruche1,*,w, L. Pizarro1,2,*, N. Maimari1,3,*, T. Poggioli1,w, C. Tomlinson4,J.Lees5, I. Zalivina1,w, A. Wheeler1,w, A. Alberts6, A. Russo2 & V.M.M. Braga1 In spite of extensive recent progress, a comprehensive understanding of how actin cytoskeleton remodelling supports stable junctions remains to be established. Here we design a platform that integrates actin functions with optimized phenotypic clustering and identify new cytoskeletal proteins, their functional hierarchy and pathways that modulate E-cadherin adhesion. Depletion of EEF1A, an actin bundling protein, increases E-cadherin levels at junctions without a corresponding reinforcement of cell–cell contacts. This unexpected result reflects a more dynamic and mobile junctional actin in EEF1A-depleted cells. A partner for EEF1A in cadherin contact maintenance is the formin DIAPH2, which interacts with EEF1A. In contrast, depletion of either the endocytic regulator TRIP10 or the Rho GTPase activator VAV2 reduces E-cadherin levels at junctions. TRIP10 binds to and requires VAV2 function for its junctional localization. Overall, we present new conceptual insights on junction stabilization, which integrate known and novel pathways with impact for epithelial morphogenesis, homeostasis and diseases. 1 National Heart and Lung Institute, Faculty of Medicine, Imperial College London, London SW7 2AZ, UK. 2 Computing Department, Imperial College London, London SW7 2AZ, UK. 3 Bioengineering Department, Faculty of Engineering, Imperial College London, London SW7 2AZ, UK. 4 Department of Surgery & Cancer, Faculty of Medicine, Imperial College London, London SW7 2AZ, UK. -

Circmyo10 Promotes Osteosarcoma Progression by Regulating Mir-370
Chen et al. Molecular Cancer (2019) 18:150 https://doi.org/10.1186/s12943-019-1076-1 RESEARCH Open Access CircMYO10 promotes osteosarcoma progression by regulating miR-370-3p/ RUVBL1 axis to enhance the transcriptional activity of β-catenin/LEF1 complex via effects on chromatin remodeling Junxin Chen1†, Gang Liu1†, Yizheng Wu1†, Jianjun Ma1†, Hongfei Wu2, Ziang Xie1, Shuai Chen1, Yute Yang1, Shengyu Wang1, Panyang Shen1, Yifan Fang3, Shunwu Fan1, Shuying Shen1* and Xiangqian Fang1* Abstract Background: CircMYO10 is a circular RNA generated by back-splicing of gene MYO10 and is upregulated in osteosarcoma cell lines, but its functional role in osteosarcoma is still unknown. This study aimed to clarify the mechanism of circMYO10 in osteosarcoma. Methods: CircMYO10 expression in 10 paired osteosarcoma and chondroma tissues was assessed by quantitative reverse transcription polymerase chain reaction (PCR). The function of circMYO10/miR-370-3p/RUVBL1 axis was assessed regarding two key characteristics: proliferation and endothelial–mesenchymal transition (EMT). Bioinformatics analysis, western blotting, real-time PCR, fluorescence in situ hybridization, immunoprecipitation, RNA pull-down assays, luciferase reporter assays, chromatin immunoprecipitation, and rescue experiments were used to evaluate the mechanism. Stably transfected MG63 cells were injected via tail vein or subcutaneously into nude mice to assess the role of circMYO10 in vivo. Results: CircMYO10 was significantly upregulated, while miR-370-3p was downregulated, in osteosarcoma cell lines and human osteosarcoma samples. Silencing circMYO10 inhibited cell proliferation and EMT in vivo and in vitro. Mechanistic investigations revealed that miR-370-3p targets RUVBL1 directly, and inhibits the interaction between RUVBL1 and β-catenin/LEF1 complex while circMYO10 showed a contrary effect via the inhibition of miR-370-3p. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -
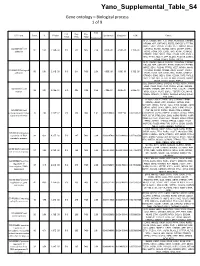
Supplemental Tables4.Pdf
Yano_Supplemental_Table_S4 Gene ontology – Biological process 1 of 9 Fold List Pop Pop GO Term Count % PValue Bonferroni Benjamini FDR Genes Total Hits Total Enrichment DLC1, CADM1, NELL2, CLSTN1, PCDHGA8, CTNNB1, NRCAM, APP, CNTNAP2, FERT2, RAPGEF1, PTPRM, MPDZ, SDK1, PCDH9, PTPRS, VEZT, NRXN1, MYH9, GO:0007155~cell CTNNA2, NCAM1, NCAM2, DDR1, LSAMP, CNTN1, 50 5.61 2.14E-08 510 311 7436 2.34 4.50E-05 4.50E-05 3.70E-05 adhesion ROR2, VCAN, DST, LIMS1, TNC, ASTN1, CTNND2, CTNND1, CDH2, NEO1, CDH4, CD24A, FAT3, PVRL3, TRO, TTYH1, MLLT4, LPP, NLGN1, PCDH19, LAMA1, ITGA9, CDH13, CDON, PSPC1 DLC1, CADM1, NELL2, CLSTN1, PCDHGA8, CTNNB1, NRCAM, APP, CNTNAP2, FERT2, RAPGEF1, PTPRM, MPDZ, SDK1, PCDH9, PTPRS, VEZT, NRXN1, MYH9, GO:0022610~biological CTNNA2, NCAM1, NCAM2, DDR1, LSAMP, CNTN1, 50 5.61 2.14E-08 510 311 7436 2.34 4.50E-05 4.50E-05 3.70E-05 adhesion ROR2, VCAN, DST, LIMS1, TNC, ASTN1, CTNND2, CTNND1, CDH2, NEO1, CDH4, CD24A, FAT3, PVRL3, TRO, TTYH1, MLLT4, LPP, NLGN1, PCDH19, LAMA1, ITGA9, CDH13, CDON, PSPC1 DCC, ENAH, PLXNA2, CAPZA2, ATP5B, ASTN1, PAX6, ZEB2, CDH2, CDH4, GLI3, CD24A, EPHB1, NRCAM, GO:0006928~cell CTTNBP2, EDNRB, APP, PTK2, ETV1, CLASP2, STRBP, 36 4.04 3.46E-07 510 205 7436 2.56 7.28E-04 3.64E-04 5.98E-04 motion NRG1, DCLK1, PLAT, SGPL1, TGFBR1, EVL, MYH9, YWHAE, NCKAP1, CTNNA2, SEMA6A, EPHA4, NDEL1, FYN, LRP6 PLXNA2, ADCY5, PAX6, GLI3, CTNNB1, LPHN2, EDNRB, LPHN3, APP, CSNK2A1, GPR45, NRG1, RAPGEF1, WWOX, SGPL1, TLE4, SPEN, NCAM1, DDR1, GRB10, GRM3, GNAQ, HIPK1, GNB1, HIPK2, PYGO1, GO:0007166~cell RNF138, ROR2, CNTN1, -
The Reproductive System
27 The Reproductive System PowerPoint® Lecture Presentations prepared by Steven Bassett Southeast Community College Lincoln, Nebraska © 2012 Pearson Education, Inc. Introduction • The reproductive system is designed to perpetuate the species • The male produces gametes called sperm cells • The female produces gametes called ova • The joining of a sperm cell and an ovum is fertilization • Fertilization results in the formation of a zygote © 2012 Pearson Education, Inc. Anatomy of the Male Reproductive System • Overview of the Male Reproductive System • Testis • Epididymis • Ductus deferens • Ejaculatory duct • Spongy urethra (penile urethra) • Seminal gland • Prostate gland • Bulbo-urethral gland © 2012 Pearson Education, Inc. Figure 27.1 The Male Reproductive System, Part I Pubic symphysis Ureter Urinary bladder Prostatic urethra Seminal gland Membranous urethra Rectum Corpus cavernosum Prostate gland Corpus spongiosum Spongy urethra Ejaculatory duct Ductus deferens Penis Bulbo-urethral gland Epididymis Anus Testis External urethral orifice Scrotum Sigmoid colon (cut) Rectum Internal urethral orifice Rectus abdominis Prostatic urethra Urinary bladder Prostate gland Pubic symphysis Bristle within ejaculatory duct Membranous urethra Penis Spongy urethra Spongy urethra within corpus spongiosum Bulbospongiosus muscle Corpus cavernosum Ductus deferens Epididymis Scrotum Testis © 2012 Pearson Education, Inc. Anatomy of the Male Reproductive System • The Testes • Testes hang inside a pouch called the scrotum, which is on the outside of the body -

Male Reproductive System
MALE REPRODUCTIVE SYSTEM DR RAJARSHI ASH M.B.B.S.(CAL); D.O.(EYE) ; M.D.-PGT(2ND YEAR) DEPARTMENT OF PHYSIOLOGY CALCUTTA NATIONAL MEDICAL COLLEGE PARTS OF MALE REPRODUCTIVE SYSTEM A. Gonads – Two ovoid testes present in scrotal sac, out side the abdominal cavity B. Accessory sex organs - epididymis, vas deferens, seminal vesicles, ejaculatory ducts, prostate gland and bulbo-urethral glands C. External genitalia – penis and scrotum ANATOMY OF MALE INTERNAL GENITALIA AND ACCESSORY SEX ORGANS SEMINIFEROUS TUBULE Two principal cell types in seminiferous tubule Sertoli cell Germ cell INTERACTION BETWEEN SERTOLI CELLS AND SPERM BLOOD- TESTIS BARRIER • Blood – testis barrier protects germ cells in seminiferous tubules from harmful elements in blood. • The blood- testis barrier prevents entry of antigenic substances from the developing germ cells into circulation. • High local concentration of androgen, inositol, glutamic acid, aspartic acid can be maintained in the lumen of seminiferous tubule without difficulty. • Blood- testis barrier maintains higher osmolality of luminal content of seminiferous tubules. FUNCTIONS OF SERTOLI CELLS 1.Germ cell development 2.Phagocytosis 3.Nourishment and growth of spermatids 4.Formation of tubular fluid 5.Support spermiation 6.FSH and testosterone sensitivity 7.Endocrine functions of sertoli cells i)Inhibin ii)Activin iii)Follistatin iv)MIS v)Estrogen 8.Sertoli cell secretes ‘Androgen binding protein’(ABP) and H-Y antigen. 9.Sertoli cell contributes formation of blood testis barrier. LEYDIG CELL • Leydig cells are present near the capillaries in the interstitial space between seminiferous tubules. • They are rich in mitochondria & endoplasmic reticulum. • Leydig cells secrete testosterone,DHEA & Androstenedione. • The activity of leydig cell is different in different phases of life. -

Genetic Counseling and Diagnostic Guidelines for Couples with Infertility And/Or Recurrent Miscarriage
medizinische genetik 2021; 33(1): 3–12 Margot J. Wyrwoll, Sabine Rudnik-Schöneborn, and Frank Tüttelmann* Genetic counseling and diagnostic guidelines for couples with infertility and/or recurrent miscarriage https://doi.org/10.1515/medgen-2021-2051 to both partners prior to undergoing assisted reproduc- Received January 16, 2021; accepted February 11, 2021 tive technology. In couples with recurrent miscarriages, Abstract: Around 10–15 % of all couples are infertile, karyotyping is recommended to detect balanced structural rendering infertility a widespread disease. Male and fe- chromosomal aberrations. male causes contribute equally to infertility, and, de- Keywords: female infertility, male infertility, miscarriages, pending on the defnition, roughly 1 % to 5 % of all genetic counseling, ART couples experience recurrent miscarriages. In German- speaking countries, recommendations for infertile cou- ples and couples with recurrent miscarriages are pub- lished as consensus-based (S2k) Guidelines by the “Ar- Introduction beitsgemeinschaft der Wissenschaftlichen Medizinischen Fachgesellschaften” (AWMF). This article summarizes the A large proportion of genetic consultation appointments current recommendations with regard to genetic counsel- is attributed to infertile couples and couples with recur- ing and diagnostics. rent miscarriages. Infertility, which is defned by the WHO Prior to genetic counseling, the infertile couple as the inability to achieve a pregnancy after one year of must undergo a gynecological/andrological examination, unprotected intercourse [1], afects 10–15 % of all couples, which includes anamnesis, hormonal profling, physical thus rendering infertility a widespread disease, compara- examination and genital ultrasound. Women should be ex- ble to, e. g., high blood pressure or depression. Histori- amined for the presence of hyperandrogenemia. Men must cally, the female partner has been the focus of diagnos- further undergo a semen analysis. -

Spectrum of MYO7A Mutations in an Indigenous South African
G C A T T A C G G C A T genes Article Spectrum of MYO7A Mutations in an Indigenous South African Population Further Elucidates the Nonsyndromic Autosomal Recessive Phenotype of DFNB2 to Include Both Homozygous and Compound Heterozygous Mutations Rosemary Ida Kabahuma 1,2,*, Wolf-Dieter Schubert 2, Christiaan Labuschagne 3, Denise Yan 4, Susan Halloran Blanton 4,5 , Michael Sean Pepper 6 and Xue Zhong Liu 4,5,* 1 Department of Otorhinolaryngology, University of Pretoria, Pretoria 0001, South Africa 2 Departments of Biochemistry, Genetics and Microbiology, Faculty of Natural and Agricultural Sciences, University of Pretoria, Pretoria 0001, South Africa; [email protected] 3 Inqaba Biotechnical Industries, Pretoria 0002, South Africa; [email protected] 4 Department Otolaryngology, University of Miami Miller School of Medicine, Miami, FL 33136, USA; [email protected] (D.Y.); [email protected] (S.H.B.) 5 Dr. John T. Macdonald Foundation Department of Human Genetics, and John P. Hussman Institute for Human Genomics, University of Miami Miller School of Medicine, Miami, FL 33136, USA 6 Department Immunology and SAMRC Extramural Unit for Stem Cell Research and Therapy, Faculty of Health Sciences, Institute for Cellular and Molecular Medicine, University of Pretoria, Citation: Kabahuma, R.I.; Pretoria 0001, South Africa; [email protected] Schubert, W.-D.; Labuschagne, C.; * Correspondence: [email protected] (R.I.K.); [email protected] (X.Z.L.) Yan, D.; Blanton, S.H.; Pepper, M.S.; Liu, X.Z. Spectrum of MYO7A Abstract: MYO7A gene encodes unconventional myosin VIIA, which, when mutated, causes a phe- Mutations in an Indigenous South notypic spectrum ranging from recessive hearing loss DFNB2 to deaf-blindness, Usher Type 1B African Population Further (USH1B).