Downloaded from in December 2015)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Mutations in CDK5RAP2 Cause Seckel Syndrome Go¨ Khan Yigit1,2,3,A, Karen E
ORIGINAL ARTICLE Mutations in CDK5RAP2 cause Seckel syndrome Go¨ khan Yigit1,2,3,a, Karen E. Brown4,a,Hu¨ lya Kayserili5, Esther Pohl1,2,3, Almuth Caliebe6, Diana Zahnleiter7, Elisabeth Rosser8, Nina Bo¨ gershausen1,2,3, Zehra Oya Uyguner5, Umut Altunoglu5, Gudrun Nu¨ rnberg2,3,9, Peter Nu¨ rnberg2,3,9, Anita Rauch10, Yun Li1,2,3, Christian Thomas Thiel7 & Bernd Wollnik1,2,3 1Institute of Human Genetics, University of Cologne, Cologne, Germany 2Center for Molecular Medicine Cologne (CMMC), University of Cologne, Cologne, Germany 3Cologne Excellence Cluster on Cellular Stress Responses in Aging-Associated Diseases (CECAD), University of Cologne, Cologne, Germany 4Chromosome Biology Group, MRC Clinical Sciences Centre, Imperial College School of Medicine, Hammersmith Hospital, London, W12 0NN, UK 5Department of Medical Genetics, Istanbul Medical Faculty, Istanbul University, Istanbul, Turkey 6Institute of Human Genetics, Christian-Albrechts-University of Kiel, Kiel, Germany 7Institute of Human Genetics, Friedrich-Alexander University Erlangen-Nuremberg, Erlangen, Germany 8Department of Clinical Genetics, Great Ormond Street Hospital for Children, London, WC1N 3EH, UK 9Cologne Center for Genomics, University of Cologne, Cologne, Germany 10Institute of Medical Genetics, University of Zurich, Schwerzenbach-Zurich, Switzerland Keywords Abstract CDK5RAP2, CEP215, microcephaly, primordial dwarfism, Seckel syndrome Seckel syndrome is a heterogeneous, autosomal recessive disorder marked by pre- natal proportionate short stature, severe microcephaly, intellectual disability, and Correspondence characteristic facial features. Here, we describe the novel homozygous splice-site Bernd Wollnik, Center for Molecular mutations c.383+1G>C and c.4005-9A>GinCDK5RAP2 in two consanguineous Medicine Cologne (CMMC) and Institute of families with Seckel syndrome. CDK5RAP2 (CEP215) encodes a centrosomal pro- Human Genetics, University of Cologne, tein which is known to be essential for centrosomal cohesion and proper spindle Kerpener Str. -

Defining Functional Interactions During Biogenesis of Epithelial Junctions
ARTICLE Received 11 Dec 2015 | Accepted 13 Oct 2016 | Published 6 Dec 2016 | Updated 5 Jan 2017 DOI: 10.1038/ncomms13542 OPEN Defining functional interactions during biogenesis of epithelial junctions J.C. Erasmus1,*, S. Bruche1,*,w, L. Pizarro1,2,*, N. Maimari1,3,*, T. Poggioli1,w, C. Tomlinson4,J.Lees5, I. Zalivina1,w, A. Wheeler1,w, A. Alberts6, A. Russo2 & V.M.M. Braga1 In spite of extensive recent progress, a comprehensive understanding of how actin cytoskeleton remodelling supports stable junctions remains to be established. Here we design a platform that integrates actin functions with optimized phenotypic clustering and identify new cytoskeletal proteins, their functional hierarchy and pathways that modulate E-cadherin adhesion. Depletion of EEF1A, an actin bundling protein, increases E-cadherin levels at junctions without a corresponding reinforcement of cell–cell contacts. This unexpected result reflects a more dynamic and mobile junctional actin in EEF1A-depleted cells. A partner for EEF1A in cadherin contact maintenance is the formin DIAPH2, which interacts with EEF1A. In contrast, depletion of either the endocytic regulator TRIP10 or the Rho GTPase activator VAV2 reduces E-cadherin levels at junctions. TRIP10 binds to and requires VAV2 function for its junctional localization. Overall, we present new conceptual insights on junction stabilization, which integrate known and novel pathways with impact for epithelial morphogenesis, homeostasis and diseases. 1 National Heart and Lung Institute, Faculty of Medicine, Imperial College London, London SW7 2AZ, UK. 2 Computing Department, Imperial College London, London SW7 2AZ, UK. 3 Bioengineering Department, Faculty of Engineering, Imperial College London, London SW7 2AZ, UK. 4 Department of Surgery & Cancer, Faculty of Medicine, Imperial College London, London SW7 2AZ, UK. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -
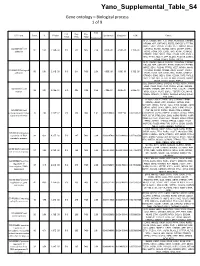
Supplemental Tables4.Pdf
Yano_Supplemental_Table_S4 Gene ontology – Biological process 1 of 9 Fold List Pop Pop GO Term Count % PValue Bonferroni Benjamini FDR Genes Total Hits Total Enrichment DLC1, CADM1, NELL2, CLSTN1, PCDHGA8, CTNNB1, NRCAM, APP, CNTNAP2, FERT2, RAPGEF1, PTPRM, MPDZ, SDK1, PCDH9, PTPRS, VEZT, NRXN1, MYH9, GO:0007155~cell CTNNA2, NCAM1, NCAM2, DDR1, LSAMP, CNTN1, 50 5.61 2.14E-08 510 311 7436 2.34 4.50E-05 4.50E-05 3.70E-05 adhesion ROR2, VCAN, DST, LIMS1, TNC, ASTN1, CTNND2, CTNND1, CDH2, NEO1, CDH4, CD24A, FAT3, PVRL3, TRO, TTYH1, MLLT4, LPP, NLGN1, PCDH19, LAMA1, ITGA9, CDH13, CDON, PSPC1 DLC1, CADM1, NELL2, CLSTN1, PCDHGA8, CTNNB1, NRCAM, APP, CNTNAP2, FERT2, RAPGEF1, PTPRM, MPDZ, SDK1, PCDH9, PTPRS, VEZT, NRXN1, MYH9, GO:0022610~biological CTNNA2, NCAM1, NCAM2, DDR1, LSAMP, CNTN1, 50 5.61 2.14E-08 510 311 7436 2.34 4.50E-05 4.50E-05 3.70E-05 adhesion ROR2, VCAN, DST, LIMS1, TNC, ASTN1, CTNND2, CTNND1, CDH2, NEO1, CDH4, CD24A, FAT3, PVRL3, TRO, TTYH1, MLLT4, LPP, NLGN1, PCDH19, LAMA1, ITGA9, CDH13, CDON, PSPC1 DCC, ENAH, PLXNA2, CAPZA2, ATP5B, ASTN1, PAX6, ZEB2, CDH2, CDH4, GLI3, CD24A, EPHB1, NRCAM, GO:0006928~cell CTTNBP2, EDNRB, APP, PTK2, ETV1, CLASP2, STRBP, 36 4.04 3.46E-07 510 205 7436 2.56 7.28E-04 3.64E-04 5.98E-04 motion NRG1, DCLK1, PLAT, SGPL1, TGFBR1, EVL, MYH9, YWHAE, NCKAP1, CTNNA2, SEMA6A, EPHA4, NDEL1, FYN, LRP6 PLXNA2, ADCY5, PAX6, GLI3, CTNNB1, LPHN2, EDNRB, LPHN3, APP, CSNK2A1, GPR45, NRG1, RAPGEF1, WWOX, SGPL1, TLE4, SPEN, NCAM1, DDR1, GRB10, GRM3, GNAQ, HIPK1, GNB1, HIPK2, PYGO1, GO:0007166~cell RNF138, ROR2, CNTN1, -

On the Turning of Xenopus Retinal Axons Induced by Ephrin-A5
Development 130, 1635-1643 1635 © 2003 The Company of Biologists Ltd doi:10.1242/dev.00386 On the turning of Xenopus retinal axons induced by ephrin-A5 Christine Weinl1, Uwe Drescher2,*, Susanne Lang1, Friedrich Bonhoeffer1 and Jürgen Löschinger1 1Max-Planck-Institute for Developmental Biology, Spemannstrasse 35, 72076 Tübingen, Germany 2MRC Centre for Developmental Neurobiology, King’s College London, New Hunt’s House, Guy’s Hospital Campus, London SE1 1UL, UK *Author for correspondence (e-mail: [email protected]) Accepted 13 January 2003 SUMMARY The Eph family of receptor tyrosine kinases and their turning or growth cone collapse when confronted with ligands, the ephrins, play important roles during ephrin-A5-Fc bound to beads. However, when added in development of the nervous system. Frequently they exert soluble form to the medium, ephrin-A5 induces growth their functions through a repellent mechanism, so that, for cone collapse, comparable to data from chick. example, an axon expressing an Eph receptor does not The analysis of growth cone behaviour in a gradient of invade a territory in which an ephrin is expressed. Eph soluble ephrin-A5 in the ‘turning assay’ revealed a receptor activation requires membrane-associated ligands. substratum-dependent reaction of Xenopus retinal axons. This feature discriminates ephrins from other molecules On fibronectin, we observed a repulsive response, with the sculpturing the nervous system such as netrins, slits and turning of growth cones away from higher concentrations class 3 semaphorins, which are secreted molecules. While of ephrin-A5. On laminin, retinal axons turned towards the ability of secreted molecules to guide axons, i.e. -

Endothelin-2 Signaling in the Neural Retina Promotes the Endothelial Tip Cell State and Inhibits Angiogenesis
Endothelin-2 signaling in the neural retina promotes PNAS PLUS the endothelial tip cell state and inhibits angiogenesis Amir Rattnera,1, Huimin Yua, John Williamsa,b, Philip M. Smallwooda,b, and Jeremy Nathansa,b,c,d,1 Departments of aMolecular Biology and Genetics, cNeuroscience, and dOphthalmology and bHoward Hughes Medical Institute, Johns Hopkins University School of Medicine, Baltimore, MD 21205 Contributed by Jeremy Nathans, August 20, 2013 (sent for review February 19, 2013) Endothelin signaling is required for neural crest migration and time lapse imaging studies of vascular development in zebrafish homeostatic regulation of blood pressure. Here, we report that and mammalian EC dynamics in explant culture show that the tip constitutive overexpression of Endothelin-2 (Edn2) in the mouse cell and stalk cell states are highly plastic, with frequent retina perturbs vascular development by inhibiting endothelial cell exchanges between the two cell states (8, 9). migration across the retinal surface and subsequent endothelial Several other signaling pathways are also essential for retinal cell invasion into the retina. Developing endothelial cells exist in vascular development. Norrin, a Muller-glia–derived ligand, and one of two states: tip cells at the growing front and stalk cells in its EC receptor Frizzled4 (Fz4), coreceptor Lrp5, and receptor the vascular plexus behind the front. This division of endothelial chaperone Tspan12 activate canonical Wnt signaling in de- cell states is one of the central organizing principles of angiogen- veloping ECs (10). In humans and mice, defects in any of these esis. In the developing retina, Edn2 overexpression leads to components lead to retinal hypovascularization. -

Serum Albumin OS=Homo Sapiens
Protein Name Cluster of Glial fibrillary acidic protein OS=Homo sapiens GN=GFAP PE=1 SV=1 (P14136) Serum albumin OS=Homo sapiens GN=ALB PE=1 SV=2 Cluster of Isoform 3 of Plectin OS=Homo sapiens GN=PLEC (Q15149-3) Cluster of Hemoglobin subunit beta OS=Homo sapiens GN=HBB PE=1 SV=2 (P68871) Vimentin OS=Homo sapiens GN=VIM PE=1 SV=4 Cluster of Tubulin beta-3 chain OS=Homo sapiens GN=TUBB3 PE=1 SV=2 (Q13509) Cluster of Actin, cytoplasmic 1 OS=Homo sapiens GN=ACTB PE=1 SV=1 (P60709) Cluster of Tubulin alpha-1B chain OS=Homo sapiens GN=TUBA1B PE=1 SV=1 (P68363) Cluster of Isoform 2 of Spectrin alpha chain, non-erythrocytic 1 OS=Homo sapiens GN=SPTAN1 (Q13813-2) Hemoglobin subunit alpha OS=Homo sapiens GN=HBA1 PE=1 SV=2 Cluster of Spectrin beta chain, non-erythrocytic 1 OS=Homo sapiens GN=SPTBN1 PE=1 SV=2 (Q01082) Cluster of Pyruvate kinase isozymes M1/M2 OS=Homo sapiens GN=PKM PE=1 SV=4 (P14618) Glyceraldehyde-3-phosphate dehydrogenase OS=Homo sapiens GN=GAPDH PE=1 SV=3 Clathrin heavy chain 1 OS=Homo sapiens GN=CLTC PE=1 SV=5 Filamin-A OS=Homo sapiens GN=FLNA PE=1 SV=4 Cytoplasmic dynein 1 heavy chain 1 OS=Homo sapiens GN=DYNC1H1 PE=1 SV=5 Cluster of ATPase, Na+/K+ transporting, alpha 2 (+) polypeptide OS=Homo sapiens GN=ATP1A2 PE=3 SV=1 (B1AKY9) Fibrinogen beta chain OS=Homo sapiens GN=FGB PE=1 SV=2 Fibrinogen alpha chain OS=Homo sapiens GN=FGA PE=1 SV=2 Dihydropyrimidinase-related protein 2 OS=Homo sapiens GN=DPYSL2 PE=1 SV=1 Cluster of Alpha-actinin-1 OS=Homo sapiens GN=ACTN1 PE=1 SV=2 (P12814) 60 kDa heat shock protein, mitochondrial OS=Homo -

Mir-146A Regulates Migration and Invasion by Targeting NRP2 in Circulating-Tumor Cell Mimicking Suspension Cells
G C A T T A C G G C A T genes Article MiR-146a Regulates Migration and Invasion by Targeting NRP2 in Circulating-Tumor Cell Mimicking Suspension Cells Yeojin Do 1, Jin Gu Cho 1, Ji Young Park 1, Sumin Oh 1, Doyeon Park 1 , Kyung Hyun Yoo 1,2 , Myeong-Sok Lee 1,2, Byung Su Kwon 3,* , Jongmin Kim 1,2,* and Young Yang 2,* 1 Division of Biological Sciences, Sookmyung Women’s University, Seoul 04310, Korea; [email protected] (Y.D.); [email protected] (J.G.C.); [email protected] (J.Y.P.); [email protected] (S.O.); [email protected] (D.P.); [email protected] (K.H.Y.); [email protected] (M.-S.L.) 2 Research Institute for Women’s Health, Sookmyung Women’s University, Seoul 04310, Korea 3 Department of Obstetrics and Gynecology, Kyung Hee University Medical Center, 23, Seoul 02447, Korea * Correspondence: [email protected] (B.S.K.); [email protected] (J.K.); [email protected] (Y.Y.); Tel.: +82-2-958-8837 (B.S.K.); +82-2-710-9553 (J.K.); +82-2-710-9590 (Y.Y.) Abstract: Cancer metastasis is the primary cause of cancer-related death and metastatic cancer has circulating-tumor cells (CTCs), which circulate in the bloodstream before invading other organs. Thus, understanding the precise role of CTCs may provide new insights into the metastasis process and reduce cancer mortality. However, the molecular characteristics of CTCs are not well understood due to a lack of number of CTCs. -

3B and Semaphorin-3F in Ovarian Cancer
Published OnlineFirst February 2, 2010; DOI: 10.1158/1535-7163.MCT-09-0664 Research Article Molecular Cancer Therapeutics Hormonal Regulation and Distinct Functions of Semaphorin- 3B and Semaphorin-3F in Ovarian Cancer Doina Joseph1, Shuk-Mei Ho2, and Viqar Syed1 Abstract Semaphorins comprise a family of molecules that influence neuronal growth and guidance. Class-3 sema- phorins, semaphorin-3B (SEMA3B) and semaphorin-3F (SEMA3F), illustrate their effects by forming a com- plex with neuropilins (NP-1 or NP-2) and plexins. We examined the status and regulation of semaphorins and their receptors in human ovarian cancer cells. A significantly reduced expression of SEMA3B (83 kDa), SE- MA3F (90 kDa), and plexin-A3 was observed in ovarian cancer cell lines when compared with normal human ovarian surface epithelial cells. The expression of NP-1, NP-2, and plexin-A1 was not altered in human ovar- ian surface epithelial and ovarian cancer cells. The decreased expression of SEMA3B, SEMA3F, and plexin-A3 was confirmed in stage 3 ovarian tumors. The treatment of ovarian cancer cells with luteinizing hormone, follicle-stimulating hormone, and estrogen induced a significant upregulation of SEMA3B, whereas SEMA3F was upregulated only by estrogen. Cotreatment of cell lines with a hormone and its specific antagonist blocked the effect of the hormone. Ectopic expression of SEMA3B or SEMA3F reduced soft-agar colony for- mation, adhesion, and cell invasion of ovarian cancer cell cultures. Forced expression of SEMA3B, but not SEMA3F, inhibited viability of ovarian cancer cells. Overexpression of SEMA3B and SEMA3F reduced focal adhesion kinase phosphorylation and matrix metalloproteinase-2 and matrix metalloproteinase-9 expression in ovarian cancer cells. -

MYO5A Rabbit Pab
Leader in Biomolecular Solutions for Life Science MYO5A Rabbit pAb Catalog No.: A9830 Basic Information Background Catalog No. This gene is one of three myosin V heavy-chain genes, belonging to the myosin gene A9830 superfamily. Myosin V is a class of actin-based motor proteins involved in cytoplasmic vesicle transport and anchorage, spindle-pole alignment and mRNA translocation. The Observed MW protein encoded by this gene is abundant in melanocytes and nerve cells. Mutations in 240kDa this gene cause Griscelli syndrome type-1 (GS1), Griscelli syndrome type-3 (GS3) and neuroectodermal melanolysosomal disease, or Elejalde disease. Multiple alternatively Calculated MW spliced transcript variants encoding different isoforms have been reported, but the full- 212kDa/215kDa/218kDa length nature of some variants has not been determined. Category Primary antibody Applications WB,IHC,IF Cross-Reactivity Human, Mouse, Rat Recommended Dilutions Immunogen Information WB 1:500 - 1:2000 Gene ID Swiss Prot 4644 Q9Y4I1 IHC 1:50 - 1:100 Immunogen 1:50 - 1:100 IF Recombinant fusion protein containing a sequence corresponding to amino acids 950-1150 of human MYO5A (NP_000250.3). Synonyms MYO5A;GS1;MYH12;MYO5;MYR12;myosin VA Contact Product Information www.abclonal.com Source Isotype Purification Rabbit IgG Affinity purification Storage Store at -20℃. Avoid freeze / thaw cycles. Buffer: PBS with 0.02% sodium azide,50% glycerol,pH7.3. Validation Data Western blot analysis of extracts of various cell lines, using MYO5A antibody (A9830) at 1:1000 dilution. Secondary antibody: HRP Goat Anti-Rabbit IgG (H+L) (AS014) at 1:10000 dilution. Lysates/proteins: 25ug per lane. Blocking buffer: 3% nonfat dry milk in TBST. -

Semaphorin Signaling Guides Cranial Neural Crest Cell Migration in Zebrafish
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Developmental Biology 280 (2005) 373–385 www.elsevier.com/locate/ydbio Semaphorin signaling guides cranial neural crest cell migration in zebrafish Hung-Hsiang Yu, Cecilia B. Moens* Howard Hughes Medical Institute and Division of Basic Science, Fred Hutchinson Cancer Research Center, 1100 Fairview Avenue N., Box 19024 Seattle, WA 98109, USA Received for publication 2 December 2004, revised 19 January 2005, accepted 28 January 2005 Abstract Cranial neural crest cells (NCCs) migrate into the pharyngeal arches in three primary streams separated by two cranial neural crest (NC)- free zones. Multiple tissues have been implicated in the guidance of cranial NCC migration; however, the signals provided by these tissues have remained elusive. We investigate the function of semaphorins (semas) and their receptors, neuropilins (nrps), in cranial NCC migration in zebrafish. We find that genes of the sema3F and sema3G class are expressed in the cranial NC-free zones, while nrp2a and nrp2b are expressed in the migrating NCCs. sema3F/3G expression is expanded homogeneously in the head periphery through which the cranial NCCs migrate in lzr/pbx4 mutants, in which the cranial NC streams are fused. Antisense morpholino knockdown of Sema3F/3G or Nrp2 suppresses the abnormal cranial NC phenotype of lzr/pbx4 mutants, demonstrating that aberrant Sema3F/3G-Nrp2 signaling is responsible for this phenotype and suggesting that repulsive Sema3F/3G-Npn2 signaling normally contributes to the guidance of migrating cranial NCCs. Furthermore, global over-expression of sema3Gb phenocopies the aberrant cranial NC phenotype of lzr/pbx4 mutants when endogenous Sema3 ligands are knocked down, consistent with a model in which the patterned expression of Sema3 ligands in the head periphery coordinates the migration of Nrp-expressing cranial NCCs. -

Hepatocyte Growth Factor: a Regulator of Inflammation and Autoimmunity
Autoimmunity Reviews 14 (2015) 293–303 Contents lists available at ScienceDirect Autoimmunity Reviews journal homepage: www.elsevier.com/locate/autrev Review Hepatocyte growth factor: A regulator of inflammation and autoimmunity Nicolas Molnarfi a,b,1, Mahdia Benkhoucha a,b,1, Hiroshi Funakoshi d, Toshikazu Nakamura e, Patrice H. Lalive a,b,c,⁎ a Department of Pathology and Immunology, Faculty of Medicine, University of Geneva, Geneva, Switzerland b Department of Clinical Neurosciences, Division of Neurology, Unit of Neuroimmunology and Multiple Sclerosis, University Hospital of Geneva, Geneva, Switzerland c Department of Genetics and Laboratory Medicine, Laboratory Medicine Service, University Hospital of Geneva, Geneva, Switzerland d Center for Advanced Research and Education, Asahikawa Medical University, Asahikawa, Japan e Neurogen Inc., Nakahozumi, Ibaraki, Osaka, Japan article info abstract Article history: Hepatocyte growth factor (HGF) is a pleiotropic cytokine that has been extensively studied over several decades, Received 20 November 2014 but was only recently recognized as a key player in mediating protection of many types of inflammatory and au- Accepted 25 November 2014 toimmune diseases. HGF was reported to prevent and attenuate disease progression by influencing multiple Available online 1 December 2014 pathophysiological processes involved in inflammatory and immune response, including cell migration, matura- tion, cytokine production, antigen presentation, and T cell effector function. In this review, we discuss the actions Keywords: fl HGF and mechanisms of HGF in in ammation and immunity and the therapeutic potential of this factor for the treat- fl c-Met ment of in ammatory and autoimmune diseases. Inflammation © 2014 Elsevier B.V. All rights reserved. Autoimmunity Autoimmune regulator Therapy Contents 1.