Myth4-FERM Myosin Based Filopodia Initiation
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Defining Functional Interactions During Biogenesis of Epithelial Junctions
ARTICLE Received 11 Dec 2015 | Accepted 13 Oct 2016 | Published 6 Dec 2016 | Updated 5 Jan 2017 DOI: 10.1038/ncomms13542 OPEN Defining functional interactions during biogenesis of epithelial junctions J.C. Erasmus1,*, S. Bruche1,*,w, L. Pizarro1,2,*, N. Maimari1,3,*, T. Poggioli1,w, C. Tomlinson4,J.Lees5, I. Zalivina1,w, A. Wheeler1,w, A. Alberts6, A. Russo2 & V.M.M. Braga1 In spite of extensive recent progress, a comprehensive understanding of how actin cytoskeleton remodelling supports stable junctions remains to be established. Here we design a platform that integrates actin functions with optimized phenotypic clustering and identify new cytoskeletal proteins, their functional hierarchy and pathways that modulate E-cadherin adhesion. Depletion of EEF1A, an actin bundling protein, increases E-cadherin levels at junctions without a corresponding reinforcement of cell–cell contacts. This unexpected result reflects a more dynamic and mobile junctional actin in EEF1A-depleted cells. A partner for EEF1A in cadherin contact maintenance is the formin DIAPH2, which interacts with EEF1A. In contrast, depletion of either the endocytic regulator TRIP10 or the Rho GTPase activator VAV2 reduces E-cadherin levels at junctions. TRIP10 binds to and requires VAV2 function for its junctional localization. Overall, we present new conceptual insights on junction stabilization, which integrate known and novel pathways with impact for epithelial morphogenesis, homeostasis and diseases. 1 National Heart and Lung Institute, Faculty of Medicine, Imperial College London, London SW7 2AZ, UK. 2 Computing Department, Imperial College London, London SW7 2AZ, UK. 3 Bioengineering Department, Faculty of Engineering, Imperial College London, London SW7 2AZ, UK. 4 Department of Surgery & Cancer, Faculty of Medicine, Imperial College London, London SW7 2AZ, UK. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -
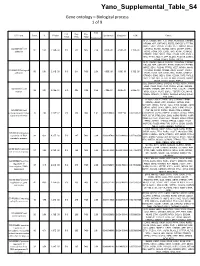
Supplemental Tables4.Pdf
Yano_Supplemental_Table_S4 Gene ontology – Biological process 1 of 9 Fold List Pop Pop GO Term Count % PValue Bonferroni Benjamini FDR Genes Total Hits Total Enrichment DLC1, CADM1, NELL2, CLSTN1, PCDHGA8, CTNNB1, NRCAM, APP, CNTNAP2, FERT2, RAPGEF1, PTPRM, MPDZ, SDK1, PCDH9, PTPRS, VEZT, NRXN1, MYH9, GO:0007155~cell CTNNA2, NCAM1, NCAM2, DDR1, LSAMP, CNTN1, 50 5.61 2.14E-08 510 311 7436 2.34 4.50E-05 4.50E-05 3.70E-05 adhesion ROR2, VCAN, DST, LIMS1, TNC, ASTN1, CTNND2, CTNND1, CDH2, NEO1, CDH4, CD24A, FAT3, PVRL3, TRO, TTYH1, MLLT4, LPP, NLGN1, PCDH19, LAMA1, ITGA9, CDH13, CDON, PSPC1 DLC1, CADM1, NELL2, CLSTN1, PCDHGA8, CTNNB1, NRCAM, APP, CNTNAP2, FERT2, RAPGEF1, PTPRM, MPDZ, SDK1, PCDH9, PTPRS, VEZT, NRXN1, MYH9, GO:0022610~biological CTNNA2, NCAM1, NCAM2, DDR1, LSAMP, CNTN1, 50 5.61 2.14E-08 510 311 7436 2.34 4.50E-05 4.50E-05 3.70E-05 adhesion ROR2, VCAN, DST, LIMS1, TNC, ASTN1, CTNND2, CTNND1, CDH2, NEO1, CDH4, CD24A, FAT3, PVRL3, TRO, TTYH1, MLLT4, LPP, NLGN1, PCDH19, LAMA1, ITGA9, CDH13, CDON, PSPC1 DCC, ENAH, PLXNA2, CAPZA2, ATP5B, ASTN1, PAX6, ZEB2, CDH2, CDH4, GLI3, CD24A, EPHB1, NRCAM, GO:0006928~cell CTTNBP2, EDNRB, APP, PTK2, ETV1, CLASP2, STRBP, 36 4.04 3.46E-07 510 205 7436 2.56 7.28E-04 3.64E-04 5.98E-04 motion NRG1, DCLK1, PLAT, SGPL1, TGFBR1, EVL, MYH9, YWHAE, NCKAP1, CTNNA2, SEMA6A, EPHA4, NDEL1, FYN, LRP6 PLXNA2, ADCY5, PAX6, GLI3, CTNNB1, LPHN2, EDNRB, LPHN3, APP, CSNK2A1, GPR45, NRG1, RAPGEF1, WWOX, SGPL1, TLE4, SPEN, NCAM1, DDR1, GRB10, GRM3, GNAQ, HIPK1, GNB1, HIPK2, PYGO1, GO:0007166~cell RNF138, ROR2, CNTN1, -

Serum Albumin OS=Homo Sapiens
Protein Name Cluster of Glial fibrillary acidic protein OS=Homo sapiens GN=GFAP PE=1 SV=1 (P14136) Serum albumin OS=Homo sapiens GN=ALB PE=1 SV=2 Cluster of Isoform 3 of Plectin OS=Homo sapiens GN=PLEC (Q15149-3) Cluster of Hemoglobin subunit beta OS=Homo sapiens GN=HBB PE=1 SV=2 (P68871) Vimentin OS=Homo sapiens GN=VIM PE=1 SV=4 Cluster of Tubulin beta-3 chain OS=Homo sapiens GN=TUBB3 PE=1 SV=2 (Q13509) Cluster of Actin, cytoplasmic 1 OS=Homo sapiens GN=ACTB PE=1 SV=1 (P60709) Cluster of Tubulin alpha-1B chain OS=Homo sapiens GN=TUBA1B PE=1 SV=1 (P68363) Cluster of Isoform 2 of Spectrin alpha chain, non-erythrocytic 1 OS=Homo sapiens GN=SPTAN1 (Q13813-2) Hemoglobin subunit alpha OS=Homo sapiens GN=HBA1 PE=1 SV=2 Cluster of Spectrin beta chain, non-erythrocytic 1 OS=Homo sapiens GN=SPTBN1 PE=1 SV=2 (Q01082) Cluster of Pyruvate kinase isozymes M1/M2 OS=Homo sapiens GN=PKM PE=1 SV=4 (P14618) Glyceraldehyde-3-phosphate dehydrogenase OS=Homo sapiens GN=GAPDH PE=1 SV=3 Clathrin heavy chain 1 OS=Homo sapiens GN=CLTC PE=1 SV=5 Filamin-A OS=Homo sapiens GN=FLNA PE=1 SV=4 Cytoplasmic dynein 1 heavy chain 1 OS=Homo sapiens GN=DYNC1H1 PE=1 SV=5 Cluster of ATPase, Na+/K+ transporting, alpha 2 (+) polypeptide OS=Homo sapiens GN=ATP1A2 PE=3 SV=1 (B1AKY9) Fibrinogen beta chain OS=Homo sapiens GN=FGB PE=1 SV=2 Fibrinogen alpha chain OS=Homo sapiens GN=FGA PE=1 SV=2 Dihydropyrimidinase-related protein 2 OS=Homo sapiens GN=DPYSL2 PE=1 SV=1 Cluster of Alpha-actinin-1 OS=Homo sapiens GN=ACTN1 PE=1 SV=2 (P12814) 60 kDa heat shock protein, mitochondrial OS=Homo -

4 Mechanics of the Cytoskeleton
4 Mechanics of the cytoskeleton 4.1 Motivation In the previous section, we have seen how biopolymers dynamically assemble and dis- assemble during polymerization. We have discussed the individual mechanical prop- erties such as Young’s modulus E, the axial stiffness EA, the bending stiffness EI, and the persistence length A for individual filaments. In particular, have talked about actin filaments, intermediate filaments, and microtubules. Now, assuming we know the me- chanical properties of the individual filaments, what does that actually tell us about the assembly of filaments that we find in the cell? Or, to put it differently, if we knew elements of the cytoskeleton microtubules intermediate filaments actin filaments Figure 4.1: The cytoskeleton provides structural stability and is responsible for forces during cell loco- motion. Microtubules are thick hollow cylinders reaching out from the nucleus to the membrane, inter- mediate filaments can be found anywhere in the cytosol, and actin filaments are usually concentrated close to the cell membrane. the structural arrangement of filaments, could we then predict the stiffness of the over- all assembly? How does the filament microstructure affect cytoskeletal properties? Or, more precisely, how can we calculate the macroscopic network properties from the in- dividual microscopic filament properties? In mechanics, the derivation of macroscopic parameters based on microscopic considerations is referred to as homogenization. In this chapter, we illustrate the homogenization by means of three different examples, the fiber bundle model for filopodia, the network model for red blood cell membranes, and the tensegrity model for generic cell structures. 4.2 Fiber bundle model for filopodia Filopodia are thin dynamic cytoplasmic projections composed of tight bundles of long actin filaments extending from the leading edge of migrating cells. -

Of Polarity Ups and Downs of Guided Vessel Sprouting
Ups and Downs of Guided Vessel Sprouting: The Role of Polarity Christina Y. Lee and Victoria L. Bautch Physiology 26:326-333, 2011. doi:10.1152/physiol.00018.2011 You might find this additional info useful... This article cites 82 articles, 38 of which can be accessed free at: /content/26/5/326.full.html#ref-list-1 This article has been cited by 2 other HighWire hosted articles Rasip1 regulates vertebrate vascular endothelial junction stability through Epac1-Rap1 signaling Christopher W. Wilson, Leon H. Parker, Christopher J. Hall, Tanya Smyczek, Judy Mak, Ailey Crow, George Posthuma, Ann De Mazière, Meredith Sagolla, Cecile Chalouni, Philip Vitorino, Merone Roose-Girma, Søren Warming, Judith Klumperman, Philip S. Crosier and Weilan Ye Blood, November 21, 2013; 122 (22): 3678-3690. [Abstract] [Full Text] [PDF] Cas and NEDD9 Contribute to Tumor Progression through Dynamic Regulation of the Cytoskeleton Michael S. Guerrero, J. Thomas Parsons and Amy H. Bouton Genes & Cancer, May , 2012; 3 (5-6): 371-381. [Abstract] [Full Text] [PDF] Downloaded from Updated information and services including high resolution figures, can be found at: /content/26/5/326.full.html Additional material and information about Physiology can be found at: http://www.the-aps.org/publications/physiol on August 25, 2014 This information is current as of August 25, 2014. Physiology (formerly published as News in Physiological Science) publishes brief review articles on major physiological developments. It is published bimonthly in February, April, June, August, October, and December by the American Physiological Society, 9650 Rockville Pike, Bethesda MD 20814-3991. Copyright © 2011 by the American Physiological Society. -

MYO5A Rabbit Pab
Leader in Biomolecular Solutions for Life Science MYO5A Rabbit pAb Catalog No.: A9830 Basic Information Background Catalog No. This gene is one of three myosin V heavy-chain genes, belonging to the myosin gene A9830 superfamily. Myosin V is a class of actin-based motor proteins involved in cytoplasmic vesicle transport and anchorage, spindle-pole alignment and mRNA translocation. The Observed MW protein encoded by this gene is abundant in melanocytes and nerve cells. Mutations in 240kDa this gene cause Griscelli syndrome type-1 (GS1), Griscelli syndrome type-3 (GS3) and neuroectodermal melanolysosomal disease, or Elejalde disease. Multiple alternatively Calculated MW spliced transcript variants encoding different isoforms have been reported, but the full- 212kDa/215kDa/218kDa length nature of some variants has not been determined. Category Primary antibody Applications WB,IHC,IF Cross-Reactivity Human, Mouse, Rat Recommended Dilutions Immunogen Information WB 1:500 - 1:2000 Gene ID Swiss Prot 4644 Q9Y4I1 IHC 1:50 - 1:100 Immunogen 1:50 - 1:100 IF Recombinant fusion protein containing a sequence corresponding to amino acids 950-1150 of human MYO5A (NP_000250.3). Synonyms MYO5A;GS1;MYH12;MYO5;MYR12;myosin VA Contact Product Information www.abclonal.com Source Isotype Purification Rabbit IgG Affinity purification Storage Store at -20℃. Avoid freeze / thaw cycles. Buffer: PBS with 0.02% sodium azide,50% glycerol,pH7.3. Validation Data Western blot analysis of extracts of various cell lines, using MYO5A antibody (A9830) at 1:1000 dilution. Secondary antibody: HRP Goat Anti-Rabbit IgG (H+L) (AS014) at 1:10000 dilution. Lysates/proteins: 25ug per lane. Blocking buffer: 3% nonfat dry milk in TBST. -

Bone Morphogenetic Protein Signaling Suppresses Wound-Induced Skin Repair by Inhibiting Keratinocyte Proliferation and Migration Christopher J
ORIGINAL ARTICLE Bone Morphogenetic Protein Signaling Suppresses Wound-Induced Skin Repair by Inhibiting Keratinocyte Proliferation and Migration Christopher J. Lewis1,2,AndreiN.Mardaryev1, Krzysztof Poterlowicz1, Tatyana Y. Sharova3, Ahmar Aziz3, David T. Sharpe2, Natalia V. Botchkareva1 and Andrey A. Sharov3 Bone morphogenetic protein (BMP) signaling plays a key role in the control of skin development and postnatal remodeling by regulating keratinocyte proliferation, differentiation, and apoptosis. To study the role of BMPs in wound-induced epidermal repair, we used transgenic mice overexpressing the BMP downstream component Smad1 under the control of a K14 promoter as an in vivo model, as well as ex vivo and in vitro assays. K14- caSmad1 mice (transgenic mice overexpressing a constitutively active form of Smad1 under K14 promoter) exhibited retarded wound healing associated with significant inhibition of proliferation and increased apoptosis in healing wound epithelium. Furthermore, microarray and quantitative real-time reverse-transcriptase–PCR (qRT– PCR) analyses revealed decreased expression of a number of cytoskeletal/cell motility–associated genes including wound-associated keratins (Krt16, Krt17)andMyosin VA (Myo5a), in the epidermis of K14-caSmad1 mice versus wild-type (WT) controls during wound healing. BMP treatment significantly inhibited keratinocyte migration ex vivo, and primary keratinocytes of K14-caSmad1 mice showed retarded migration compared with WT controls. Finally, small interfering RNA (siRNA)–mediated silencing of BMPR-1B in primary mouse keratinocytes accelerated cell migration and was associated with increased expression of Krt16, Krt17, and Myo5a compared with controls. Thus, this study demonstrates that BMPs inhibit keratinocyte proliferation, cytoskeletal organization, and migration in regenerating skin epithelium during wound healing, and raises a possibility for using BMP antagonists for the management of chronic wounds. -

Dynamics of Thin Filopodia During Sea Urchin Gastrulation
Development 121, 2501-2511 (1995) 2501 Printed in Great Britain © The Company of Biologists Limited 1995 Dynamics of thin filopodia during sea urchin gastrulation Jeffrey Miller1, Scott E. Fraser2 and David McClay1,* 1Developmental, Cell and Molecular Biology, Duke University, SRC, Box 91000, Durham, NC 27708, USA 2Division of Biology, Beckman Institute (139-74), California Institute of Technology, Pasadena CA 91125, USA *Author for correspondence: e-mail [email protected] SUMMARY At gastrulation in the sea urchin embryo, a dramatic involvement in cell-cell interactions associated with rearrangement of cells establishes the three germ layers of signaling and patterning at gastrulation. Nickel-treatment, the organism. Experiments have revealed a number of cell which is known to create a patterning defect in skeleto- interactions at this stage that transfer patterning informa- genesis due to alterations in the ectoderm, alters the normal tion from cell to cell. Of particular significance, primary position-dependent differences in the thin filopodia. The mesenchyme cells, which are responsible for production of effect is present in recombinant embryos in which the the embryonic skeleton, have been shown to obtain ectoderm alone was treated with nickel, and is absent in extensive positional information from the embryonic recombinant embryos in which only the primary mes- ectoderm. In the present study, high resolution Nomarski enchyme cells were treated, suggesting that the filopodial imaging reveals the presence of very thin filopodia (0.2-0.4 length is substratum dependent rather than being primary µm in diameter) extending from primary mesenchyme cells mesenchyme cell autonomous. The thin filopodia provide a as well as from ectodermal and secondary mesenchyme means by which cells can contact others several cell cells. -

Strong Correlation Between Air-Liquid Interface Cultures and in Vivo Transcriptomics of Nasal Brush Biopsy
Am J Physiol Lung Cell Mol Physiol 318: L1056–L1062, 2020. First published April 1, 2020; doi:10.1152/ajplung.00050.2020. RAPID REPORT Translational Physiology Strong correlation between air-liquid interface cultures and in vivo transcriptomics of nasal brush biopsy X Baishakhi Ghosh,1 Bongsoo Park,1 Debarshi Bhowmik,2 Kristine Nishida,3 Molly Lauver,3 Nirupama Putcha,3 Peisong Gao,4 Murugappan Ramanathan, Jr.,5 Nadia Hansel,3 Shyam Biswal,1 and Venkataramana K. Sidhaye1,3 1Department of Environmental Health and Engineering, Johns Hopkins Bloomberg School of Public Health, Baltimore, Maryland; 2Department of Biology, Johns Hopkins University, Baltimore, Maryland; 3Department of Pulmonary and Critical Care Medicine, Johns Hopkins School of Medicine, Baltimore, Maryland; 4Division of Allergy and Clinical Immunology, Johns Hopkins School of Medicine, Baltimore, Maryland; and 5Department of Otolaryngology-Head and Neck Surgery, Johns Hopkins School of Medicine, Baltimore, Maryland Submitted 18 February 2020; accepted in final form 24 March 2020 Ghosh B, Park B, Bhowmik D, Nishida K, Lauver M, Putcha N, RNA sequencing to determine the correlation between ALI- Gao P, Ramanathan M Jr, Hansel N, Biswal S, Sidhaye VK. cultured epithelial cells and epithelial cells obtained from nasal Strong correlation between air-liquid interface cultures and in vivo brushing and identify differences that may arise as a result of transcriptomics of nasal brush biopsy. Am J Physiol Lung Cell Mol redifferentiation. Physiol 318: L1056–L1062, 2020. First published April 1, 2020; doi:10.1152/ajplung.00050.2020.—Air-liquid interface (ALI) cultures METHODS are ex vivo models that are used extensively to study the epithelium of patients with chronic respiratory diseases. -

002 Sempozyum1 5 SON.Qxd
Abstracts www.anatomy.org.tr doi:10.2399/ana.11.001s Abstracts for the Joint Meeting of Anatomical Societies, 19-22 May 2011, Bursa, Turkey Anatomy 2011; 5 Suppl: S1-S171, © 2011 TSACA Opening Lecture New genoarchitectonic viewpoints on the developing hypothalamus Puelles L effects suggests that, rather than being a diencephalic floor ele- ment, the hypothalamus is best understood as a transverse region Department of Human Anatomy, Faculty of lying ventral to the telencephalon and rostral to the dien- Medicine, University of Murcia, Murcia, Spain cephalon; the latter separates it from the midbrain. A number of gene expression patterns observed in the developing forebrain, part of the emergent genoarchitectonic neuroanatomy, have The anatomic concept of the hypothalamus changed consider- revealed the true topologic position of the hypothalamus, as well ably since its earliest definition. Tridimensional reconstructions, as the nature of its fundamental subdivisions. There are interest- experiments and many staining methods have expanded consid- ing parallelisms with genoarchitectonic patterns in the dien- erably the number of anatomical details recognized in this terri- cephalon and midbrain. In all these cases continuous longitudi- tory, probably one of the most complex in the brain. For a long nal domains can be distinguished, as well as a number of antero- time the predominant anatomic view has interpreted the hypo- posterior (transverse) neuromeric units of the neural wall. The thalamus as a longitudinal column at the floor of the dien- hypothalamus has been newly recognized to have two antero- cephalon, connected rostrally with the telencephalon and cau- posterior neuromeric subdivisions, named terminal and pedun- dally with the midbrain. -

Molecular Signatures Differentiate Immune States in Type 1 Diabetes Families
Page 1 of 65 Diabetes Molecular signatures differentiate immune states in Type 1 diabetes families Yi-Guang Chen1, Susanne M. Cabrera1, Shuang Jia1, Mary L. Kaldunski1, Joanna Kramer1, Sami Cheong2, Rhonda Geoffrey1, Mark F. Roethle1, Jeffrey E. Woodliff3, Carla J. Greenbaum4, Xujing Wang5, and Martin J. Hessner1 1The Max McGee National Research Center for Juvenile Diabetes, Children's Research Institute of Children's Hospital of Wisconsin, and Department of Pediatrics at the Medical College of Wisconsin Milwaukee, WI 53226, USA. 2The Department of Mathematical Sciences, University of Wisconsin-Milwaukee, Milwaukee, WI 53211, USA. 3Flow Cytometry & Cell Separation Facility, Bindley Bioscience Center, Purdue University, West Lafayette, IN 47907, USA. 4Diabetes Research Program, Benaroya Research Institute, Seattle, WA, 98101, USA. 5Systems Biology Center, the National Heart, Lung, and Blood Institute, the National Institutes of Health, Bethesda, MD 20824, USA. Corresponding author: Martin J. Hessner, Ph.D., The Department of Pediatrics, The Medical College of Wisconsin, Milwaukee, WI 53226, USA Tel: 011-1-414-955-4496; Fax: 011-1-414-955-6663; E-mail: [email protected]. Running title: Innate Inflammation in T1D Families Word count: 3999 Number of Tables: 1 Number of Figures: 7 1 For Peer Review Only Diabetes Publish Ahead of Print, published online April 23, 2014 Diabetes Page 2 of 65 ABSTRACT Mechanisms associated with Type 1 diabetes (T1D) development remain incompletely defined. Employing a sensitive array-based bioassay where patient plasma is used to induce transcriptional responses in healthy leukocytes, we previously reported disease-specific, partially IL-1 dependent, signatures associated with pre and recent onset (RO) T1D relative to unrelated healthy controls (uHC).