Coracoid Fractures and Dislocations
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
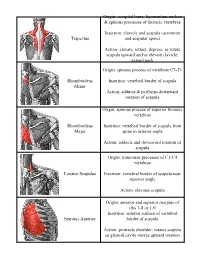
Trapezius Origin: Occipital Bone, Ligamentum Nuchae & Spinous Processes of Thoracic Vertebrae Insertion: Clavicle and Scapul
Origin: occipital bone, ligamentum nuchae & spinous processes of thoracic vertebrae Insertion: clavicle and scapula (acromion Trapezius and scapular spine) Action: elevate, retract, depress, or rotate scapula upward and/or elevate clavicle; extend neck Origin: spinous process of vertebrae C7-T1 Rhomboideus Insertion: vertebral border of scapula Minor Action: adducts & performs downward rotation of scapula Origin: spinous process of superior thoracic vertebrae Rhomboideus Insertion: vertebral border of scapula from Major spine to inferior angle Action: adducts and downward rotation of scapula Origin: transverse precesses of C1-C4 vertebrae Levator Scapulae Insertion: vertebral border of scapula near superior angle Action: elevates scapula Origin: anterior and superior margins of ribs 1-8 or 1-9 Insertion: anterior surface of vertebral Serratus Anterior border of scapula Action: protracts shoulder: rotates scapula so glenoid cavity moves upward rotation Origin: anterior surfaces and superior margins of ribs 3-5 Insertion: coracoid process of scapula Pectoralis Minor Action: depresses & protracts shoulder, rotates scapula (glenoid cavity rotates downward), elevates ribs Origin: supraspinous fossa of scapula Supraspinatus Insertion: greater tuberacle of humerus Action: abduction at the shoulder Origin: infraspinous fossa of scapula Infraspinatus Insertion: greater tubercle of humerus Action: lateral rotation at shoulder Origin: clavicle and scapula (acromion and adjacent scapular spine) Insertion: deltoid tuberosity of humerus Deltoid Action: -

Coracoid Process Anatomy: a Cadaveric Study of Surgically Relevant Structures Jorge Chahla, M.D., Ph.D., Daniel Cole Marchetti, B.A., Gilbert Moatshe, M.D., Márcio B
Quantitative Assessment of the Coracoacromial and the Coracoclavicular Ligaments With 3-Dimensional Mapping of the Coracoid Process Anatomy: A Cadaveric Study of Surgically Relevant Structures Jorge Chahla, M.D., Ph.D., Daniel Cole Marchetti, B.A., Gilbert Moatshe, M.D., Márcio B. Ferrari, M.D., George Sanchez, B.S., Alex W. Brady, M.Sc., Jonas Pogorzelski, M.D., M.H.B.A., George F. Lebus, M.D., Peter J. Millett, M.D., M.Sc., Robert F. LaPrade, M.D., Ph.D., and CAPT Matthew T. Provencher, M.D., M.C., U.S.N.R. Purpose: To perform a quantitative anatomic evaluation of the (1) coracoid process, specifically the attachment sites of the conjoint tendon, the pectoralis minor, the coracoacromial ligament (CAL), and the coracoclavicular (CC) ligaments in relation to pertinent osseous and soft tissue landmarks; (2) CC ligaments’ attachments on the clavicle; and (3) CAL attachment on the acromion in relation to surgically relevant anatomic landmarks to assist in planning of the Latarjet procedure, acromioclavicular (AC) joint reconstructions, and CAL resection distances avoiding iatrogenic injury to sur- rounding structures. Methods: Ten nonpaired fresh-frozen human cadaveric shoulders (mean age 52 years, range 33- 64 years) were included in this study. A 3-dimensional coordinate measuring device was used to quantify the location of pertinent bony landmarks and soft tissue attachment areas. The ligament and tendon attachment perimeters and center points on the coracoid, clavicle, and acromion were identified and subsequently dissected off the bone. Coordinates of points along the perimeters of attachment sites were used to calculate areas, whereas coordinates of center points were used to determine distances between surgically relevant attachment sites and pertinent bony landmarks. -

Reconstructing Pectoral Appendicular Muscle Anatomy in Fossil Fish and Tetrapods Over the Fins-To-Limbs Transition
Biol. Rev. (2017), pp. 000–000. 1 doi: 10.1111/brv.12386 Reconstructing pectoral appendicular muscle anatomy in fossil fish and tetrapods over the fins-to-limbs transition Julia L. Molnar1,∗ , Rui Diogo2, John R. Hutchinson3 and Stephanie E. Pierce4 1Department of Anatomy, New York Institute of Technology College of Osteopathic Medicine, Northern Boulevard, Old Westbury, NY, U.S.A. 2Department of Anatomy, Howard University College of Medicine, 520 W St. NW, Numa Adams Building, Washington, DC 20059, U.S.A. 3Structure and Motion Lab, Royal Veterinary College, Hawkshead Lane, Hatfield, Hertfordshire AL9 7TA, UK 4Museum of Comparative Zoology and Department of Organismic and Evolutionary Biology, Harvard University, 26 Oxford Street, Cambridge, MA 02138, U.S.A. ABSTRACT The question of how tetrapod limbs evolved from fins is one of the great puzzles of evolutionary biology. While palaeontologists, developmental biologists, and geneticists have made great strides in explaining the origin and early evolution of limb skeletal structures, that of the muscles remains largely unknown. The main reason is the lack of consensus about appendicular muscle homology between the closest living relatives of early tetrapods: lobe-finned fish and crown tetrapods. In the light of a recent study of these homologies, we re-examined osteological correlates of muscle attachment in the pectoral girdle, humerus, radius, and ulna of early tetrapods and their close relatives. Twenty-nine extinct and six extant sarcopterygians were included in a meta-analysis using information from the literature and from original specimens, when possible. We analysed these osteological correlates using parsimony-based character optimization in order to reconstruct muscle anatomy in ancestral lobe-finned fish, tetrapodomorph fish, stem tetrapods, and crown tetrapods. -

Breathing and Locomotion in Birds
Breathing and locomotion in birds. A thesis submitted to the University of Manchester for the degree of Doctor of Philosophy in the Faculty of Life Sciences. 2010 Peter George Tickle Contents Abstract 4 Declaration 5 Copyright Statement 6 Author Information 7 Acknowledgements 9 Organisation of this PhD thesis 10 Chapter 1 General Introduction 13 1. Introduction 14 1.1 The Avian Respiratory System 14 1.1.1 Structure of the lung and air sacs 16 1.1.2 Airflow in the avian respiratory system 21 1.1.3 The avian aspiration pump 25 1.2 The uncinate processes in birds 29 1.2.1 Uncinate process morphology and biomechanics 32 1.3 Constraints on breathing in birds 33 1.3.1 Development 33 1.3.2 Locomotion 35 1.3.2.1 The appendicular skeleton 35 1.3.2.2 Overcoming the trade-off between breathing 36 and locomotion 1.3.2.3 Energetics of locomotion in birds 38 1.4 Evolution of the ventilatory pump in birds 41 1.5 Overview and Thesis Aims 42 2 Chapter 2 Functional significance of the uncinate processes in birds. 44 Chapter 3 Ontogenetic development of the uncinate processes in the 45 domestic turkey (Meleagris gallopavo). Chapter 4 Uncinate process length in birds scales with resting metabolic rate. 46 Chapter 5 Load carrying during locomotion in the barnacle goose (Branta 47 leucopsis): The effect of load placement and size. Chapter 6 A continuum in ventilatory mechanics from early theropods to 48 extant birds. Chapter 7 General Discussion 49 References 64 3 Abstract of a thesis by Peter George Tickle submitted to the University of Manchester for the degree of PhD in the Faculty of Life Sciences and entitled ‘Breathing and Locomotion in Birds’. -

Some Notes on the Diverse Brachiosaurid Sauropods of the Late Jurassic of North America, Europe and Africa
Some Notes on the Diverse Brachiosaurid Sauropods of the Late Jurassic of North America, Europe and Africa GREGORY S. PAUL 3109 N. Calvert St. Baltimore, Maryland 21218 USA [email protected] June 2012 Abstract: The unusual placement of the parapophyses on elongotated peduncles further distinguishes Brachiosaurus from other brachiosaurid genera including Giraffatitan. Late Jurassic brachiosaurid taxonomy is probably more complex than has been realized, in part because of evolution over the considerable periods of times recorded in the Morrison and Tendaguru formations. The presence of reduced tails on late Jurassic brachiosaurids is confirmed. INTRODUCTION It was long assumed that the brachiosaurid material found in the Late Jurassic Morrison, Tendaguru, and Lourinha Formations from three continents belonged to the genus Brachiosaurus (as per Janensch 1950, 1961, Lapparent and Zbyszewski 1957, Jensen 1987) until Paul (1988) noted significant differences between the type species B. altithorax (Fig. 1A) from North America and the African B. brancai (Fig. 1B) indicated at least a subgeneric separation, and Taylor (2009) formally separated the sauropods, leaving much of the Tendaguru brachiosaurids material titled Giraffatitan brancai; the generic separation has been tentatively questioned (Chure et al. 2010). In addition European B. alatalaiensis has been retitled Lusotitan alatalaiensis (Antunes and Mateus 2003), and a dwarf brachiosaur from the European Mittlere Kimmeridge-Stufe deposits has been designated Europasaurus holgeri (Sander et al. 2006). Institutional Abbreviations: AMNH, American Museum of Natural History, New York; BYU, Brigham Young University, Provo; FMNH, Field Museum of Natural History, Chicago; HMN, Humboldt Museum fur Naturkunde, Berlin; NHMUK, Natural History Museum, London; USNM, United States Natural History Museum, Washington DC. -

Rehabilitation Guidelines for Latarjet/Coracoid Process Transfer Josef K
Rehabilitation Guidelines for Latarjet/Coracoid Process Transfer Josef K. Eichinger, MD Shoulder instability may be caused from congenital deformity, recurrent overuse activity, and/or traumatic dislocation. Surgical stabilization of the glenohumeral joint is indicated after conservative treatment fails and recurrent instability/subluxation continues. A number of different surgical procedures may be indicated in this situation, often divided into soft tissue or bony procedures. Shoulder Instability – Soft Tissue: Surgical reconstruction targeting the glenohumeral joint’s soft tissues for shoulder instability, typically involves labral repairs, the most common being the Bankart repair. A Bankart lesion typically occurs from an anterior-inferior dislocation of the humerus, tearing the labrum from it’s attachment to the glenoid, thereby detaching the inferior gleno-humeral ligament (IGHL). Surgical management of this revolves around labral repair to reattach the IGHL under appropriate tension. This may be accomplished either arthroscopically or through an open approach.1 Most traumatic glenohumeral dislocations may not only cause a Bankart lesion, but may create impression fractures in the postero-superior humeral head termed Hill-Sachs lesions.2 An adverse effect from this procedure includes suturing the capsule too tightly, causing a shortening of the capsule, and thus decreasing the external rotation allowed at the glenohumeral joint. Other complications are extremely rare, but may include axillary nerve damage, subscapularis rupture (seen only in open repairs performed with subscapularis detachment and repair), and recurrent instability. If there is bony deficiency in the glenoid, which represents 20% or more of the antero-inferior glenoid, it is biomechanically impossible to restore the same stability and is therefore more prone to recurrent instability and failure. -

Basic Avian Anatomy
Basic Avian Anatomy Peter S. Sakas DVM, MS Niles Animal Hospital and Bird Medical Center 7278 N. Milwaukee Ave. Niles, IL 60714 (847)-647-9325 FAX (847)-647-8498 Introduction Everyone is familiar with the anatomy of mammals and may also have some knowledge of a few avian anatomical characteristics. The purpose of this discussion is to provide a deeper insight into avian anatomy and provide some comparisons to mammalian features. An understanding of avian anatomy is essential for avian practitioners. Sources of information for this discussion include the fine work of Dr. Howard Evans and Dr. Robert Clipsham. Feathers Feathers are unique to birds. Birds grow feathers in and around eight well- defined feather tracts or pterylae; they are not haphazardly arranged. Feathers compromise from 10-20% of a bird’s body weight. Each feather can be raised by a separate skin muscle (‘raising their hackles’ or fanning tail).Feathers are outgrowths of the feather follicles of the skin and are the counterpart to hairs and hair follicles in mammals. Feathers provide many functions for birds, attracts mate or deceives predator, heat control, flight, aerodynamic streamlining and water buoyancy. Feathers are not really “bird hairs” but are probably modified scales passed down from their reptilian ancestors. Feathers can be grouped into three categories: 1) Contour feathers or penna – These feathers cover the body, wings and tail, and are the feathers most obviously visible on the bird. 2) Down feathers or plumules – These tiny, soft down feathers are found associated with contour feathers and/or the spaces between them. 3) Tufted bristle feathers or filoplumes- Feathers which are modified and appear as ‘eyelashes and nose hairs.’ Contour Feathers The contour feather consists of a shaft with a vane. -

DEINOCHEIRIDAE, a NEW FAMILY of THEROPOD DINOSAURS (Plates I-V)
HALSZKA OSMQLSKA & EWA RONIEWICZ DEINOCHEIRIDAE, A NEW FAMILY OF THEROPOD DINOSAURS (plates I-V) Abstract,- A new carnosaurian dinosaur, Deinochelrus mirificus n, gen., n, sp., is described from the Upper Cretaceous of the Gobi Desert. The material consists of a shoulder girdle, fore limbs and fragmentary ribs. The fore limbs are about 240 cm long and tridactyl, all the digits being equally developed and terminating in powerful claws. Carnosaurian fore limbs of such large size have not been previously reported. The gigantic size of the fore limbs appears ample justification for establishment of a new family, Deinocheiridae, within Carnosauria. INTRODUCTION During the course of the Polish-Mongolian Palaeontological Expedition of 1965, an unusual, incomplete skeleton of a gigantic carnosaurian dinosaur was recovered. This skeleton was found by Prof. Z. KIELAN-]AWOROWSKA at Altan Ula III locality, Nemegt Basin, Gobi Desert, Mongolian People's Republic, in the Upper Cretaceous sandstones, designated by GRA DZINSKI et a/..(l968/69) as Upper Nemegt Beds; its exact location IS given by GRADZINSKI et al. (I. c., Text-fig. 6; see also KIELAN-]AWOROWSKA & DOVCHIN, 1968/69, p. 24, PI. 4, Fig. 1). The specimen in question, consisting ofa shoulder girdle, fore limbs and other incomplete bone fragments, is described in the present paper as Deinocheirus mirificus n. gen., n. sp. Because of its extreme dimensions and the unreduced tridactyl structure of the manus, the new genus cannot be assigned to any known family of theropod dinosaurs. Consequently, a new family Deinocheiridae is erected for it, within the superfamily Megalosauroidea Walker, faute de mieux, infraorder Carnosauria (non Megalosauroidea sensu MALEYEV, 1968). -

A Nearly Complete Ornithocheirid Pterosaur from the Aptian (Early Cretaceous) Crato Formation of NE Brazil
A nearly complete ornithocheirid pterosaur from the Aptian (Early Cretaceous) Crato Formation of NE Brazil ROSS A. ELGIN and EBERHARD FREY Elgin, R.A. and Frey, E. 2012. A nearly complete ornithocheirid pterosaur from the Aptian (Early Cretaceous) Crato Formation of NE Brazil. Acta Palaeontologica Polonica 57 (1): 101–110. A partial ornithocheirid, representing a rare example of a pterosaurian body fossil from the Nova Olinda Member of the Crato Formation, NE Brazil, is described from the collections of the State Museum of Natural History, Karlsruhe. While similar in preservation and taphonomy to Arthurdactylus conandoylei, it is distinguished by slight differences in biomet− ric ratios, but the absence of a skull prevents closer identification. Mostly complete body fossils belonging to ornitho− cheiroid pterosaurs appear to be relatively more abundant in the younger Romualdo Member of the Santana Formation, making the described specimen one of only two well documented ornithocheiroids known from the Nova Olinda Lagerstätte. Key words: Ornithocheiroidea, pterosaur, taphonomy, Aptian, Cretaceous, Crato Formation, Brazil. Ross A. Elgin [[email protected]] and Eberhard Frey [[email protected]], Staatliches Museum für Naturkunde Karlsruhe (SMNK), Abteilung Geologie, Erbprinzenstraße 13, 76133 Karlsruhe, Germany. Received 4 August 2010, accepted 17 March 2011, available online 31 March 2011. Introduction A new specimen from the Nova Olinda Member in the collections of the State Museum of Natural History, Karls− The Araripe Basin of NE Brazil contains two Early Creta− ruhe (SMNK PAL 3854), is described here, representing the ceous Lagerstätten that are world renowned for their excep− rare occurrence of a largely complete ornithocheirid ptero− tional preservation of insects and vertebrate fossils (Unwin saur from this locality. -

Scapula Fractures
Ministry of Defence Synopsis of Causation Scapula Fractures Author: Mr Darren P Forward, Queen’s Medical Centre, Nottingham Professor W Angus Wallace, Queen’s Medical Centre, Nottingham Validator: Mr Sheo Tibrewal, Queen Elizabeth Hospital London September 2008 Disclaimer This synopsis has been completed by medical practitioners. It is based on a literature search at the standard of a textbook of medicine and generalist review articles. It is not intended to be a meta-analysis of the literature on the condition specified. Every effort has been taken to ensure that the information contained in the synopsis is accurate and consistent with current knowledge and practice and to do this the synopsis has been subject to an external validation process by consultants in a relevant specialty nominated by the Royal Society of Medicine. The Ministry of Defence accepts full responsibility for the contents of this synopsis, and for any claims for loss, damage or injury arising from the use of this synopsis by the Ministry of Defence. 2 1. Definition 1.1. A scapular fracture is a break in the continuity of the cortex of the scapula bone. Scapular fractures are uncommon. They represent 1%1 of all fractures and 3%1 of shoulder-girdle injuries. 1.2. To aid prognosis, they can be classified into 3 groups.2 1.2.1. Type 1: 54% Fractures of the body. 1.2.2. Type 2: 17% Fractures of the acromion, spine, and coracoid process. 1.2.3. Type 3: 29% Fractures of the glenoid rim and fossa, and anatomical and surgical necks. 1.3. -

Splinting Avian Fractures
SPLINTING AVIAN FRACTURES Rebecca Duerr D V M M PV M International Bird Rescue Research Center Cordelia, CA © 2004, 2010: 2nd Edition, Rebecca Duerr. All drawings and images are by the author unless otherwise marked. TABLE OF CONTENTS Section Page • Considerations for wild bird care 2 • Glossary of terms 2 • Physical examination 3 • Before beginning the splint 4 • Compound fractures 5 • Prognoses of typical fractures 6 • The avian skeleton 10 • Examining the wing for possible fractures 11 • Splinting fractures of the wing: Humerus or radius/ulna fractures with support 12 • Splinting fractures of the wing: Metacarpal fractures with support 14 • Metacarpal wrap 15 • Calcium supplementation for fractured birds 15 • Slit wing wrap 16 • Examining the leg for possible fractures 17 • Splinting the femur: To immobilize prior to surgery or as the only treatment 18 • Tibiotarsus: making the splint 20 • Tibiotarsus: applying the splint 21 • Splinting the tarsometatarsus 22 • Splinting the foot—applying a shoe 23 • Mallards: walking/swimming splint for tarsometatarsus fractures 25 1 Considerations for wild bird care Treating wild birds with fractures requires the consideration of a number of factors that are not issues in treating domestic pets. First and foremost, each bird must be fit to be released when healed; even with raptors, available placement for disabled birds is a rare thing. It is even difficult to place charismatic species such as eagles. Consequently, reality (and usually rehabilitation licensing) dictates that a bird with an injury that will render it unable to fly or forage should be humanely euthanized. It is both illegal and inhumane to keep most wild birds as pets. -

Pectoral Girdle Anatomy & Physiology Fundamentals > Skeletal > Skeletal
Pectoral Girdle Anatomy & Physiology Fundamentals > Skeletal > Skeletal Pectoral girdle • Connects the upper limb to the axial skeleton and provides muscle attachment sites for both the arm and back • Comprises the clavicle (collarbone) and the scapula (shoulder blade) Clavicle Anatomy: • Sternal end - flat medial end, articulates with manubrium of sternum • Conoid tubercle - small bony projection where the conoid ligament attaches • Acromion end – rounded lateral end, articulates with acromion of scapula • Costal tuberosity (aka impression) - provides an attachment site for the costoclavicular ligament • Superior surface - smooth • Inferior surface - rough due to muscular attachments Key Points: • Cannot withstand heavy forces; fractures are common and can cause damage to the underlying brachial plexus and associated vessels. • The clavicle is one of the first bones to begin ossification during fetal development, but is the last to complete ossification, during the mid-twenties. • It is the only long bone that ossifies intramembraneously. Scapula Anatomy: • Supraspinous fossa - superior to the spine • Infraspinous fossa - inferior to the spine • Body – triangular surface of scapula • Superior angle – projects superomedially • Inferior angle - projects inferiorly • Lateral angle – projects laterally; includes the glenoid cavity 1 / 2 • Superior border - extends from the glenoid cavity to the superior angle • Lateral (aka axillary) border - extends from the inferior angle to the lateral angle • Medial (aka vertebral) border - extends from