The Reaction of Diphenyl Cadium with Phosphorus Trichloride
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

(A) 1) at First a Pair of Each 10G. of Sample Was Extracted with Ether by the Soxhlet's Apparatus for a Week
52 [vol. 6, THE DISTRIBUTION OF PHOSPHORUS IN COW'S MILK AND THE SCHEME FOR THE SEPARATION OF PHOSPHATIDES By Rinjiro SASAKI (From the Institute of Agricultural Chemistry, College of Agriculture, Tokyo Imperial University) (Received 9th September 1930) A. The Distribution of Phosphorus in Cow's milk In order to extract the phosphatides, it is necessary first to determine the distribution of phosphorus in cow's milk. Liquid milk can not be extracted with ether or with other organic solvents owing to its large content of water. The substance applied in this experiment is the milk powder, dried by the Buflovak drum drier below 70•Ž. Its content of moisture is 4.498%, deter mined by the method of drying in the air bath of 105•Ž. The content of total phosphorus was determined(5) in the ash of sample which was burned with a little of fusing mixture. In 100g. of milk powder In 100g. of dry matter Total phosphorus 0.745g. 0.781g. The amounts of phosphorus, which are soluble in the various organic sol vents, were determined by extracting with three solvents in the following orders. (A) Ether-Acetone-Alcohol (B) Acetone-Ether-Alcohol (C) Alcohol-Ether-Acetone Experiment (A) 1) At first a pair of each 10g. of sample was extracted with ether by the Soxhlet's apparatus for a week. After the extraction had been comp leted, the extract was freed from ether and weighed. In 100g.of milkpowder In 100g.of dry matter Total ether matters soluble 3.609g. 3.779g. 2) The above extract was dissolved in a very little of ether. -

Phosphorus Oxychloride CAS No
Product Safety Summary Phosphorus Oxychloride CAS No. 10025-87-3 This Product Safety Summary is intended to provide a general overview of the chemical substance. The information in the summary is basic information and is not intended to provide emergency response information, medical information or treatment information. The summary should not be used to provide in-depth safety and health information. In-depth safety and health information can be found in the Safety Data Sheet (SDS) for the chemical substance. Names Phosphorus oxychloride Phosphorus trichloride oxide Phosphoric trichloride POCl 3 Product Overview Solvay Novecare does not sell phosphorus oxychloride directly to consumers. Phosphorus oxychloride (POCl3) is used as a chemical intermediate to produce a variety of products which are used in several applications including manufacture of triarylphosphate esters which are used as flame retardants as well as an intermediate in the production of pharmaceutical, textile and agricultural chemicals. Phosphorus oxychloride is used in industrial applications and other processes where workplace exposures can occur. Consumer exposure does not occur as phosphorus oxychloride is not used in any commercially available product. Phosphorus oxychloride is dangerous to human health. Phosphorus oxychloride may be fatal if inhaled, highly toxic if swallowed, harmful if absorbed through skin and can cause severe burns which may result in scarring. Phosphorus oxychloride is consumed in manufacturing processes. POCl3 can make its way into the environment through unintentional releases (spills). POCl3 will not bioaccumulate but is not biodegradable. POCl3 in higher concentrations can be harmful to aquatic life due to formation of acids from the hydrolysis of POCl3. When released into the atmosphere, phosphorus oxychloride exists as vapor. -

5 Phosphorus Oxychloride1 Acute Exposure Guideline Levels
Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 10 Committee on Acute Exposure Guideline Levels Committee on Toxicology Board on Environmental Studies and Toxicology Division on Earth and Life Studies Copyright © National Academy of Sciences. All rights reserved. Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 10 THE NATIONAL ACADEMIES PRESS 500 FIFTH STREET, NW WASHINGTON, DC 20001 NOTICE: The project that is the subject of this report was approved by the Governing Board of the National Research Council, whose members are drawn from the councils of the National Academy of Sciences, the National Academy of Engineering, and the Insti- tute of Medicine. The members of the committee responsible for the report were chosen for their special competences and with regard for appropriate balance. This project was supported by Contract No. W81K04-06-D-0023 and EP-W-09-007 be- tween the National Academy of Sciences and the U.S. Department of Defense and the U.S. Environmental Protection Agency. Any opinions, findings, conclusions, or recom- mendations expressed in this publication are those of the author(s) and do not necessarily reflect the view of the organizations or agencies that provided support for this project. International Standard Book Number-13: 978-0-309-21987-7 International Standard Book Number-10: 0-309-21987-6 Additional copies of this report are available from The National Academies Press 500 Fifth Street, NW Box 285 Washington, DC 20055 800-624-6242 202-334-3313 (in the Washington metropolitan area) http://www.nap.edu Copyright 2011 by the National Academy of Sciences. -

United States Patent Office Patented Apr
3,030,421 United States Patent Office Patented Apr. 17, 1962 2 3,030,421 perature and under a reduced pressure. In the case of PROCESS FOR PREPARNG TRHYDROXY most of the catalysts the reaction products, which are free METHYL-PHOSPHINE s from solvents, solidify already when being cooled to room Martin Reuter and Ludwig Ortane, Frankfurt an Main, temperature. If, in the case of some catalysts, they Germany, assignors to Farbwerke Hoechst Aktienger 5 solidify only at a lower temperature and still contain to sellschaft vormas Meister Lucius & Briining, Frank a greater extent oily by-products and/or phosphonium furt am Main, Germany, a corporation of Gerinally hydroxide, they can be separated from the latter by filter No Drawing. Fied Jaa. 14, 1958, Ser. No. 708,764 ing or pressing. - - - Claims priority, application Germany Jan. 23, 1957 It is to be assumed that the crystalline main product 6 Canas. (C. 260-606.5) O of the present invention constitutes the hitherto unknown We have found that a new and valuable phosphorus trihydroxymethyl-phosphine. Main and by-products are compound carrying hydroxymethyl groups at the phos easily soluble in water and methanol and sparingly solu phorus atom can be prepared by reacting 1 mol of formal ble in fat dissolvers. dehyde with 4 mol of phosphine, preferably in the pres The reaction products of the invention can be used as ence of water, and in the presence of Small quantities of 5 insecticides, additives for lubricants, flame-proofing agents finely distributed metals that do not belong to the alkali for wood and textiles and as intermediates for these sub metals or alkaline earth metals and/or their compounds Staces. -

NPR72: the Pavlodar Chemical Weapons Plant in Kazakhstan
GULBARSHYN BOZHEYEVA Report The Pavlodar Chemical Weapons Plant in Kazakhstan: History and Legacy GULBARSHYN BOZHEYEVA Gulbarshyn Bozheyeva holds a Ph.D. degree in Chemical Physics from the Kazakh State University. From 1996-97, she was a Visiting Scholar and Research Associate at the Center for Nonproliferation Studies at the Monterey Institute of International Studies. She is now completing a master’s degree in International Development Policy at Duke University. he former Soviet Union’s chemical weapons theless, manufacturing lines and equipment for primary (CW) program consisted of many production and intermediate CW precursors and buildings for fill- plants that created the world’s largest stockpile ing CW munitions were constructed at Pavlodar. The T 1 of chemical weapons. Most of the CW production and plant also acquired personnel with expertise in CW pro- storage facilities were located in Russia, but a few fa- duction.5 cilities existed in other Soviet republics. In recent years, This report is devoted to the role of the Pavlodar Western countries have provided significant financial Chemical Plant in the former Soviet CW program and assistance for dismantling former CW facilities in Rus- its current status. The first part of the report describes sia and converting former CW production facilities for the history of the Pavlodar plant and its military and 2 commercial use. Although a fair amount has been writ- civilian infrastructures. The second part deals with the ten about Russian CW facilities, little is known about CW capability of the plant and the nature of the chemi- the CW programs in other former Soviet republics. -

United States Patent Office Patented Oct
3,346,562 United States Patent Office Patented Oct. 10, 1967 2 3,346,562 cg METHOD FOR THE PRODUCTION OF PO-CE Base RBONUCLEOSDE-5'-PHOSPHATE / O Mikio Honjo, Takatsuki, and Ryuji Maremoto, Minoo, Cl EO Japan, assignors to Takeda Chemical industries, Ltd., Osaka, Japan No Drawing. Filed May 31, 1966, Ser. No. 553,718 Claims priority, application Japan, May 29, 1965, R. X R. 40/31,814 9 Claims. (Cl. 260-21.5) HO. O. BIO O 10 N1 N1 This invention is concerned with a method for the pro Po-H, Base Po-H, Base duction of ribonucleoside-5'-phosphate. EIO k" wE+ EO k". Ribonucleoside-5'-phosphate is very useful as condi H HO - ment for food and also in the pharmaceutical industry, O O OH OH and has been chemically produced by at first protecting 15 X the hydroxyl groups at the 2'- and 3'-positions of its ribose R1 R2 moiety with acyl or isopropylidene groups and then phos phorylating the 5'-hydroxyl group of the thus-protected RN compound with pentavalent phosphorus compound such C=O: aliphatic ketone or aromatic aldehyde as phosphorus pentachloride, phosphorus oxychloride, 20 R?2 etc., followed by removing the protecting groups. As "ribonucleoside' in the present method there are However, this hitherto-known method requires a long used those containing purine base such as adenosine, time (about 7 to about 30 hours) for completing the pro inosine, etc. or those containing pyrimidine base such as tection and phosphorylation, and therefore is not desirable uridine, cytidine, etc. As the aliphatic ketone having 3 from an industrial viewpoint. -

Thermodynamic Properties of Mixtures of Aqueous Solutions of Hydrochloric Acid and Cadmium Chloride, Copper Chloride, Manganese Chloride, and Zinc Chloride
Western Michigan University ScholarWorks at WMU Master's Theses Graduate College 12-1993 Thermodynamic Properties of Mixtures of Aqueous Solutions of Hydrochloric Acid and Cadmium Chloride, Copper Chloride, Manganese Chloride, and Zinc Chloride Samia A. Kosa Follow this and additional works at: https://scholarworks.wmich.edu/masters_theses Part of the Chemistry Commons Recommended Citation Kosa, Samia A., "Thermodynamic Properties of Mixtures of Aqueous Solutions of Hydrochloric Acid and Cadmium Chloride, Copper Chloride, Manganese Chloride, and Zinc Chloride" (1993). Master's Theses. 4363. https://scholarworks.wmich.edu/masters_theses/4363 This Masters Thesis-Open Access is brought to you for free and open access by the Graduate College at ScholarWorks at WMU. It has been accepted for inclusion in Master's Theses by an authorized administrator of ScholarWorks at WMU. For more information, please contact [email protected]. THERMODYNAMIC PROPERTIES OF MIXTURES OF AQUEOUS SOLUTIONS OF HYDROCHLORIC ACID AND CADMIUM CHLORIDE, COPPER CHLORIDE, MANGANESE CHLORIDE, AND ZINC CHLORIDE by Samia A. Kosa A Thesis Submitted to the Faculty of The Graduate College in partial fulfillment of the requirements for the Degree of Master of Arts Department of Chemistry Western Michigan University Kalamazoo, Michigan December 1993 ACKNOWLEDGEMENTS I wish to express my deep gratitude and sincere appreciation to my committee advisor, Dr. Donald Schriber, for his continual guidance, direction, and assistance in this research. I would also like to acknowledge the help of the other committee members, Dr. Thomass Houser and Dr. Ralph Steinhaus, towards this work. My family continuously provided me support and encouragement and to them I owe more than I can say. -

Download Author Version (PDF)
Journal of Materials Chemistry A Accepted Manuscript This is an Accepted Manuscript, which has been through the Royal Society of Chemistry peer review process and has been accepted for publication. Accepted Manuscripts are published online shortly after acceptance, before technical editing, formatting and proof reading. Using this free service, authors can make their results available to the community, in citable form, before we publish the edited article. We will replace this Accepted Manuscript with the edited and formatted Advance Article as soon as it is available. You can find more information about Accepted Manuscripts in the Information for Authors. Please note that technical editing may introduce minor changes to the text and/or graphics, which may alter content. The journal’s standard Terms & Conditions and the Ethical guidelines still apply. In no event shall the Royal Society of Chemistry be held responsible for any errors or omissions in this Accepted Manuscript or any consequences arising from the use of any information it contains. www.rsc.org/materialsA Page 1 of 9 Journal of Materials Chemistry A ARTICLE JMCA Safer Salts for CdTe Nanocrystal Solution Processed Solar Cells: The Dual Roles of Ligand Exchange and Grain Growth Received 00th January 20xx, a b c d e Accepted 00th January 20xx Troy K. Townsend, † William B. Heuer, Edward E. Foos, Eric Kowalski, Woojun Yoon and Joseph G. Tischler e DOI: 10.1039/x0xx00000x Inorganic CdSe/CdTe nanocrystals for solid-state photovoltaic devices are typically sintered into a bulk-like material after www.rsc.org/ annealing in the presence of solid cadmium chloride. -
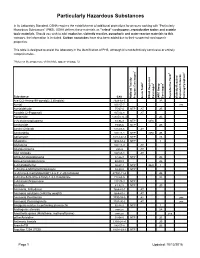
Particularly Hazardous Substances
Particularly Hazardous Substances In its Laboratory Standard, OSHA requires the establishment of additional protections for persons working with "Particularly Hazardous Substances" (PHS). OSHA defines these materials as "select" carcinogens, reproductive toxins and acutely toxic materials. Should you wish to add: explosive, violently reactive, pyrophoric and water-reactve materials to this category, the information is included. Carbon nanotubes have also been added due to their suspected carcinogenic properties. This table is designed to assist the laboratory in the identification of PHS, although it is not definitively conclusive or entirely comprehensive. *Notes on the proper use of this table appear on page 12. 1 6 5 2 3 4 Substance CAS National Toxicity National Program Carcinogen Toxin Acute Regulated OSHA Carcinogen Group IARC Carcinogen Toxin Reproductive Violently Reactive/ Explosive/Peroxide Forming/Pyrophoric A-a-C(2-Amino-9H-pyrido[2,3,b]indole) 2648-68-5 2B Acetal 105-57-7 yes Acetaldehyde 75-07-0 NTP AT 2B Acrolein (2-Propenal) 107-02-8 AT Acetamide 126850-14-4 2B 2-Acetylaminofluorene 53-96-3 NTP ORC Acrylamide 79-06-6 NTP 2B Acrylyl Chloride 814-68-6 AT Acrylonitrile 107-13-1 NTP ORC 2B Adriamycin 23214-92-8 NTP 2A Aflatoxins 1402-68-2 NTP 1 Allylamine 107-11-9 AT Alkylaluminums varies AT Allyl Chloride 107-05-1 AT ortho-Aminoazotoluene 97-56-3 NTP 2B para-aminoazobenzene 60-09-3 2B 4-Aminobiphenyl 92-67-1 NTP ORC 1 1-Amino-2-Methylanthraquinone 82-28-0 NTP (2-Amino-6-methyldipyrido[1,2-a:3’,2’-d]imidazole) 67730-11-4 2B -

The Effect of Sargassum Angustifolium Ethanol Extract on Cadmium Chloride-Induced Hypertension in Rat
Research Journal of Pharmacognosy (RJP) 8(1), 2021: 81-89 Received: 31 Oct 2020 Accepted: 17 Dec 2020 Published online: 19 Dec 2020 DOI: 10.22127/RJP.2020.255203.1637 Original article The Effect of Sargassum angustifolium Ethanol Extract on Cadmium Chloride-Induced Hypertension in Rat Leila Safaeian1* , Afsaneh Yegdaneh2, Masoud Mobasherian1 1Department of Pharmacology and Toxicology, Isfahan Pharmaceutical Sciences Research Center, School of Pharmacy and Pharmaceutical Sciences, Isfahan University of Medical Sciences, Isfahan, Iran. 2Department of Pharmacognosy, School of Pharmacy and Pharmaceutical Sciences, Isfahan University of Medical Sciences, Isfahan, Iran. Abstract Background and objectives: Sargassum angustifolium is a brown alga in southwestern coastline of Persian Gulf. Regarding the presence of various bioactive compounds and evidence of antihypertensive effects in other species of Sargassum, we evaluated the effect of S. angustifolium ethanol extract in CdCl2-induced hypertension in Wistar rats. Methods: Alga extract was prepared by maceration method using 70% ethanol and assessed for total phenolics and salt content. CdCl2 (1.5 mg/kg/day) was administered intraperitoneally to the rats for two weeks. Treatment groups received S. angustifolium extract (20, 40 and 80 mg/kg) or nifedipine (10 mg/kg) orally and simultaneously were given CdCl2 for two weeks. Systolic blood pressure (SBP) and heart rate were measured using tail-cuff method. Total antioxidant capacity, urea, creatinine, electrolytes including sodium, potassium, calcium and chloride were estimated in blood samples. The weight and histopathology of kidney tissues were also evaluated. Results: The content of total phenolic as gallic acid equivalent and the salt as NaCl was 67.42 ± 9.5 mg/g and 6.9 g/100 g in dried ethanol extract, respectively. -

Product Safety Summary
Product Safety Summary Phosphorus Trichloride CAS No. 7719-12-2 This Product Safety Summary is intended to provide a general overview of the chemical substance. The information in the summary is basic information and is not intended to provide emergency response information, medical information or treatment information. The summary should not be used to provide in-depth safety and health information. In-depth safety and health information can be found in the Safety Data Sheet (SDS) for the chemical substance. Names Phosphorus trichloride Phosphorus (III) chloride Phosphorous chloride PCl 3 Product Overview Solvay Novecare does not sell phosphorus trichloride directly to consumers and phosphorus trichloride has no known uses except as an intermediate to produce other chemicals. Phosphorus trichloride (PCl3) is used as a chemical intermediate to produce a variety of products which are used in several applications including agricultural products, surfactants and metal extractants, flame retardants, additives for lubricants and stabilizers for plastics. Phosphorus trichloride is used in industrial applications and other processes where workplace exposures can occur. Consumer exposure does not occur as phosphorus trichloride is not used in any commercially available product. Phosphorus trichloride is dangerous to human health. Phosphorus trichloride may be fatal if inhaled, highly toxic if swallowed, harmful if absorbed through skin and can cause severe burns which may result in scarring. Phosphorus trichloride is consumed in manufacturing processes. PCl3 can make its way into the environment through unintentional releases (spills). PCl3 will not bioaccumulate but is not biodegradable. Based on ecotoxicological testing performed on fish and fresh-water invertebrates, PCl3 in higher concentrations can be harmful to aquatic life due to formation of acids from the hydrolysis of PCl3. -

UNITED STATES PATENT OFFICE 2,683,168 PREPARATHON of ORGANO PHOSPHONY, CHELORADES Warren Jensen, Ponca City, Okla., and James O
Patented July 6, 1954 2,683,168 UNITED STATES PATENT OFFICE 2,683,168 PREPARATHON OF ORGANO PHOSPHONY, CHELORADES Warren Jensen, Ponca City, Okla., and James O. Claytoia, Berkeley, Calif., assigno's to California, Research Corporation, San Francisco, Calif., a corporation of Delaware No Drawing. Application December 22, 1950, Serial No. 202,396 8 Claims. (CI. 260-543) 1. 2 This invention relates to a method of prepar vide a means of preparing phosphonyl compounds ing phosphonyl chlorides and the like by the wherein the phosphorus atom is directly con reaction of an organic compound with phosphorus nected to an aliphatic carbon aton. trichloride in the presence of oxygen. it is another object of this invention to provide This application is a continuation-in-part of 5 a means of converting an aliphatic carbon-to Our Copending application, Serial No. 86,856 hydrogen bond to an aliphatic carbon-to-phoS (filed April 11, 1949 which has been abandoned, phorus bond. and was a continuation-in-part of application It is a further object of this invention to pro Serial No. 716,182 (filed December 12, 1946), which wide a more economical means of obtaining or has also been abandoned). O gano phosphorus compounds having a carbon-to Phosphonyl chloride and their derivatives are phosphorus linkage. useful in various arts. For example, certain It is a still further object of this invention to phosphonyl chloride derivatives (e.g., phosphonic provide a means of obtaining organo phosphorus acids and Salts and esters thereof) are useful compounds having