Product Safety Summary
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

This PDF Was Created from the British Library's Microfilm Copy of The
IMAGING SERVICES NORTH Boston Spa, Wetherby West Yorkshire, LS23 7BQ www.bl.uk This PDF was created from the British Library’s microfilm copy of the original thesis. As such the images are greyscale and no colour was captured. Due to the scanning process, an area greater than the page area is recorded and extraneous details can be captured. This is the best available copy THE BRITISH LIBRARY DOCUMENT SUPPLY CENTRE TITLE Synthesis and Biological Activities of a-aminoalkanephosphonic acids and a-aminoalkanephosphonous acids and their derivatives AUTHOR Fatima Bawa INSTITUTION and DATE The Polytechnic of North London, Attention is drawn to the fact that the copyright of this thesis rests with its author. This copy of the thesis has been supplied on condition that anyone who consults it is understood to recognise that its copyright rests with its author and that no information derived from it may be published without the author’s prior written consent THE BRITISH LIBRARY DOCUMENT SUPPLY CENTRE “ T* ~ n “T4I 1 II 1 II Bocton Spa, W atharty WaitYoffaMr« 20 cmt __L I 1 Unkad Kkifdoin RBOUCnONX'. The Polytechnic of North London in collaboration with Kenogard AB (Sweden) Synthesis and Biological Activities of a-aminoalkanephosphonic acids and a-amlnoalkanephosphonous acids and their derivatives by Fatima Bawa A thesis submitted for the Degree of Doctor of Philosophy of the Council for National Academic Awards. THE BRITISH LIBRARY DOCUMENT SUPPLY CENTRE BRITISH THESES NOT! C E The quality of this reproduction is heavily dependent upon the quality of the original thesis submitted for microfilming. Every effort has been made to ensure the highest quality of reproduction possible. -

Phosphorus Oxychloride CAS No
Product Safety Summary Phosphorus Oxychloride CAS No. 10025-87-3 This Product Safety Summary is intended to provide a general overview of the chemical substance. The information in the summary is basic information and is not intended to provide emergency response information, medical information or treatment information. The summary should not be used to provide in-depth safety and health information. In-depth safety and health information can be found in the Safety Data Sheet (SDS) for the chemical substance. Names Phosphorus oxychloride Phosphorus trichloride oxide Phosphoric trichloride POCl 3 Product Overview Solvay Novecare does not sell phosphorus oxychloride directly to consumers. Phosphorus oxychloride (POCl3) is used as a chemical intermediate to produce a variety of products which are used in several applications including manufacture of triarylphosphate esters which are used as flame retardants as well as an intermediate in the production of pharmaceutical, textile and agricultural chemicals. Phosphorus oxychloride is used in industrial applications and other processes where workplace exposures can occur. Consumer exposure does not occur as phosphorus oxychloride is not used in any commercially available product. Phosphorus oxychloride is dangerous to human health. Phosphorus oxychloride may be fatal if inhaled, highly toxic if swallowed, harmful if absorbed through skin and can cause severe burns which may result in scarring. Phosphorus oxychloride is consumed in manufacturing processes. POCl3 can make its way into the environment through unintentional releases (spills). POCl3 will not bioaccumulate but is not biodegradable. POCl3 in higher concentrations can be harmful to aquatic life due to formation of acids from the hydrolysis of POCl3. When released into the atmosphere, phosphorus oxychloride exists as vapor. -

5 Phosphorus Oxychloride1 Acute Exposure Guideline Levels
Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 10 Committee on Acute Exposure Guideline Levels Committee on Toxicology Board on Environmental Studies and Toxicology Division on Earth and Life Studies Copyright © National Academy of Sciences. All rights reserved. Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 10 THE NATIONAL ACADEMIES PRESS 500 FIFTH STREET, NW WASHINGTON, DC 20001 NOTICE: The project that is the subject of this report was approved by the Governing Board of the National Research Council, whose members are drawn from the councils of the National Academy of Sciences, the National Academy of Engineering, and the Insti- tute of Medicine. The members of the committee responsible for the report were chosen for their special competences and with regard for appropriate balance. This project was supported by Contract No. W81K04-06-D-0023 and EP-W-09-007 be- tween the National Academy of Sciences and the U.S. Department of Defense and the U.S. Environmental Protection Agency. Any opinions, findings, conclusions, or recom- mendations expressed in this publication are those of the author(s) and do not necessarily reflect the view of the organizations or agencies that provided support for this project. International Standard Book Number-13: 978-0-309-21987-7 International Standard Book Number-10: 0-309-21987-6 Additional copies of this report are available from The National Academies Press 500 Fifth Street, NW Box 285 Washington, DC 20055 800-624-6242 202-334-3313 (in the Washington metropolitan area) http://www.nap.edu Copyright 2011 by the National Academy of Sciences. -

Phosphorus and Sulfur Cosmochemistry: Implications for the Origins of Life
Phosphorus and Sulfur Cosmochemistry: Implications for the Origins of Life Item Type text; Electronic Dissertation Authors Pasek, Matthew Adam Publisher The University of Arizona. Rights Copyright © is held by the author. Digital access to this material is made possible by the University Libraries, University of Arizona. Further transmission, reproduction or presentation (such as public display or performance) of protected items is prohibited except with permission of the author. Download date 07/10/2021 06:16:37 Link to Item http://hdl.handle.net/10150/194288 PHOSPHORUS AND SULFUR COSMOCHEMISTRY: IMPLICATIONS FOR THE ORIGINS OF LIFE by Matthew Adam Pasek ________________________ A Dissertation Submitted to the Faculty of the DEPARTMENT OF PLANETARY SCIENCE In Partial Fulfillment of the Requirements For the Degree of DOCTOR OF PHILOSOPHY In the Graduate College UNIVERSITY OF ARIZONA 2 0 0 6 2 THE UNIVERSITY OF ARIZONA GRADUATE COLLEGE As members of the Dissertation Committee, we certify that we have read the dissertation prepared by Matthew Adam Pasek entitled Phosphorus and Sulfur Cosmochemistry: Implications for the Origins of Life and recommend that it be accepted as fulfilling the dissertation requirement for the Degree of Doctor of Philosophy _______________________________________________________________________ Date: 04/11/2006 Dante Lauretta _______________________________________________________________________ Date: 04/11/2006 Timothy Swindle _______________________________________________________________________ Date: 04/11/2006 -

NPR72: the Pavlodar Chemical Weapons Plant in Kazakhstan
GULBARSHYN BOZHEYEVA Report The Pavlodar Chemical Weapons Plant in Kazakhstan: History and Legacy GULBARSHYN BOZHEYEVA Gulbarshyn Bozheyeva holds a Ph.D. degree in Chemical Physics from the Kazakh State University. From 1996-97, she was a Visiting Scholar and Research Associate at the Center for Nonproliferation Studies at the Monterey Institute of International Studies. She is now completing a master’s degree in International Development Policy at Duke University. he former Soviet Union’s chemical weapons theless, manufacturing lines and equipment for primary (CW) program consisted of many production and intermediate CW precursors and buildings for fill- plants that created the world’s largest stockpile ing CW munitions were constructed at Pavlodar. The T 1 of chemical weapons. Most of the CW production and plant also acquired personnel with expertise in CW pro- storage facilities were located in Russia, but a few fa- duction.5 cilities existed in other Soviet republics. In recent years, This report is devoted to the role of the Pavlodar Western countries have provided significant financial Chemical Plant in the former Soviet CW program and assistance for dismantling former CW facilities in Rus- its current status. The first part of the report describes sia and converting former CW production facilities for the history of the Pavlodar plant and its military and 2 commercial use. Although a fair amount has been writ- civilian infrastructures. The second part deals with the ten about Russian CW facilities, little is known about CW capability of the plant and the nature of the chemi- the CW programs in other former Soviet republics. -

United States Patent Office Patented Oct
3,346,562 United States Patent Office Patented Oct. 10, 1967 2 3,346,562 cg METHOD FOR THE PRODUCTION OF PO-CE Base RBONUCLEOSDE-5'-PHOSPHATE / O Mikio Honjo, Takatsuki, and Ryuji Maremoto, Minoo, Cl EO Japan, assignors to Takeda Chemical industries, Ltd., Osaka, Japan No Drawing. Filed May 31, 1966, Ser. No. 553,718 Claims priority, application Japan, May 29, 1965, R. X R. 40/31,814 9 Claims. (Cl. 260-21.5) HO. O. BIO O 10 N1 N1 This invention is concerned with a method for the pro Po-H, Base Po-H, Base duction of ribonucleoside-5'-phosphate. EIO k" wE+ EO k". Ribonucleoside-5'-phosphate is very useful as condi H HO - ment for food and also in the pharmaceutical industry, O O OH OH and has been chemically produced by at first protecting 15 X the hydroxyl groups at the 2'- and 3'-positions of its ribose R1 R2 moiety with acyl or isopropylidene groups and then phos phorylating the 5'-hydroxyl group of the thus-protected RN compound with pentavalent phosphorus compound such C=O: aliphatic ketone or aromatic aldehyde as phosphorus pentachloride, phosphorus oxychloride, 20 R?2 etc., followed by removing the protecting groups. As "ribonucleoside' in the present method there are However, this hitherto-known method requires a long used those containing purine base such as adenosine, time (about 7 to about 30 hours) for completing the pro inosine, etc. or those containing pyrimidine base such as tection and phosphorylation, and therefore is not desirable uridine, cytidine, etc. As the aliphatic ketone having 3 from an industrial viewpoint. -
![United States Patent [19] [11] Patent Number: 5,368,832 Buckholtz Et A1](https://docslib.b-cdn.net/cover/1511/united-states-patent-19-11-patent-number-5-368-832-buckholtz-et-a1-641511.webp)
United States Patent [19] [11] Patent Number: 5,368,832 Buckholtz Et A1
USOO5368832A United States Patent [19] [11] Patent Number: 5,368,832 Buckholtz et a1. [45] Date of Patent: Nov. 29, 1994 [54] ZERO DISCI'IARGE PRQCESS FOR 4,330,515 5/1982 Campbell .......................... .. 423/316 MANUFACI'URING OF PHOSPHOROUS 4,380,531 4/1983 Wisnouskas ....................... .. 423/316 ACID AND HYPOPHOSPHOROUS ACID FOREIGN PATENT DOCUMENTS [75] 1I1v¢m°rs= HWY E- Buckh‘?tz, Lewiswn; 0007493 2/1980 European Pat. Off. .......... .. 423/317 Mohan S. Saran, Grand Island, both 254166 11/1986 Japan _ Of N.Y.; Frederick C. Leiterf, 7900920 11/1979 WIPO ............................... .. 423/316 Madison; David A. Flautt, Ashtabula, both of Ohio Primary iExaminer-Michael Lewis Assistant Examiner—-Stephen G. Kalinchak [73] Assignee: gggiggg?gg? Corporation’ gltltglrgey, Agent, or Firm-Wayne A. Jones; Richard D. [21] Appl. No.: 711,841 [57] ' ABS Cr [22] Flled' : Jun . 7 ’ 1991 Dlsclosed. 1s. a method of making. phosphorous acid. or [51] Int. Cl.5 .................. .. C01B 25/165; COlB 25/163 hypophosphomus acid by reacting hydrogen chloride U:S. C1. ................................... .. 423/ 316; 423/ 317 with a sodium phosphite or a sodium hypophosphite’ [58] Field of Search ............. .. 423/ 167, 307, 316, 317, respectively, in the presence of water to precipitate 423/321 R sodium chloride crystals and form the acid. The acid is [56] References Cited separated from the sodium chloride crystals and-can be US PATENT DOCUMENTS passed through amon exchange column that 1s pref erably loaded with phosphite or hypophosphite lons, 2,595,198 4/1952 Leffore et a1. ............ .. 423/316 respectively, to remove'residual chloride ions_ 2,711,388 6/1955 Mottern et a1. -

Phosphorous Acid
Phosphorous acid sc-286667 Material Safety Data Sheet Hazard Alert Code EXTREME HIGH MODERATE LOW Key: Section 1 - CHEMICAL PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME Phosphorous acid STATEMENT OF HAZARDOUS NATURE CONSIDERED A HAZARDOUS SUBSTANCE ACCORDING TO OSHA 29 CFR 1910.1200. NFPA FLAMMABILITY0 HEALTH3 HAZARD INSTABILITY1 SUPPLIER Santa Cruz Biotechnology, Inc. 2145 Delaware Avenue Santa Cruz, California 95060 800.457.3801 or 831.457.3800 EMERGENCY ChemWatch Within the US & Canada: 877-715-9305 Outside the US & Canada: +800 2436 2255 (1-800-CHEMCALL) or call +613 9573 3112 SYNONYMS H3-O3-P, H3PO3, "phosphonic acid", "orthophosphorous acid", "phosphorus trihydroxide", trihydroxyphosphine, "phosphorus acid (sic)", "o-phosphorous acid", "o-phosphorus acid (sic)" Section 2 - HAZARDS IDENTIFICATION CHEMWATCH HAZARD RATINGS Min Max Flammability: 0 Toxicity: 2 Body Contact: 4 Min/Nil=0 Low=1 Reactivity: 1 Moderate=2 High=3 Chronic: 2 Extreme=4 CANADIAN WHMIS SYMBOLS 1 of 9 EMERGENCY OVERVIEW RISK Harmful if swallowed. Causes severe burns. Risk of serious damage to eyes. May cause long-term adverse effects in the aquatic environment. Cumulative effects may result following exposure*. * (limited evidence). POTENTIAL HEALTH EFFECTS ACUTE HEALTH EFFECTS SWALLOWED ■ Accidental ingestion of the material may be harmful; animal experiments indicate that ingestion of less than 150 gram may be fatal or may produce serious damage to the health of the individual. ■ Ingestion of acidic corrosives may produce burns around and in the mouth, the throat and oesophagus. Immediate pain and difficulties in swallowing and speaking may also be evident. ■ As absorption of phosphates from the bowel is poor, poisoning this way is less likely. -

Neutralized Phosphorous Acid to Control Phytophthora Diseases
Plant Pathology Bulletin 18: 155-165, 2009 A Simple Technique, Concentration and Application Schedule for Using Neutralized Phosphorous Acid to Control Phytophthora Diseases Pao-Jen Ann 1, 4, Jyh-Nong Tsai 1, In-Ting Wong 1, Ting-Fang Hsieh 2, and Chien-Yih Lin 3 1. Plant Pathology Division, Taiwan Agriculture Research Institute, Taichung, 413, Taiwan 2. Floriculture Research Center, Taiwan Agriculture Research Institute, Yulin, 646, Taiwan 3. College of Health Science, Asia University, Taichung 413, Taiwan 4. Corresponding author, E-mail: [email protected]; Fax: 886-4-2330-2803 Accepted for publication: September 19, 2009 ABSTRACT Ann, P. J., Tsai, J. N., Wong, I. T., Hsieh, T. F., and Lin, C. Y. 2009. A simple technique, concentration and application schedule for using neutralized phosphorous acid to control Phytophthora diseases. Plant Pathol. Bull. 18: 155-165. A simple method for application of phosphorous acid to control Phytophthora diseases was developed. The proposed method involves measuring equal weight of phosphorous acid and potassium hydroxide and dissolving phosphorous acid in water before adding potassium hydroxide. The pH of the neutralized phosphorous acid solution (NPA) is 6.2-6.7. The NPA concentration at 5000 mg/L was effective in controlling tomato seedling blight caused by Phytophthora capsici when using soil drench method. For suppression of lily leaf and blossom blight caused by Phytophthora parasitica, foliar application is much more effective than soil drench. However, NPA concentrations exceeding 2000 mg/L were phytotoxic to young plant tissues. The best results were achieved by applying NPA at the rate of 1000 mg/L for 3 times at one week intervals. -

United States Patent (19) 11) 4,278,647 Jüdden Et Al
United States Patent (19) 11) 4,278,647 Jüdden et al. 45) Jul. 14, 1981 54 PRODUCTION OF PHOSPHOROUS ACID 56) References Cited 75 Inventors: Klaus Jödden, Hirth; Hans-Werner FOREIGN PATENT DOCUMENTS Stephan, Cologne; Gero Heymer, 1218063 12/1958 France ..................................... 423/317 Erftstadt, all of Fed. Rep. of Primary Examiner-O. R. Vertiz Germany Assistant Examiner-Gregory A. Heller 73) Assignee: Hoechst Aktiengesellschaft, Fed. 57 ABSTRACT Rep. of Germany The invention relates to a process for making aqueous 21 Appl. No.: 60,659 phosphorous acid solutions by admixing an aqueous sodium phosphite solution with hydrochloric acid. 22 Filed: Jul. 25, 1979 More specifically, phosphorous acid solutions substan tially free from sodium in hydrochloric acid are pro (30) Foreign Application Priority Data duced by introducing hydrogen chloride, into the so Jul, 29, 1978 DE Fed. Rep. of Germany ....... 2833380 dium phosphite solution until the latter is saturated therewith and separating precipitating sodium chloride 51 Int. Cl. ....................... C01B 25/16; CO1B 25/26 from the resulting phosphorous acid solution in hydro 52 U.S.C. ..................................... 423/317; 423/307 chloric acid. 58 Field of Search ............... 423/317, 307, 499, 481, 423/316 8 Claims, No Drawings 4,278,647 2 adversely affect the work up of the resulting phospho PRODUCTION OF PHOSPHOROUS ACID rous acid solution, it is good practice, prior to the intro duction of hydrogen chloride, to pass a free oxygen This invention relates to a process for making aque containing gas, e.g. air or oxygen, through the sodium ous phosphorous acid solutions by admixing an aqueous phosphite solution at elevated temperature, preferably sodium phosphite solution with hydrochloric acid. -
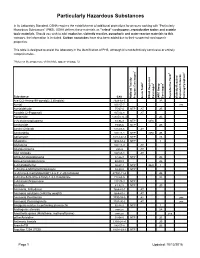
Particularly Hazardous Substances
Particularly Hazardous Substances In its Laboratory Standard, OSHA requires the establishment of additional protections for persons working with "Particularly Hazardous Substances" (PHS). OSHA defines these materials as "select" carcinogens, reproductive toxins and acutely toxic materials. Should you wish to add: explosive, violently reactive, pyrophoric and water-reactve materials to this category, the information is included. Carbon nanotubes have also been added due to their suspected carcinogenic properties. This table is designed to assist the laboratory in the identification of PHS, although it is not definitively conclusive or entirely comprehensive. *Notes on the proper use of this table appear on page 12. 1 6 5 2 3 4 Substance CAS National Toxicity National Program Carcinogen Toxin Acute Regulated OSHA Carcinogen Group IARC Carcinogen Toxin Reproductive Violently Reactive/ Explosive/Peroxide Forming/Pyrophoric A-a-C(2-Amino-9H-pyrido[2,3,b]indole) 2648-68-5 2B Acetal 105-57-7 yes Acetaldehyde 75-07-0 NTP AT 2B Acrolein (2-Propenal) 107-02-8 AT Acetamide 126850-14-4 2B 2-Acetylaminofluorene 53-96-3 NTP ORC Acrylamide 79-06-6 NTP 2B Acrylyl Chloride 814-68-6 AT Acrylonitrile 107-13-1 NTP ORC 2B Adriamycin 23214-92-8 NTP 2A Aflatoxins 1402-68-2 NTP 1 Allylamine 107-11-9 AT Alkylaluminums varies AT Allyl Chloride 107-05-1 AT ortho-Aminoazotoluene 97-56-3 NTP 2B para-aminoazobenzene 60-09-3 2B 4-Aminobiphenyl 92-67-1 NTP ORC 1 1-Amino-2-Methylanthraquinone 82-28-0 NTP (2-Amino-6-methyldipyrido[1,2-a:3’,2’-d]imidazole) 67730-11-4 2B -

CHEM 1411 Nomenclature Homework - Answers Part I
1 CHEM 1411 Nomenclature Homework - Answers Part I 1. The following are a list of binary and pseudobinary ionic compounds. Write the name when the formula is given. Write the formula when the name is given. (a) AlCl3 aluminum chloride (k) rubidium oxide Rb2O (b) AuBr3 gold (III) bromide (l) chromium (III) selenide Cr2Se3 (c) Na2S sodium sulfide (m) barium iodide BaI2 (d) Cu3P2 copper (II) phosphide (n) copper (I) fluoride CuF (e) Fe(OH)2 iron (II) hydroxide (o) copper (II) fluoride CuF2 (f) NH4OH ammonium hydroxide (p) strontium cyanide Sr(CN)2 (g) Co(CH3COO)3 cobalt (III) acetate (q) mercury (II) bromide HgBr2 (h) Zn(SCN)2 zinc thiocyanate (r) mercury (I) bromide Hg2Br2 (i) CaCrO4 calcium chromate (s) magnesium permanganate Mg(MnO4)2 (j) K2Cr2O7 potassium dichromate (t) lithium nitride Li3N 2. The following are lists of covalent compounds. Write the name when a formula is given. Write the formula when given a name. (a) CSe2 carbon diselenide (h) dichlorine heptoxide Cl2O7 (b) SF6 sulfur hexafluoride (i) xenon tetrafluoride XeF4 (c) BrF5 bromine pentafluoride (j) carbon monoxide CO (d) P4O10 tetraphosphorous decoxide (k) oxygen O2 (e) Cl2O dichlorine oxide (l) diboron trioxide B2B O3 (f) NH3 ammonia (m) arsenic trifluoride AsF3 (g) N2 dinitrogen or nitrogen (n) diiodine I2 2 3. The following are lists of acids or acid-forming compounds. Write the name when the formula is given. Write the formula when the name is given. (a) H3PO2 hypophosphorous acid (k) hydrogen cyanide HCN (g) (b) H2SO4 sulfuric acid (l) periodic acid HIO4 (c) HClO hypochlorous acid (m) hypochlorous acid HClO (d) H3PO4 phosphoric acid (n) nitric acid HNO3 (e) HBrO4 perbromic acid (o) acetic acid CH3CO2H (f) HIO2 iodous acid (p) chloric acid HClO3 (g) HI (g) hydrogen iodide (q) perbromic acid HBrO4 (h) HI (aq) hydroiodic acid (r) hydrofluoric acid HF (aq) (i) HCN (aq) hydrocyanic acid (s) phosphorous acid H3PO3 (j) HBrO hypobromous acid (t) hydrosulfuric acid H2S (aq) 4.