Proquest Dissertations
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Sequencing As a Way of Work
Edinburgh Research Explorer A new insight into Sanger’s development of sequencing Citation for published version: Garcia-Sancho, M 2010, 'A new insight into Sanger’s development of sequencing: From proteins to DNA, 1943-77', Journal of the History of Biology, vol. 43, no. 2, pp. 265-323. https://doi.org/10.1007/s10739-009- 9184-1 Digital Object Identifier (DOI): 10.1007/s10739-009-9184-1 Link: Link to publication record in Edinburgh Research Explorer Document Version: Peer reviewed version Published In: Journal of the History of Biology Publisher Rights Statement: © Garcia-Sancho, M. (2010). A new insight into Sanger’s development of sequencing: From proteins to DNA, 1943-77. Journal of the History of Biology, 43(2), 265-323. 10.1007/s10739-009-9184-1 General rights Copyright for the publications made accessible via the Edinburgh Research Explorer is retained by the author(s) and / or other copyright owners and it is a condition of accessing these publications that users recognise and abide by the legal requirements associated with these rights. Take down policy The University of Edinburgh has made every reasonable effort to ensure that Edinburgh Research Explorer content complies with UK legislation. If you believe that the public display of this file breaches copyright please contact [email protected] providing details, and we will remove access to the work immediately and investigate your claim. Download date: 28. Sep. 2021 THIS IS AN ADVANCED DRAFT OF A PUBLISHED PAPER. REFERENCES AND QUOTATIONS SHOULD ALWAYS BE MADE TO THE PUBLISHED VERION, WHICH CAN BE FOUND AT: García-Sancho M. -

Environmental Influences on Endothelial Gene Expression
ENDOTHELIAL CELL GENE EXPRESSION John Matthew Jeff Herbert Supervisors: Prof. Roy Bicknell and Dr. Victoria Heath PhD thesis University of Birmingham August 2012 University of Birmingham Research Archive e-theses repository This unpublished thesis/dissertation is copyright of the author and/or third parties. The intellectual property rights of the author or third parties in respect of this work are as defined by The Copyright Designs and Patents Act 1988 or as modified by any successor legislation. Any use made of information contained in this thesis/dissertation must be in accordance with that legislation and must be properly acknowledged. Further distribution or reproduction in any format is prohibited without the permission of the copyright holder. ABSTRACT Tumour angiogenesis is a vital process in the pathology of tumour development and metastasis. Targeting markers of tumour endothelium provide a means of targeted destruction of a tumours oxygen and nutrient supply via destruction of tumour vasculature, which in turn ultimately leads to beneficial consequences to patients. Although current anti -angiogenic and vascular targeting strategies help patients, more potently in combination with chemo therapy, there is still a need for more tumour endothelial marker discoveries as current treatments have cardiovascular and other side effects. For the first time, the analyses of in-vivo biotinylation of an embryonic system is performed to obtain putative vascular targets. Also for the first time, deep sequencing is applied to freshly isolated tumour and normal endothelial cells from lung, colon and bladder tissues for the identification of pan-vascular-targets. Integration of the proteomic, deep sequencing, public cDNA libraries and microarrays, delivers 5,892 putative vascular targets to the science community. -

Anfinsen Experiments
Lecture Notes - 2 7.24/7.88J/5.48J The Protein Folding Problem Handouts: An Anfinsen paper Reading List Anfinsen Experiments The Problem of the title refers to how the amino acid sequence of a polypeptide chain determines the folded three-dimensional organization of the chain: • Does the sequence determine the structure?? • Protein Denaturation • Emergence of the Problem • Ribonuclease A • Refolding of ribonuclease in vitro. A. Protein Denaturation/Inactivation One of the features that identified proteins as distinctive polymers was 1) Unusual phase transition in semi purified proteins When exposed to relatively gentle conditions outside the range of physiological • heat • pH • salt • organic solvents, e.g. alcohols Lets consider the familiar transition I mentioned last week; • Heat denaturation; cooking the white of an egg • Acid denaturation; Milk turning sour Macroscopic changes in bulk solution; scatters visible light, increase in viscosity - white of egg: >>heat; opaque, hard; cool down; no change: Coagulation, Aggregation, precipitation, denaturation: One of the components is egg white lysozyme: activity assay; hydrolysis of bacterial cell walls: Activity remaining vs. temperature on same axes This transition: is Denaturation. These transitions were in general found to be irreversible: activity was not recovered upon cooling: Historically the study of the folding and unfolding of proteins emerged from trying to understand this unusual change of state or phase transitions. B. Emergence of the Protein Folding Problem In the period 1958-1960, the first structure of a protein molecule - myoglobin, the oxygen binding protein of muscle - was solved by John Kendrew in 1958, followed soon after by the related tetrameric red blood cell oxygen binding protein, hemoglobin, solved by Max Perutz, both of the British Medical Research Council Labs in Cambridge, U.K. -

Peptide Chemistry up to Its Present State
Appendix In this Appendix biographical sketches are compiled of many scientists who have made notable contributions to the development of peptide chemistry up to its present state. We have tried to consider names mainly connected with important events during the earlier periods of peptide history, but could not include all authors mentioned in the text of this book. This is particularly true for the more recent decades when the number of peptide chemists and biologists increased to such an extent that their enumeration would have gone beyond the scope of this Appendix. 250 Appendix Plate 8. Emil Abderhalden (1877-1950), Photo Plate 9. S. Akabori Leopoldina, Halle J Plate 10. Ernst Bayer Plate 11. Karel Blaha (1926-1988) Appendix 251 Plate 12. Max Brenner Plate 13. Hans Brockmann (1903-1988) Plate 14. Victor Bruckner (1900- 1980) Plate 15. Pehr V. Edman (1916- 1977) 252 Appendix Plate 16. Lyman C. Craig (1906-1974) Plate 17. Vittorio Erspamer Plate 18. Joseph S. Fruton, Biochemist and Historian Appendix 253 Plate 19. Rolf Geiger (1923-1988) Plate 20. Wolfgang Konig Plate 21. Dorothy Hodgkins Plate. 22. Franz Hofmeister (1850-1922), (Fischer, biograph. Lexikon) 254 Appendix Plate 23. The picture shows the late Professor 1.E. Jorpes (r.j and Professor V. Mutt during their favorite pastime in the archipelago on the Baltic near Stockholm Plate 24. Ephraim Katchalski (Katzir) Plate 25. Abraham Patchornik Appendix 255 Plate 26. P.G. Katsoyannis Plate 27. George W. Kenner (1922-1978) Plate 28. Edger Lederer (1908- 1988) Plate 29. Hennann Leuchs (1879-1945) 256 Appendix Plate 30. Choh Hao Li (1913-1987) Plate 31. -

The Early Years-Across the Bench from Bruce (1963-1966)
The Early Years—Across the Bench From Bruce (1963–1966) The Early Years—Across the Bench From Bruce (1963–1966) Garland R. Marshall1,2 1Department of Biochemistry and Molecular Biophysics, Center for Computational Biology, Washington University, St. Louis, MO 63110 2Department of Biomedical Engineering, Center for Computational Biology, Washington University, St. Louis, MO 63110 Received 14 July 2007; revised 20 September 2007; accepted 5 October 2007 Published online 16 October 2007 in Wiley InterScience (www.interscience.wiley.com). DOI 10.1002/bip.20870 a Nobel Laureate, Chairman of the Department of Biology at ABSTRACT: Caltech and a member of the National Academy of Science, and was still willing to recommend me for graduate studies This personal reflection on the author’s experience as at Rockefeller. Bruce Merrifield’s first graduate student has been I was convinced at the time that I was chosen to study adapted from a talk given at the Merrifield Memorial neurophysiology, having failed miserably to isolate the acetyl- Symposium at the Rockefeller University on November choline receptor from denervated rabbit muscle as an under- graduate at Caltech. The outstanding neurophysiologists at 13, 2006. # 2007 Wiley Periodicals, Inc. Biopolymers Rockefeller including H. Keffer Hartline, Nobel Laureate, (Pept Sci) 90: 190–199, 2008. were more interested, however, in the wiring diagrams of the Keywords: solid phase synthesis; Merrifield; DNA synthe- eye of the horseshoe crab2 than in how a small molecule sis; combinatorial chemistry could trigger the action potential. Thus, my first laboratory experience at Rockefeller was with Prof. Henry Kunkel, a This article was originally published online as an accepted prominent immunologist.3 Prof. -

Genome-Wide Screen of Cell-Cycle Regulators in Normal and Tumor Cells
bioRxiv preprint doi: https://doi.org/10.1101/060350; this version posted June 23, 2016. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Genome-wide screen of cell-cycle regulators in normal and tumor cells identifies a differential response to nucleosome depletion Maria Sokolova1, Mikko Turunen1, Oliver Mortusewicz3, Teemu Kivioja1, Patrick Herr3, Anna Vähärautio1, Mikael Björklund1, Minna Taipale2, Thomas Helleday3 and Jussi Taipale1,2,* 1Genome-Scale Biology Program, P.O. Box 63, FI-00014 University of Helsinki, Finland. 2Science for Life laboratory, Department of Biosciences and Nutrition, Karolinska Institutet, SE- 141 83 Stockholm, Sweden. 3Science for Life laboratory, Division of Translational Medicine and Chemical Biology, Department of Medical Biochemistry and Biophysics, Karolinska Institutet, S-171 21 Stockholm, Sweden To identify cell cycle regulators that enable cancer cells to replicate DNA and divide in an unrestricted manner, we performed a parallel genome-wide RNAi screen in normal and cancer cell lines. In addition to many shared regulators, we found that tumor and normal cells are differentially sensitive to loss of the histone genes transcriptional regulator CASP8AP2. In cancer cells, loss of CASP8AP2 leads to a failure to synthesize sufficient amount of histones in the S-phase of the cell cycle, resulting in slowing of individual replication forks. Despite this, DNA replication fails to arrest, and tumor cells progress in an elongated S-phase that lasts several days, finally resulting in death of most of the affected cells. -

Histone-Related Genes Are Hypermethylated in Lung Cancer
Published OnlineFirst October 1, 2019; DOI: 10.1158/0008-5472.CAN-19-1019 Cancer Genome and Epigenome Research Histone-Related Genes Are Hypermethylated in Lung Cancer and Hypermethylated HIST1H4F Could Serve as a Pan-Cancer Biomarker Shihua Dong1,Wei Li1, Lin Wang2, Jie Hu3,Yuanlin Song3, Baolong Zhang1, Xiaoguang Ren1, Shimeng Ji3, Jin Li1, Peng Xu1, Ying Liang1, Gang Chen4, Jia-Tao Lou2, and Wenqiang Yu1 Abstract Lung cancer is the leading cause of cancer-related deaths lated in all 17 tumor types from TCGA datasets (n ¼ 7,344), worldwide. Cytologic examination is the current "gold stan- which was further validated in nine different types of cancer dard" for lung cancer diagnosis, however, this has low sensi- (n ¼ 243). These results demonstrate that HIST1H4F can tivity. Here, we identified a typical methylation signature of function as a universal-cancer-only methylation (UCOM) histone genes in lung cancer by whole-genome DNA methyl- marker, which may aid in understanding general tumorigen- ation analysis, which was validated by The Cancer Genome esis and improve screening for early cancer diagnosis. Atlas (TCGA) lung cancer cohort (n ¼ 907) and was further confirmed in 265 bronchoalveolar lavage fluid samples with Significance: These findings identify a new biomarker for specificity and sensitivity of 96.7% and 87.0%, respectively. cancer detection and show that hypermethylation of histone- More importantly, HIST1H4F was universally hypermethy- related genes seems to persist across cancers. Introduction to its low specificity, LDCT is far from satisfactory as a screening tool for clinical application, similar to other currently used cancer Lung cancer is one of the most common malignant tumors and biomarkers, such as carcinoembryonic antigen (CEA), neuron- the leading cause of cancer-related deaths worldwide (1, 2). -

JUAN MANUEL 2016 NOBEL PEACE PRIZE RECIPIENT Culture Friendship Justice
Friendship Volume 135, № 1 Character Culture JUAN MANUEL SANTOS 2016 NOBEL PEACE PRIZE RECIPIENT Justice LETTER FROM THE PRESIDENT Dear Brothers, It is an honor and a privilege as your president to have the challenges us and, perhaps, makes us question our own opportunity to share my message with you in each edition strongly held beliefs. But it also serves to open our minds of the Quarterly. I generally try to align my comments and our hearts to our fellow neighbor. It has to start with specific items highlighted in each publication. This with a desire to listen, to understand, and to be tolerant time, however, I want to return to the theme “living our of different points of view and a desire to be reasonable, Principles,” which I touched upon in a previous article. As patient and respectful.” you may recall, I attempted to outline and describe how Kelly concludes that it is the diversity of Southwest’s utilization of the Four Founding Principles could help people and “treating others like you would want to be undergraduates make good decisions and build better treated” that has made the organization successful. In a men. It occurred to me that the application of our values similar way, Stephen Covey’s widely read “Seven Habits of to undergraduates only is too limiting. These Principles are Highly Effective People” takes a “values-based” approach to indeed critical for each of us at this particularly turbulent organizational success. time in our society. For DU to be a successful organization, we too, must As I was flying back recently from the Delta Upsilon be able to work effectively with our varied constituents: International Fraternity Board of Directors meeting in undergraduates, parents, alumni, higher education Arizona, I glanced through the February 2017 edition professionals, etc. -

Transcriptomic Profiles of High and Low Antibody Responders to Smallpox
Genes and Immunity (2013) 14, 277–285 & 2013 Macmillan Publishers Limited All rights reserved 1466-4879/13 www.nature.com/gene ORIGINAL ARTICLE Transcriptomic profiles of high and low antibody responders to smallpox vaccine RB Kennedy1,2, AL Oberg1,3, IG Ovsyannikova1,2, IH Haralambieva1,2, D Grill1,3 and GA Poland1,2 Despite its eradication over 30 years ago, smallpox (as well as other orthopox viruses) remains a pathogen of interest both in terms of biodefense and for its use as a vector for vaccines and immunotherapies. Here we describe the application of mRNA-Seq transcriptome profiling to understanding immune responses in smallpox vaccine recipients. Contrary to other studies examining gene expression in virally infected cell lines, we utilized a mixed population of peripheral blood mononuclear cells in order to capture the essential intercellular interactions that occur in vivo, and would otherwise be lost, using single cell lines or isolated primary cell subsets. In this mixed cell population we were able to detect expression of all annotated vaccinia genes. On the host side, a number of genes encoding cytokines, chemokines, complement factors and intracellular signaling molecules were downregulated upon viral infection, whereas genes encoding histone proteins and the interferon response were upregulated. We also identified a small number of genes that exhibited significantly different expression profiles in subjects with robust humoral immunity compared with those with weaker humoral responses. Our results provide evidence that differential gene regulation patterns may be at work in individuals with robust humoral immunity compared with those with weaker humoral immune responses. Genes and Immunity (2013) 14, 277–285; doi:10.1038/gene.2013.14; published online 18 April 2013 Keywords: Next-generation sequencing; mRNA-Seq; vaccinia virus; smallpox vaccine INTRODUCTION these 44 subjects had two samples (uninfected and vaccinia Vaccinia virus (VACV) is the immunologically cross-protective infected). -

Rap1-Mediated Chromatin and Gene Expression Changes at Senescence
University of Pennsylvania ScholarlyCommons Publicly Accessible Penn Dissertations 2019 Rap1-Mediated Chromatin And Gene Expression Changes At Senescence Shufei Song University of Pennsylvania Follow this and additional works at: https://repository.upenn.edu/edissertations Part of the Biochemistry Commons, and the Cell Biology Commons Recommended Citation Song, Shufei, "Rap1-Mediated Chromatin And Gene Expression Changes At Senescence" (2019). Publicly Accessible Penn Dissertations. 3557. https://repository.upenn.edu/edissertations/3557 This paper is posted at ScholarlyCommons. https://repository.upenn.edu/edissertations/3557 For more information, please contact [email protected]. Rap1-Mediated Chromatin And Gene Expression Changes At Senescence Abstract ABSTRACT RAP1-MEDIATED CHROMATIN AND GENE EXPRESSION CHANGES AT SENESCENCE The telomeric protein Rap1 has been extensively studied for its roles as a transcriptional activator and repressor. Indeed, in both yeast and mammals, Rap1 is known to bind throughout the genome to reorganize chromatin and regulate gene transcription. Previously, our lab published evidence that Rap1 plays important roles in cellular senescence. In telomerase-deficient S. cerevisiae, Rap1 relocalizes from telomeres and subtelomeres to new Rap1 target at senescence (NRTS). This leads to two types of histone loss: Rap1 lowers global histone levels by repressing histone gene transcription and it also results in local nucleosome displacement at the promoters of the activated NRTS. Here, I examine mechanisms of site-specific histone loss by presenting evidence that Rap1 can directly interact with histone tetramers H3/H4, and map this interaction to a three-amino-acid-patch within the DNA binding domain. Functional studies are performed in vivo using a mutant form of Rap1 with weakened histone interactions, and deficient promoter clearance as well as blunted gene activation is observed, indicating that direct Rap1-H3/H4 interactions are involved in nucleosome displacement. -

HIST1H2AK (HIST1H2AM) (NM 003514) Human Recombinant Protein Product Data
OriGene Technologies, Inc. 9620 Medical Center Drive, Ste 200 Rockville, MD 20850, US Phone: +1-888-267-4436 [email protected] EU: [email protected] CN: [email protected] Product datasheet for TP760612 HIST1H2AK (HIST1H2AM) (NM_003514) Human Recombinant Protein Product data: Product Type: Recombinant Proteins Description: Purified recombinant protein of Human histone cluster 1, H2am (HIST1H2AM), full length, with N-terminal HIS tag, expressed in E.Coli, 50ug Species: Human Expression Host: E. coli Tag: N-His Predicted MW: 13.9 kDa Concentration: >50 ug/mL as determined by microplate BCA method Purity: > 80% as determined by SDS-PAGE and Coomassie blue staining Buffer: 50mM Tris,8M Urea,pH8.0. Storage: Store at -80°C. Stability: Stable for 12 months from the date of receipt of the product under proper storage and handling conditions. Avoid repeated freeze-thaw cycles. RefSeq: NP_003505 Locus ID: 8336 UniProt ID: P0C0S8, A4FTV9 RefSeq Size: 487 Cytogenetics: 6p22.1 RefSeq ORF: 390 Synonyms: dJ193B12.1; H2A.1; H2A/n; H2AC11; H2AC13; H2AC15; H2AC16; H2AFN; HIST1H2AM This product is to be used for laboratory only. Not for diagnostic or therapeutic use. View online » ©2021 OriGene Technologies, Inc., 9620 Medical Center Drive, Ste 200, Rockville, MD 20850, US 1 / 2 HIST1H2AK (HIST1H2AM) (NM_003514) Human Recombinant Protein – TP760612 Summary: Histones are basic nuclear proteins that are responsible for the nucleosome structure of the chromosomal fiber in eukaryotes. Two molecules of each of the four core histones (H2A, H2B, H3, and H4) form an octamer, around which approximately 146 bp of DNA is wrapped in repeating units, called nucleosomes. -
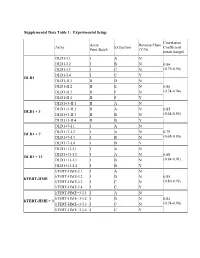
Table 3: Average Gene Expression Profiles by Chromosome
Supplemental Data Table 1: Experimental Setup Correlation Array Reverse Fluor Array Extraction Coefficient Print Batch (Y/N) mean (range) DLD1-I.1 I A N DLD1-I.2 I B N 0.86 DLD1-I.3 I C N (0.79-0.90) DLD1-I.4 I C Y DLD1 DLD1-II.1 II D N DLD1-II.2 II E N 0.86 DLD1-II.3 II F N (0.74-0.94) DLD1-II.4 II F Y DLD1+3-II.1 II A N DLD1+3-II.2 II A N 0.85 DLD1 + 3 DLD1+3-II.3 II B N (0.64-0.95) DLD1+3-II.4 II B Y DLD1+7-I.1 I A N DLD1+7-I.2 I A N 0.79 DLD1 + 7 DLD1+7-I.3 I B N (0.68-0.90) DLD1+7-I.4 I B Y DLD1+13-I.1 I A N DLD1+13-I.2 I A N 0.88 DLD1 + 13 DLD1+13-I.3 I B N (0.84-0.91) DLD1+13-I.4 I B Y hTERT-HME-I.1 I A N hTERT-HME-I.2 I B N 0.85 hTERT-HME hTERT-HME-I.3 I C N (0.80-0.92) hTERT-HME-I.4 I C Y hTERT-HME+3-I.1 I A N hTERT-HME+3-I.2 I B N 0.84 hTERT-HME + 3 hTERT-HME+3-I.3 I C N (0.74-0.90) hTERT-HME+3-I.4 I C Y Supplemental Data Table 2: Average gene expression profiles by chromosome arm DLD1 hTERT-HME Ratio.7 Ratio.1 Ratio.3 Ratio.3 Chrom.