Theoretical and Experimental Predictions of Neural Elements
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Brainstem: Midbrainmidbrain
Brainstem:Brainstem: MidbrainMidbrain 1.1. MidbrainMidbrain –– grossgross externalexternal anatomyanatomy 2.2. InternalInternal structurestructure ofof thethe midbrain:midbrain: cerebral peduncles tegmentum tectum (guadrigeminal plate) Midbrain MidbrainMidbrain –– generalgeneral featuresfeatures location – between forebrain and hindbrain the smallest region of the brainstem – 6-7g the shortest brainstem segment ~ 2 cm long least differentiated brainstem division human midbrain is archipallian – shared general architecture with the most ancient of vertebrates embryonic origin – mesencephalon main functions:functions a sort of relay station for sound and visual information serves as a nerve pathway of the cerebral hemispheres controls the eye movement involved in control of body movement Prof. Dr. Nikolai Lazarov 2 Midbrain MidbrainMidbrain –– grossgross anatomyanatomy dorsal part – tectum (quadrigeminal plate): superior colliculi inferior colliculi cerebral aqueduct ventral part – cerebral peduncles:peduncles dorsal – tegmentum (central part) ventral – cerebral crus substantia nigra Prof. Dr. Nikolai Lazarov 3 Midbrain CerebralCerebral cruscrus –– internalinternal structurestructure CerebralCerebral peduncle:peduncle: crus cerebri tegmentum mesencephali substantia nigra two thick semilunar white matter bundles composition – somatotopically arranged motor tracts: corticospinal } pyramidal tracts – medial ⅔ corticobulbar corticopontine fibers: frontopontine tracts – medially temporopontine tracts – laterally -

The Brain Stem Medulla Oblongata
Chapter 14 The Brain Stem Medulla Oblongata Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Central sulcus Parietal lobe • embryonic myelencephalon becomes Cingulate gyrus leaves medulla oblongata Corpus callosum Parieto–occipital sulcus Frontal lobe Occipital lobe • begins at foramen magnum of the skull Thalamus Habenula Anterior Epithalamus commissure Pineal gland • extends for about 3 cm rostrally and ends Hypothalamus Posterior commissure at a groove between the medulla and Optic chiasm Mammillary body pons Cerebral aqueduct Pituitary gland Fourth ventricle Temporal lobe • slightly wider than spinal cord Cerebellum Midbrain • pyramids – pair of external ridges on Pons Medulla anterior surface oblongata – resembles side-by-side baseball bats (a) • olive – a prominent bulge lateral to each pyramid • posteriorly, gracile and cuneate fasciculi of the spinal cord continue as two pair of ridges on the medulla • all nerve fibers connecting the brain to the spinal cord pass through the medulla • four pairs of cranial nerves begin or end in medulla - IX, X, XI, XII Medulla Oblongata Associated Functions • cardiac center – adjusts rate and force of heart • vasomotor center – adjusts blood vessel diameter • respiratory centers – control rate and depth of breathing • reflex centers – for coughing, sneezing, gagging, swallowing, vomiting, salivation, sweating, movements of tongue and head Medulla Oblongata Nucleus of hypoglossal nerve Fourth ventricle Gracile nucleus Nucleus of Cuneate nucleus vagus -

Sylvian Aqueduct Syndrome and Global Rostral Midbrain Dysfunction Associated with Shunt Malfunction
Sylvian aqueduct syndrome and global rostral midbrain dysfunction associated with shunt malfunction Giuseppe Cinalli, M.D., Christian Sainte-Rose, M.D., Isabelle Simon, M.D., Guillaume Lot, M.D., and Spiros Sgouros, M.D. Department of Pediatric Neurosurgery and Pediatric Radiology, Hôpital Necker•Enfants Malades, Université René Decartes; and Department of Neurosurgery, Hôpital Lariboisiere, Paris, France Object. This study is a retrospective analysis of clinical data obtained in 28 patients affected by obstructive hydrocephalus who presented with signs of midbrain dysfunction during episodes of shunt malfunction. Methods. All patients presented with an upward gaze palsy, sometimes associated with other signs of oculomotor dysfunction. In seven cases the ocular signs remained isolated and resolved rapidly after shunt revision. In 21 cases the ocular signs were variably associated with other clinical manifestations such as pyramidal and extrapyramidal deficits, memory disturbances, mutism, or alterations in consciousness. Resolution of these symptoms after shunt revision was usually slow. In four cases a transient paradoxical aggravation was observed at the time of shunt revision. In 11 cases ventriculocisternostomy allowed resolution of the symptoms and withdrawal of the shunt. Simultaneous supratentorial and infratentorial intracranial pressure recordings performed in seven of the patients showed a pressure gradient between the supratentorial and infratentorial compartments with a higher supratentorial pressure before shunt revision. Inversion of this pressure gradient was observed after shunt revision and resolution of the gradient was observed in one case after third ventriculostomy. In six recent cases, a focal midbrain hyperintensity was evidenced on T2-weighted magnetic resonance imaging sequences at the time of shunt malfunction. This rapidly resolved after the patient underwent third ventriculostomy. -

MRI Atlas of the Human Deep Brain Jean-Jacques Lemaire
MRI Atlas of the Human Deep Brain Jean-Jacques Lemaire To cite this version: Jean-Jacques Lemaire. MRI Atlas of the Human Deep Brain. 2019. hal-02116633 HAL Id: hal-02116633 https://hal.uca.fr/hal-02116633 Preprint submitted on 1 May 2019 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Distributed under a Creative Commons Attribution - NonCommercial - NoDerivatives| 4.0 International License MRI ATLAS of the HUMAN DEEP BRAIN Jean-Jacques Lemaire, MD, PhD, neurosurgeon, University Hospital of Clermont-Ferrand, Université Clermont Auvergne, CNRS, SIGMA, France This work is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/ or send a letter to Creative Commons, PO Box 1866, Mountain View, CA 94042, USA. Terminologia Foundational Model Terminologia MRI Deep Brain Atlas NeuroNames (ID) neuroanatomica usages, classical and french terminologies of Anatomy (ID) Anatomica 1998 (ID) 2017 http://fipat.library.dal.ca In -
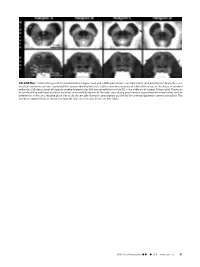
ON-LINE FIG 1. Selected Images of the Caudal Midbrain (Upper Row
ON-LINE FIG 1. Selected images of the caudal midbrain (upper row) and middle pons (lower row) from 4 of 13 total postmortem brains illustrate excellent anatomic contrast reproducibility across individual datasets. Subtle variations are present. Note differences in the shape of cerebral peduncles (24), decussation of superior cerebellar peduncles (25), and spinothalamic tract (12) in the midbrain of subject D (top right). These can be attributed to individual anatomic variation, some mild distortion of the brain stem during procurement at postmortem examination, and/or differences in the axial imaging plane not easily discernable during its prescription parallel to the anterior/posterior commissure plane. The numbers in parentheses in the on-line legends refer to structures in the On-line Table. AJNR Am J Neuroradiol ●:●●2019 www.ajnr.org E1 ON-LINE FIG 3. Demonstration of the dentatorubrothalamic tract within the superior cerebellar peduncle (asterisk) and rostral brain stem. A, Axial caudal midbrain image angled 10° anterosuperior to posteroinferior relative to the ACPC plane demonstrates the tract traveling the midbrain to reach the decussation (25). B, Coronal oblique image that is perpendicular to the long axis of the hippocam- pus (structure not shown) at the level of the ventral superior cerebel- lar decussation shows a component of the dentatorubrothalamic tract arising from the cerebellar dentate nucleus (63), ascending via the superior cerebellar peduncle to the decussation (25), and then enveloping the contralateral red nucleus (3). C, Parasagittal image shows the relatively long anteroposterior dimension of this tract, which becomes less compact and distinct as it ascends toward the thalamus. ON-LINE FIG 2. -

Neuroimaging Advances in Deep Brain Stimulation: Review of Indications, Anatomy, and Brain Connectomics
Published August 13, 2020 as 10.3174/ajnr.A6693 REVIEW ARTICLE Neuroimaging Advances in Deep Brain Stimulation: Review of Indications, Anatomy, and Brain Connectomics E.H. Middlebrooks, R.A. Domingo, T. Vivas-Buitrago, L. Okromelidze, T. Tsuboi, J.K. Wong, R.S. Eisinger, L. Almeida, M.R. Burns, A. Horn, R.J. Uitti, R.E. Wharen Jr, V.M. Holanda, and S.S. Grewal ABSTRACT SUMMARY: Deep brain stimulation is an established therapy for multiple brain disorders, with rapidly expanding potential indi- cations. Neuroimaging has advanced the field of deep brain stimulation through improvements in delineation of anatomy, and, more recently, application of brain connectomics. Older lesion-derived, localizationist theories of these conditions have evolved to newer, network-based “circuitopathies,” aided by the ability to directly assess these brain circuits in vivo through the use of advanced neuroimaging techniques, such as diffusion tractography and fMRI. In this review, we use a combination of ultra-high-field MR imaging and diffusion tractography to highlight relevant anatomy for the currently approved indications for deep brain stimulation in the United States: essential tremor, Parkinson disease, drug-resistant epilepsy, dystonia, and obsessive-compulsive disorder. We also review the literature regarding the use of fMRI and diffusion tractography in under- standing the role of deep brain stimulation in these disorders, as well as their potential use in both surgical targeting and de- vice programming. ABBREVIATIONS: AL ¼ ansa lenticularis; ALIC -

High-Yield Neuroanatomy
LWBK110-3895G-FM[i-xviii].qxd 8/14/08 5:57 AM Page i Aptara Inc. High-Yield TM Neuroanatomy FOURTH EDITION LWBK110-3895G-FM[i-xviii].qxd 8/14/08 5:57 AM Page ii Aptara Inc. LWBK110-3895G-FM[i-xviii].qxd 8/14/08 5:57 AM Page iii Aptara Inc. High-Yield TM Neuroanatomy FOURTH EDITION James D. Fix, PhD Professor Emeritus of Anatomy Marshall University School of Medicine Huntington, West Virginia With Contributions by Jennifer K. Brueckner, PhD Associate Professor Assistant Dean for Student Affairs Department of Anatomy and Neurobiology University of Kentucky College of Medicine Lexington, Kentucky LWBK110-3895G-FM[i-xviii].qxd 8/14/08 5:57 AM Page iv Aptara Inc. Acquisitions Editor: Crystal Taylor Managing Editor: Kelley Squazzo Marketing Manager: Emilie Moyer Designer: Terry Mallon Compositor: Aptara Fourth Edition Copyright © 2009, 2005, 2000, 1995 Lippincott Williams & Wilkins, a Wolters Kluwer business. 351 West Camden Street 530 Walnut Street Baltimore, MD 21201 Philadelphia, PA 19106 Printed in the United States of America. All rights reserved. This book is protected by copyright. No part of this book may be reproduced or transmitted in any form or by any means, including as photocopies or scanned-in or other electronic copies, or utilized by any information storage and retrieval system without written permission from the copyright owner, except for brief quotations embodied in critical articles and reviews. Materials appearing in this book prepared by individuals as part of their official duties as U.S. government employees are not covered by the above-mentioned copyright. To request permission, please contact Lippincott Williams & Wilkins at 530 Walnut Street, Philadelphia, PA 19106, via email at [email protected], or via website at http://www.lww.com (products and services). -

Split Cerebral Aqueduct: a Neuroendoscopic Illustration
Split cerebral aqueduct: a neuroendoscopic illustration Alberto Feletti, Alessandro Fiorindi & Pierluigi Longatti Child's Nervous System ISSN 0256-7040 Volume 32 Number 1 Childs Nerv Syst (2016) 32:199-203 DOI 10.1007/s00381-015-2827-y 1 23 Your article is protected by copyright and all rights are held exclusively by Springer- Verlag Berlin Heidelberg. This e-offprint is for personal use only and shall not be self- archived in electronic repositories. If you wish to self-archive your article, please use the accepted manuscript version for posting on your own website. You may further deposit the accepted manuscript version in any repository, provided it is only made publicly available 12 months after official publication or later and provided acknowledgement is given to the original source of publication and a link is inserted to the published article on Springer's website. The link must be accompanied by the following text: "The final publication is available at link.springer.com”. 1 23 Author's personal copy Childs Nerv Syst (2016) 32:199–203 DOI 10.1007/s00381-015-2827-y CASE REPORT Split cerebral aqueduct: a neuroendoscopic illustration Alberto Feletti1 & Alessandro Fiorindi2 & Pierluigi Longatti2 Received: 30 June 2015 /Accepted: 9 July 2015 /Published online: 1 August 2015 # Springer-Verlag Berlin Heidelberg 2015 Abstract Introduction Purpose Forking of the cerebral aqueduct is a developmental malformation that is infrequently encountered by neurosur- Although some authors have described the forking of the ce- geons as a rare cause of hydrocephalus, sometimes with a rebral aqueduct (CA) as a common cause of hydrocephalus, a delayed onset. -

Motor Systems Basal Ganglia
Motor systems 409 Basal Ganglia You have just read about the different motor-related cortical areas. Premotor areas are involved in planning, while MI is involved in execution. What you don’t know is that the cortical areas involved in movement control need “help” from other brain circuits in order to smoothly orchestrate motor behaviors. One of these circuits involves a group of structures deep in the brain called the basal ganglia. While their exact motor function is still debated, the basal ganglia clearly regulate movement. Without information from the basal ganglia, the cortex is unable to properly direct motor control, and the deficits seen in Parkinson’s and Huntington’s disease and related movement disorders become apparent. Let’s start with the anatomy of the basal ganglia. The important “players” are identified in the adjacent figure. The caudate and putamen have similar functions, and we will consider them as one in this discussion. Together the caudate and putamen are called the neostriatum or simply striatum. All input to the basal ganglia circuit comes via the striatum. This input comes mainly from motor cortical areas. Notice that the caudate (L. tail) appears twice in many frontal brain sections. This is because the caudate curves around with the lateral ventricle. The head of the caudate is most anterior. It gives rise to a body whose “tail” extends with the ventricle into the temporal lobe (the “ball” at the end of the tail is the amygdala, whose limbic functions you will learn about later). Medial to the putamen is the globus pallidus (GP). -

High-Yield Neuroanatomy, FOURTH EDITION
LWBK110-3895G-FM[i-xviii].qxd 8/14/08 5:57 AM Page i Aptara Inc. High-Yield TM Neuroanatomy FOURTH EDITION LWBK110-3895G-FM[i-xviii].qxd 8/14/08 5:57 AM Page ii Aptara Inc. LWBK110-3895G-FM[i-xviii].qxd 8/14/08 5:57 AM Page iii Aptara Inc. High-Yield TM Neuroanatomy FOURTH EDITION James D. Fix, PhD Professor Emeritus of Anatomy Marshall University School of Medicine Huntington, West Virginia With Contributions by Jennifer K. Brueckner, PhD Associate Professor Assistant Dean for Student Affairs Department of Anatomy and Neurobiology University of Kentucky College of Medicine Lexington, Kentucky LWBK110-3895G-FM[i-xviii].qxd 8/14/08 5:57 AM Page iv Aptara Inc. Acquisitions Editor: Crystal Taylor Managing Editor: Kelley Squazzo Marketing Manager: Emilie Moyer Designer: Terry Mallon Compositor: Aptara Fourth Edition Copyright © 2009, 2005, 2000, 1995 Lippincott Williams & Wilkins, a Wolters Kluwer business. 351 West Camden Street 530 Walnut Street Baltimore, MD 21201 Philadelphia, PA 19106 Printed in the United States of America. All rights reserved. This book is protected by copyright. No part of this book may be reproduced or transmitted in any form or by any means, including as photocopies or scanned-in or other electronic copies, or utilized by any information storage and retrieval system without written permission from the copyright owner, except for brief quotations embodied in critical articles and reviews. Materials appearing in this book prepared by individuals as part of their official duties as U.S. government employees are not covered by the above-mentioned copyright. To request permission, please contact Lippincott Williams & Wilkins at 530 Walnut Street, Philadelphia, PA 19106, via email at [email protected], or via website at http://www.lww.com (products and services). -

…Going One Step Further
…going one step further C20 (1017868) 2 Latin A Encephalon Mesencephalon B Telencephalon 31 Lamina tecti B1 Lobus frontalis 32 Tegmentum mesencephali B2 Lobus temporalis 33 Crus cerebri C Diencephalon 34 Aqueductus mesencephali D Mesencephalon E Metencephalon Metencephalon E1 Cerebellum 35 Cerebellum F Myelencephalon a Vermis G Circulus arteriosus cerebri (Willisii) b Tonsilla c Flocculus Telencephalon d Arbor vitae 1 Lobus frontalis e Ventriculus quartus 2 Lobus parietalis 36 Pons 3 Lobus occipitalis f Pedunculus cerebellaris superior 4 Lobus temporalis g Pedunculus cerebellaris medius 5 Sulcus centralis h Pedunculus cerebellaris inferior 6 Gyrus precentralis 7 Gyrus postcentralis Myelencephalon 8 Bulbus olfactorius 37 Medulla oblongata 9 Commissura anterior 38 Oliva 10 Corpus callosum 39 Pyramis a Genu 40 N. cervicalis I. (C1) b Truncus ® c Splenium Nervi craniales d Rostrum I N. olfactorius 11 Septum pellucidum II N. opticus 12 Fornix III N. oculomotorius 13 Commissura posterior IV N. trochlearis 14 Insula V N. trigeminus 15 Capsula interna VI N. abducens 16 Ventriculus lateralis VII N. facialis e Cornu frontale VIII N. vestibulocochlearis f Pars centralis IX N. glossopharyngeus g Cornu occipitale X N. vagus h Cornu temporale XI N. accessorius 17 V. thalamostriata XII N. hypoglossus 18 Hippocampus Circulus arteriosus cerebri (Willisii) Diencephalon 1 A. cerebri anterior 19 Thalamus 2 A. communicans anterior 20 Sulcus hypothalamicus 3 A. carotis interna 21 Hypothalamus 4 A. cerebri media 22 Adhesio interthalamica 5 A. communicans posterior 23 Glandula pinealis 6 A. cerebri posterior 24 Corpus mammillare sinistrum 7 A. superior cerebelli 25 Hypophysis 8 A. basilaris 26 Ventriculus tertius 9 Aa. pontis 10 A. -

Chapter 3: Internal Anatomy of the Central Nervous System
10353-03_CH03.qxd 8/30/07 1:12 PM Page 82 3 Internal Anatomy of the Central Nervous System LEARNING OBJECTIVES Nuclear structures and fiber tracts related to various functional systems exist side by side at each level of the After studying this chapter, students should be able to: nervous system. Because disease processes in the brain • Identify the shapes of corticospinal fibers at different rarely strike only one anatomic structure or pathway, there neuraxial levels is a tendency for a series of related and unrelated clinical symptoms to emerge after a brain injury. A thorough knowl- • Recognize the ventricular cavity at various neuroaxial edge of the internal brain structures, including their shape, levels size, location, and proximity, makes it easier to understand • Recognize major internal anatomic structures of the their functional significance. In addition, the proximity of spinal cord and describe their functions nuclear structures and fiber tracts explains multiple symp- toms that may develop from a single lesion site. • Recognize important internal anatomic structures of the medulla and explain their functions • Recognize important internal anatomic structures of the ANATOMIC ORIENTATION pons and describe their functions LANDMARKS • Identify important internal anatomic structures of the midbrain and discuss their functions Two distinct anatomic landmarks used for visual orientation to the internal anatomy of the brain are the shapes of the • Recognize important internal anatomic structures of the descending corticospinal fibers and the ventricular cavity forebrain (diencephalon, basal ganglia, and limbic (Fig. 3-1). Both are present throughout the brain, although structures) and describe their functions their shape and size vary as one progresses caudally from the • Follow the continuation of major anatomic structures rostral forebrain (telencephalon) to the caudal brainstem.