Disentangling the Roles of Form and Motion in Fish Swimming Performance
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Dissertação Final.Pdf
INSTITUTO NACIONAL DE PESQUISAS DA AMAZÔNIA – INPA PROGRAMA DE PÓS-GRADUAÇÃO EM BIOLOGIA DE ÁGUA DOCE E PESCA INTERIOR - BADPI CARACTERIZAÇÃO CITOGENÉTICA EM ESPÉCIES DE Cichla BLOCH & SCHNEIDER, 1801 (PERCIFORMES, CICHLIDAE) COM ÊNFASE NOS HÍBRIDOS INTERESPECÍFICOS JANICE MACHADO DE QUADROS MANAUS-AM 2019 JANICE MACHADO DE QUADROS CARACTERIZAÇÃO CITOGENÉTICA EM ESPÉCIES DE Cichla BLOCH & SCHNEIDER, 1801 (PERCIFORMES, CICHLIDAE) COM ÊNFASE NOS HÍBRIDOS INTERESPECÍFICOS ORIENTADORA: Eliana Feldberg, Dra. COORIENTADOR: Efrem Jorge Gondim Ferreira, Dr. Dissertação apresentada ao Programa de Pós- Graduação do Instituto Nacional de Pesquisas da Amazônia como parte dos requisitos para obtenção do título de Mestre em CIÊNCIAS BIOLÓGICAS, área de concentração Biologia de Água Doce e Pesca Interior. MANAUS-AM 2019 Ficha Catalográfica Q1c Quadros, Janice Machado de CARACTERIZAÇÃO CITOGENÉTICA EM ESPÉCIES DE Cichla BLOCH & SCHNEIDER, 1801 (PERCIFORMES, CICHLIDAE) COM ÊNFASE NOS HÍBRIDOS INTERESPECÍFICOS / Janice Machado de Quadros; orientadora Eliana Feldberg; coorientadora Efrem Ferreira. -- Manaus:[s.l], 2019. 56 f. Dissertação (Mestrado - Programa de Pós Graduação em Biologia de Água Doce e Pesca Interior) -- Coordenação do Programa de Pós- Graduação, INPA, 2019. 1. Hibridação. 2. Tucunaré. 3. Heterocromatina. 4. FISH. 5. DNAr. I. Feldberg, Eliana, orient. II. Ferreira, Efrem, coorient. III. Título. CDD: 639.2 Sinopse: A fim de investigar a existência de híbridos entre espécies de Cichla, nós analisamos, por meio de técnicas de citogenética clássica e molecular indivíduos de C. monoculus, C. temensis, C. pinima, C. kelberi, C. piquiti, C. vazzoleri de diferentes locais da bacia amazônica e entre eles, três indivíduos foram considerados, morfologicamente, híbridos. Todos os indivíduos apresentaram 2n=48 cromossomos acrocêntricos. A heterocromatina foi encontrada preferencialmente em regiões centroméricas e terminais, com variações entre indivíduos de C. -

§4-71-6.5 LIST of CONDITIONALLY APPROVED ANIMALS November
§4-71-6.5 LIST OF CONDITIONALLY APPROVED ANIMALS November 28, 2006 SCIENTIFIC NAME COMMON NAME INVERTEBRATES PHYLUM Annelida CLASS Oligochaeta ORDER Plesiopora FAMILY Tubificidae Tubifex (all species in genus) worm, tubifex PHYLUM Arthropoda CLASS Crustacea ORDER Anostraca FAMILY Artemiidae Artemia (all species in genus) shrimp, brine ORDER Cladocera FAMILY Daphnidae Daphnia (all species in genus) flea, water ORDER Decapoda FAMILY Atelecyclidae Erimacrus isenbeckii crab, horsehair FAMILY Cancridae Cancer antennarius crab, California rock Cancer anthonyi crab, yellowstone Cancer borealis crab, Jonah Cancer magister crab, dungeness Cancer productus crab, rock (red) FAMILY Geryonidae Geryon affinis crab, golden FAMILY Lithodidae Paralithodes camtschatica crab, Alaskan king FAMILY Majidae Chionocetes bairdi crab, snow Chionocetes opilio crab, snow 1 CONDITIONAL ANIMAL LIST §4-71-6.5 SCIENTIFIC NAME COMMON NAME Chionocetes tanneri crab, snow FAMILY Nephropidae Homarus (all species in genus) lobster, true FAMILY Palaemonidae Macrobrachium lar shrimp, freshwater Macrobrachium rosenbergi prawn, giant long-legged FAMILY Palinuridae Jasus (all species in genus) crayfish, saltwater; lobster Panulirus argus lobster, Atlantic spiny Panulirus longipes femoristriga crayfish, saltwater Panulirus pencillatus lobster, spiny FAMILY Portunidae Callinectes sapidus crab, blue Scylla serrata crab, Samoan; serrate, swimming FAMILY Raninidae Ranina ranina crab, spanner; red frog, Hawaiian CLASS Insecta ORDER Coleoptera FAMILY Tenebrionidae Tenebrio molitor mealworm, -

Fish, Various Invertebrates
Zambezi Basin Wetlands Volume II : Chapters 7 - 11 - Contents i Back to links page CONTENTS VOLUME II Technical Reviews Page CHAPTER 7 : FRESHWATER FISHES .............................. 393 7.1 Introduction .................................................................... 393 7.2 The origin and zoogeography of Zambezian fishes ....... 393 7.3 Ichthyological regions of the Zambezi .......................... 404 7.4 Threats to biodiversity ................................................... 416 7.5 Wetlands of special interest .......................................... 432 7.6 Conservation and future directions ............................... 440 7.7 References ..................................................................... 443 TABLE 7.2: The fishes of the Zambezi River system .............. 449 APPENDIX 7.1 : Zambezi Delta Survey .................................. 461 CHAPTER 8 : FRESHWATER MOLLUSCS ................... 487 8.1 Introduction ................................................................. 487 8.2 Literature review ......................................................... 488 8.3 The Zambezi River basin ............................................ 489 8.4 The Molluscan fauna .................................................. 491 8.5 Biogeography ............................................................... 508 8.6 Biomphalaria, Bulinis and Schistosomiasis ................ 515 8.7 Conservation ................................................................ 516 8.8 Further investigations ................................................. -

Consistency of Individual Differences in Behaviour of the Lion-Headed Cichlid, Steatocranus Casuarius
Behavioural Processes 48 (1999) 49–55 www.elsevier.com/locate/behavproc Consistency of individual differences in behaviour of the lion-headed cichlid, Steatocranus casuarius Sergey V. Budaev *, Dmitry D. Zworykin, Andrei D. Mochek A.N. Se6ertso6 Institute of Ecology and E6olution, Russian Academy of Sciences, Leninsky prospect 33, 117071 Moscow, Russia Received 29 April 1999; received in revised form 9 September 1999; accepted 14 September 1999 Abstract The development of individual differences in behaviour in a novel environment, in the presence of a strange fish and during aggressive interactions with a mirror-image was studied in the lion-headed cichlid (Steatocranus casuarius, Teleostei, Cichlidae). No consistency in behaviour was found at 4–5.5 months of age. However, behaviours scored in situations involving a discrete source of stress (a strange fish or conspecific) become significantly consistent at the age of 12 months. At 4–5.5 but not 12 months of age, larger individuals approached and attacked the strange fish significantly more than smaller ones. These patterns may be associated with development and integration of motivational systems and alternative coping strategies. © 1999 Elsevier Science B.V. All rights reserved. Keywords: Aggression; Consistency; Individual differences; Exploratory behaviour; Temperament 1. Introduction ysis of the development of individuality. More- over, while various aspects of its ontogeny have Individual differences and alternative strategies been studied in mammals (e.g. Fox, 1972; Mac- have been documented in animals of many spe- Donald, 1983; Stevenson-Hinde, 1983; Lyons et cies. It is known that they may be adaptive and al., 1988; Loughry and Lazari, 1994), the develop- arise within the population for a variety of rea- ment of individual differences in behaviour, espe- sons (Slater, 1981; Clark and Ehlinger, 1987; cially in consistent behavioural traits, is almost an Magurran, 1993; Wilson et al., 1994). -

AN ECOLOGICAL and SYSTEMATIC SURVEY of FISHES in the RAPIDS of the LOWER ZA.Fre OR CONGO RIVER
AN ECOLOGICAL AND SYSTEMATIC SURVEY OF FISHES IN THE RAPIDS OF THE LOWER ZA.fRE OR CONGO RIVER TYSON R. ROBERTS1 and DONALD J. STEWART2 CONTENTS the rapids habitats, and the adaptations and mode of reproduction of the fishes discussed. Abstract ______________ ----------------------------------------------- 239 Nineteen new species are described from the Acknowledgments ----------------------------------- 240 Lower Zaire rapids, belonging to the genera Introduction _______________________________________________ 240 Mormyrus, Alestes, Labeo, Bagrus, Chrysichthys, Limnology ---------------------------------------------------------- 242 Notoglanidium, Gymnallabes, Chiloglanis, Lampro Collecting Methods and Localities __________________ 244 logus, Nanochromis, Steatocranus, Teleogramma, Tabulation of species ---------------------------------------- 249 and Mastacembelus, most of them with obvious Systematics -------------------------------------------------------- 249 modifications for life in the rapids. Caecomasta Campylomormyrus _______________ 255 cembelus is placed in the synonymy of Mastacem M ormyrus ____ --------------------------------- _______________ 268 belus, and morphologically intermediate hybrids Alestes __________________ _________________ 270 reported between blind, depigmented Mastacem Bryconaethiops -------------------------------------------- 271 belus brichardi and normally eyed, darkly pig Labeo ---------------------------------------------------- _______ 274 mented M astacembelus brachyrhinus. The genera Bagrus -

A New Colorful Species of Geophagus (Teleostei: Cichlidae), Endemic to the Rio Aripuanã in the Amazon Basin of Brazil
Neotropical Ichthyology, 12(4): 737-746, 2014 Copyright © 2014 Sociedade Brasileira de Ictiologia DOI: 10.1590/1982-0224-20140038 A new colorful species of Geophagus (Teleostei: Cichlidae), endemic to the rio Aripuanã in the Amazon basin of Brazil Gabriel C. Deprá1, Sven O. Kullander2, Carla S. Pavanelli1,3 and Weferson J. da Graça4 Geophagus mirabilis, new species, is endemic to the rio Aripuanã drainage upstream from Dardanelos/Andorinhas falls. The new species is distinguished from all other species of the genus by the presence of one to five large black spots arranged longitudinally along the middle of the flank, in addition to the black midlateral spot that is characteristic of species in the genus and by a pattern of iridescent spots and lines on the head in living specimens. It is further distinguished from all congeneric species, except G. camopiensis and G. crocatus, by the presence of seven (vs. eight or more) scale rows in the circumpeduncular series below the lateral line (7 in G. crocatus; 7-9 in G. camopiensis). Including the new species, five cichlids and 11 fish species in total are known only from the upper rio Aripuanã, and 15 fish species in total are known only from the rio Aripuanã drainage. Geophagus mirabilis, espécie nova, é endêmica da drenagem do rio Aripuanã, a montante das quedas de Dardanelos/ Andorinhas. A espécie nova se distingue de todas as outras espécies do gênero pela presença de uma a cinco manchas pretas grandes distribuídas longitudinalmente ao longo do meio do flanco, em adição à mancha preta no meio do flanco característica das espécies do gênero, e por um padrão de pontos e linhas iridescentes sobre a cabeça em espécimes vivos. -

Summary Report of Freshwater Nonindigenous Aquatic Species in U.S
Summary Report of Freshwater Nonindigenous Aquatic Species in U.S. Fish and Wildlife Service Region 4—An Update April 2013 Prepared by: Pam L. Fuller, Amy J. Benson, and Matthew J. Cannister U.S. Geological Survey Southeast Ecological Science Center Gainesville, Florida Prepared for: U.S. Fish and Wildlife Service Southeast Region Atlanta, Georgia Cover Photos: Silver Carp, Hypophthalmichthys molitrix – Auburn University Giant Applesnail, Pomacea maculata – David Knott Straightedge Crayfish, Procambarus hayi – U.S. Forest Service i Table of Contents Table of Contents ...................................................................................................................................... ii List of Figures ............................................................................................................................................ v List of Tables ............................................................................................................................................ vi INTRODUCTION ............................................................................................................................................. 1 Overview of Region 4 Introductions Since 2000 ....................................................................................... 1 Format of Species Accounts ...................................................................................................................... 2 Explanation of Maps ................................................................................................................................ -

Species Composition and Invasion Risks of Alien Ornamental Freshwater
www.nature.com/scientificreports OPEN Species composition and invasion risks of alien ornamental freshwater fshes from pet stores in Klang Valley, Malaysia Abdulwakil Olawale Saba1,2, Ahmad Ismail1, Syaizwan Zahmir Zulkifi1, Muhammad Rasul Abdullah Halim3, Noor Azrizal Abdul Wahid4 & Mohammad Noor Azmai Amal1* The ornamental fsh trade has been considered as one of the most important routes of invasive alien fsh introduction into native freshwater ecosystems. Therefore, the species composition and invasion risks of fsh species from 60 freshwater fsh pet stores in Klang Valley, Malaysia were studied. A checklist of taxa belonging to 18 orders, 53 families, and 251 species of alien fshes was documented. Fish Invasiveness Screening Test (FIST) showed that seven (30.43%), eight (34.78%) and eight (34.78%) species were considered to be high, medium and low invasion risks, respectively. After the calibration of the Fish Invasiveness Screening Kit (FISK) v2 using the Receiver Operating Characteristics, a threshold value of 17 for distinguishing between invasive and non-invasive fshes was identifed. As a result, nine species (39.13%) were of high invasion risk. In this study, we found that non-native fshes dominated (85.66%) the freshwater ornamental trade in Klang Valley, while FISK is a more robust tool in assessing the risk of invasion, and for the most part, its outcome was commensurate with FIST. This study, for the frst time, revealed the number of high-risk ornamental fsh species that give an awareness of possible future invasion if unmonitored in Klang Valley, Malaysia. As a global hobby, fshkeeping is cherished by both young and old people. -

View/Download
CICHLIFORMES: Cichlidae (part 5) · 1 The ETYFish Project © Christopher Scharpf and Kenneth J. Lazara COMMENTS: v. 10.0 - 11 May 2021 Order CICHLIFORMES (part 5 of 8) Family CICHLIDAE Cichlids (part 5 of 7) Subfamily Pseudocrenilabrinae African Cichlids (Palaeoplex through Yssichromis) Palaeoplex Schedel, Kupriyanov, Katongo & Schliewen 2020 palaeoplex, a key concept in geoecodynamics representing the total genomic variation of a given species in a given landscape, the analysis of which theoretically allows for the reconstruction of that species’ history; since the distribution of P. palimpsest is tied to an ancient landscape (upper Congo River drainage, Zambia), the name refers to its potential to elucidate the complex landscape evolution of that region via its palaeoplex Palaeoplex palimpsest Schedel, Kupriyanov, Katongo & Schliewen 2020 named for how its palaeoplex (see genus) is like a palimpsest (a parchment manuscript page, common in medieval times that has been overwritten after layers of old handwritten letters had been scraped off, in which the old letters are often still visible), revealing how changes in its landscape and/or ecological conditions affected gene flow and left genetic signatures by overwriting the genome several times, whereas remnants of more ancient genomic signatures still persist in the background; this has led to contrasting hypotheses regarding this cichlid’s phylogenetic position Pallidochromis Turner 1994 pallidus, pale, referring to pale coloration of all specimens observed at the time; chromis, a name -

28. Annexe 2 Haplochromis
Description de ‘Haplochromis’ snoeksi ‘Haplochromis’ snoeksi (Perciformes: Cichlidae) a new species from the Inkisi River basin, Lower Congo Soleil WAMUINI L. *, ** Emmanuel VREVEN *** and Pierre VANDEWALLE** * I.S.P. Mbanza-Ngungu, Département de Biologie, B.P. 127 - Mbanza-Ngungu, Democratic Republic of Congo (DRC). E-mail: [email protected] ** Université de Liège, Faculté des Sciences, Laboratoire de Morphologie fonctionnelle et évolutive, B-4000 Liège, Belgium. E-mail: [email protected] *** Royal Museum for Central Africa, Vertebrate Section, Ichthyology, B-3080 Tervuren, Belgium; E-mail: [email protected] Abstract ‘Haplochromis’ snoeksi , new species, is described from a single locality, Ngeba village on the Ngeba /Ngufu River, a right bank affluent of the Inkisi River basin (Lower Congo; Democratic Republic of Congo). The species is distinguished from its riverine Congo River basin congeners by the following unique combination of characters: thorax partially naked, i.e. without small scales; 16 scales around caudal peduncle; and an upper lateral line with 23 scales. Résumé Une nouvelle espèce, ‘ Haplochromis ’ snoeksi , est décrite d’une seule localité, le village Ngeba sur la rivière Ngeba/Ngufu, un affluent de la rive droite de l’Inkisi (Bas-Congo; République Démocratique du Congo). Cette espèce se distingue des ses congénères des rivières de la province ichtyologiques du bassin du Congo par la combinaison des caractères suivants : un espace thoracique partiellement nu, c'est-à-dire dépourvu d’écailles, 16 écailles autour du pédoncule caudal et une ligne latéral supérieure avec 23 écailles. Introduction The family Cichlidae is characterized by a single nostril on either side of the head, a lateral line which is divided in two parts and a single continuous dorsal fin which has a spiny anterior and soft posterior fin-ray part (Snoeks, 1994; Nelson, 2006; Stiassny et al ., 2008). -

Nabs 2004 Final
CURRENT AND SELECTED BIBLIOGRAPHIES ON BENTHIC BIOLOGY 2004 Published August, 2005 North American Benthological Society 2 FOREWORD “Current and Selected Bibliographies on Benthic Biology” is published annu- ally for the members of the North American Benthological Society, and summarizes titles of articles published during the previous year. Pertinent titles prior to that year are also included if they have not been cited in previous reviews. I wish to thank each of the members of the NABS Literature Review Committee for providing bibliographic information for the 2004 NABS BIBLIOGRAPHY. I would also like to thank Elizabeth Wohlgemuth, INHS Librarian, and library assis- tants Anna FitzSimmons, Jessica Beverly, and Elizabeth Day, for their assistance in putting the 2004 bibliography together. Membership in the North American Benthological Society may be obtained by contacting Ms. Lucinda B. Johnson, Natural Resources Research Institute, Uni- versity of Minnesota, 5013 Miller Trunk Highway, Duluth, MN 55811. Phone: 218/720-4251. email:[email protected]. Dr. Donald W. Webb, Editor NABS Bibliography Illinois Natural History Survey Center for Biodiversity 607 East Peabody Drive Champaign, IL 61820 217/333-6846 e-mail: [email protected] 3 CONTENTS PERIPHYTON: Christine L. Weilhoefer, Environmental Science and Resources, Portland State University, Portland, O97207.................................5 ANNELIDA (Oligochaeta, etc.): Mark J. Wetzel, Center for Biodiversity, Illinois Natural History Survey, 607 East Peabody Drive, Champaign, IL 61820.................................................................................................................6 ANNELIDA (Hirudinea): Donald J. Klemm, Ecosystems Research Branch (MS-642), Ecological Exposure Research Division, National Exposure Re- search Laboratory, Office of Research & Development, U.S. Environmental Protection Agency, 26 W. Martin Luther King Dr., Cincinnati, OH 45268- 0001 and William E. -
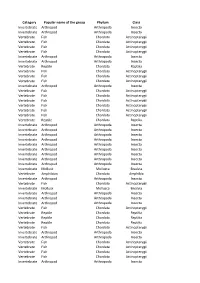
Category Popular Name of the Group Phylum Class Invertebrate
Category Popular name of the group Phylum Class Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Vertebrate Reptile Chordata Reptilia Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Invertebrate Arthropod Arthropoda Insecta Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Reptile Chordata Reptilia Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Mollusk Mollusca Bivalvia Vertebrate Amphibian Chordata Amphibia Invertebrate Arthropod Arthropoda Insecta Vertebrate Fish Chordata Actinopterygii Invertebrate Mollusk Mollusca Bivalvia Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Vertebrate