HNRPH3 Antibody Cat
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Genetic and Pharmacological Approaches to Preventing Neurodegeneration
University of Pennsylvania ScholarlyCommons Publicly Accessible Penn Dissertations 2012 Genetic and Pharmacological Approaches to Preventing Neurodegeneration Marco Boccitto University of Pennsylvania, [email protected] Follow this and additional works at: https://repository.upenn.edu/edissertations Part of the Neuroscience and Neurobiology Commons Recommended Citation Boccitto, Marco, "Genetic and Pharmacological Approaches to Preventing Neurodegeneration" (2012). Publicly Accessible Penn Dissertations. 494. https://repository.upenn.edu/edissertations/494 This paper is posted at ScholarlyCommons. https://repository.upenn.edu/edissertations/494 For more information, please contact [email protected]. Genetic and Pharmacological Approaches to Preventing Neurodegeneration Abstract The Insulin/Insulin-like Growth Factor 1 Signaling (IIS) pathway was first identified as a major modifier of aging in C.elegans. It has since become clear that the ability of this pathway to modify aging is phylogenetically conserved. Aging is a major risk factor for a variety of neurodegenerative diseases including the motor neuron disease, Amyotrophic Lateral Sclerosis (ALS). This raises the possibility that the IIS pathway might have therapeutic potential to modify the disease progression of ALS. In a C. elegans model of ALS we found that decreased IIS had a beneficial effect on ALS pathology in this model. This beneficial effect was dependent on activation of the transcription factor daf-16. To further validate IIS as a potential therapeutic target for treatment of ALS, manipulations of IIS in mammalian cells were investigated for neuroprotective activity. Genetic manipulations that increase the activity of the mammalian ortholog of daf-16, FOXO3, were found to be neuroprotective in a series of in vitro models of ALS toxicity. -

Apoptotic Genes As Potential Markers of Metastatic Phenotype in Human Osteosarcoma Cell Lines
17-31 10/12/07 14:53 Page 17 INTERNATIONAL JOURNAL OF ONCOLOGY 32: 17-31, 2008 17 Apoptotic genes as potential markers of metastatic phenotype in human osteosarcoma cell lines CINZIA ZUCCHINI1, ANNA ROCCHI2, MARIA CRISTINA MANARA2, PAOLA DE SANCTIS1, CRISTINA CAPANNI3, MICHELE BIANCHINI1, PAOLO CARINCI1, KATIA SCOTLANDI2 and LUISA VALVASSORI1 1Dipartimento di Istologia, Embriologia e Biologia Applicata, Università di Bologna, Via Belmeloro 8, 40126 Bologna; 2Laboratorio di Ricerca Oncologica, Istituti Ortopedici Rizzoli; 3IGM-CNR, Unit of Bologna, c/o Istituti Ortopedici Rizzoli, Via di Barbiano 1/10, 40136 Bologna, Italy Received May 29, 2007; Accepted July 19, 2007 Abstract. Metastasis is the most frequent cause of death among malignant primitive bone tumor, usually developing in children patients with osteosarcoma. We have previously demonstrated and adolescents, with a high tendency to metastasize (2). in independent experiments that the forced expression of Metastases in osteosarcoma patients spread through peripheral L/B/K ALP and CD99 in U-2 OS osteosarcoma cell lines blood very early and colonize primarily the lung, and later markedly reduces the metastatic ability of these cancer cells. other skeleton districts (3). Since disseminated hidden micro- This behavior makes these cell lines a useful model to assess metastases are present in 80-90% of OS patients at the time the intersection of multiple and independent gene expression of diagnosis, the identification of markers of invasiveness signatures concerning the biological problem of dissemination. and metastasis forms a target of paramount importance in With the aim to characterize a common transcriptional profile planning the treatment of osteosarcoma lesions and enhancing reflecting the essential features of metastatic behavior, we the prognosis. -

Mouse Hnrnph3 Knockout Project (CRISPR/Cas9)
https://www.alphaknockout.com Mouse Hnrnph3 Knockout Project (CRISPR/Cas9) Objective: To create a Hnrnph3 knockout Mouse model (C57BL/6J) by CRISPR/Cas-mediated genome engineering. Strategy summary: The Hnrnph3 gene (NCBI Reference Sequence: NM_001079824 ; Ensembl: ENSMUSG00000020069 ) is located on Mouse chromosome 10. 10 exons are identified, with the ATG start codon in exon 2 and the TGA stop codon in exon 10 (Transcript: ENSMUST00000020263). Exon 2~3 will be selected as target site. Cas9 and gRNA will be co-injected into fertilized eggs for KO Mouse production. The pups will be genotyped by PCR followed by sequencing analysis. Note: Exon 2 starts from the coding region. Exon 2~3 covers 24.18% of the coding region. The size of effective KO region: ~768 bp. The KO region does not have any other known gene. Page 1 of 9 https://www.alphaknockout.com Overview of the Targeting Strategy Wildtype allele 5' gRNA region gRNA region 3' 1 2 3 10 Legends Exon of mouse Hnrnph3 Knockout region Page 2 of 9 https://www.alphaknockout.com Overview of the Dot Plot (up) Window size: 15 bp Forward Reverse Complement Sequence 12 Note: The 2000 bp section upstream of Exon 2 is aligned with itself to determine if there are tandem repeats. Tandem repeats are found in the dot plot matrix. The gRNA site is selected outside of these tandem repeats. Overview of the Dot Plot (down) Window size: 15 bp Forward Reverse Complement Sequence 12 Note: The 534 bp section downstream of Exon 3 is aligned with itself to determine if there are tandem repeats. -

Supplementary Tables S1-S3
Supplementary Table S1: Real time RT-PCR primers COX-2 Forward 5’- CCACTTCAAGGGAGTCTGGA -3’ Reverse 5’- AAGGGCCCTGGTGTAGTAGG -3’ Wnt5a Forward 5’- TGAATAACCCTGTTCAGATGTCA -3’ Reverse 5’- TGTACTGCATGTGGTCCTGA -3’ Spp1 Forward 5'- GACCCATCTCAGAAGCAGAA -3' Reverse 5'- TTCGTCAGATTCATCCGAGT -3' CUGBP2 Forward 5’- ATGCAACAGCTCAACACTGC -3’ Reverse 5’- CAGCGTTGCCAGATTCTGTA -3’ Supplementary Table S2: Genes synergistically regulated by oncogenic Ras and TGF-β AU-rich probe_id Gene Name Gene Symbol element Fold change RasV12 + TGF-β RasV12 TGF-β 1368519_at serine (or cysteine) peptidase inhibitor, clade E, member 1 Serpine1 ARE 42.22 5.53 75.28 1373000_at sushi-repeat-containing protein, X-linked 2 (predicted) Srpx2 19.24 25.59 73.63 1383486_at Transcribed locus --- ARE 5.93 27.94 52.85 1367581_a_at secreted phosphoprotein 1 Spp1 2.46 19.28 49.76 1368359_a_at VGF nerve growth factor inducible Vgf 3.11 4.61 48.10 1392618_at Transcribed locus --- ARE 3.48 24.30 45.76 1398302_at prolactin-like protein F Prlpf ARE 1.39 3.29 45.23 1392264_s_at serine (or cysteine) peptidase inhibitor, clade E, member 1 Serpine1 ARE 24.92 3.67 40.09 1391022_at laminin, beta 3 Lamb3 2.13 3.31 38.15 1384605_at Transcribed locus --- 2.94 14.57 37.91 1367973_at chemokine (C-C motif) ligand 2 Ccl2 ARE 5.47 17.28 37.90 1369249_at progressive ankylosis homolog (mouse) Ank ARE 3.12 8.33 33.58 1398479_at ryanodine receptor 3 Ryr3 ARE 1.42 9.28 29.65 1371194_at tumor necrosis factor alpha induced protein 6 Tnfaip6 ARE 2.95 7.90 29.24 1386344_at Progressive ankylosis homolog (mouse) -

Endocrine System Local Gene Expression
Copyright 2008 By Nathan G. Salomonis ii Acknowledgments Publication Reprints The text in chapter 2 of this dissertation contains a reprint of materials as it appears in: Salomonis N, Hanspers K, Zambon AC, Vranizan K, Lawlor SC, Dahlquist KD, Doniger SW, Stuart J, Conklin BR, Pico AR. GenMAPP 2: new features and resources for pathway analysis. BMC Bioinformatics. 2007 Jun 24;8:218. The co-authors listed in this publication co-wrote the manuscript (AP and KH) and provided critical feedback (see detailed contributions at the end of chapter 2). The text in chapter 3 of this dissertation contains a reprint of materials as it appears in: Salomonis N, Cotte N, Zambon AC, Pollard KS, Vranizan K, Doniger SW, Dolganov G, Conklin BR. Identifying genetic networks underlying myometrial transition to labor. Genome Biol. 2005;6(2):R12. Epub 2005 Jan 28. The co-authors listed in this publication developed the hierarchical clustering method (KP), co-designed the study (NC, AZ, BC), provided statistical guidance (KV), co- contributed to GenMAPP 2.0 (SD) and performed quantitative mRNA analyses (GD). The text of this dissertation contains a reproduction of a figure from: Yeo G, Holste D, Kreiman G, Burge CB. Variation in alternative splicing across human tissues. Genome Biol. 2004;5(10):R74. Epub 2004 Sep 13. The reproduction was taken without permission (chapter 1), figure 1.3. iii Personal Acknowledgments The achievements of this doctoral degree are to a large degree possible due to the contribution, feedback and support of many individuals. To all of you that helped, I am extremely grateful for your support. -
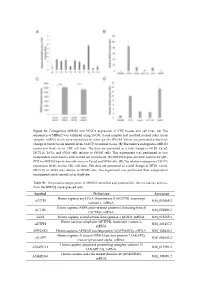
Figure S1. Endogenous MIR45
Figure S1. Endogenous MIR452 and VEGFA expression in CRC tissues and cell lines. (A) The expression of MIR452 was validated using 10 CRC tissue samples and matched normal colon tissue samples. miRNA levels were normalized to colon-specific RNU48. Values are presented as the fold- change in tumor tissue relative levels (ΔΔCT) to normal tissue. (B) The relative endogenous MIR452 expression levels in six CRC cell lines. The data are presented as a fold change in HT29, Caco2, HCT116, LoVo, and SW48 cells relative to SW480 cells. This experiment was performed as two independent experiments, each carried out in triplicate. (C) MIR452 expression level analysis by qRT- PCR for MIR452 transfection efficiency in Caco2 and SW48 cells. (D) The relative endogenous VEGFA expression levels in five CRC cell lines. The data are presented as a fold change in HT29, Caco2, HCT116, or SW48 cells relative to SW480 cells. This experiment was performed three independent experiments, each carried out in duplicate. Table S1. The putative target genes of MIR452 identified and predicted by the microarray analysis from the MIR452 overexpressed cells. Symbol Definition Accession Homo sapiens acyl-CoA thioesterase 8 (ACOT8), transcript ACOT8 NM_005469.2 variant 1, mRNA. Homo sapiens ARP6 actin-related protein 6 homolog (yeast) ACTR6 NM_022496.3 (ACTR6), mRNA. ADI1 Homo sapiens acireductone dioxygenase 1 (ADI1), mRNA. NM_018269.1 Homo sapiens aftiphilin (AFTPH), transcript variant 1, AFTPH NM_203437.2 mRNA. AHNAK2 Homo sapiens AHNAK nucleoprotein 2 (AHNAK2), mRNA. NM_138420.2 Homo sapiens A kinase (PRKA) anchor protein 7 (AKAP7), AKAP7 NM_004842.2 transcript variant alpha, mRNA. Homo sapiens anaphase promoting complex subunit 13 ANAPC13 NM_015391.2 (ANAPC13), mRNA. -

(12) Patent Application Publication (10) Pub. No.: US 2003/0170678A1 Tanzi Et Al
US 2003.017O678A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2003/0170678A1 Tanzi et al. (43) Pub. Date: Sep. 11, 2003 (54) GENETIC MARKERS FOR ALZHEIMER'S Related U.S. Application Data DISEASE AND METHODS USING THE SAME (60) Provisional application No. 60/348,065, filed on Oct. (75) Inventors: Rudolph E. Tanzi, Hull, MA (US); 25, 2001. Provisional application No. 60/336,983, Kenneth David Becker, San Diego, CA filed on Nov. 2, 2001. (US); Gonul Velicelebi, San Diego, CA (US); Kathryn J. Elliott, San Diego, Publication Classification CA (US); Xin Wang, San Diego, CA (US); Lars Bertram, Brighton, MA (51) Int. C.7 - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - C12O 1/68 (US); Aleister J. Saunders, (52) U.S. Cl. .................................................................. 435/6 Philadelphia, PA (US); Deborah Lynne Blacker, Newton, MA (US) (57) ABSTRACT Correspondence Address: HELLER EHRMAN WHITE & MCAULIFFE LLP Genetic markers associated with Alzheimer's disease are 4350 LA JOLLAVILLAGE DRIVE provided. Also provided are methods of determining the 7TH FLOOR presence or absence in a Subject of one or more polymor SAN DIEGO, CA 92122-1246 (US) phisms associated with Alzheimer's disease and methods of determining the level of risk for Alzheimer's disease in a (73) Assignee: Neurogenetics, Inc. Subject. Further provided are nucleic acid compositions and kits for use in determining the presence or absence in a (21) Appl. No.: 10/281,456 Subject of one or more polymorphisms associated with Alzheimer's disease and kits for determining the level of (22) Filed: Oct. 25, 2002 risk for Alzheimer's disease in a Subject. Patent Application Publication Sep. -

Translational Regulation of 5' Terminal Oligopyrimidine Tract (TOP) Mrnas by Insulin
Markou et al. BMC Genomics 2010, 11:343 http://www.biomedcentral.com/1471-2164/11/343 RESEARCH ARTICLE Open Access RegulationResearch article of the cardiomyocyte transcriptome vs translatome by endothelin-1 and insulin: translational regulation of 5' terminal oligopyrimidine tract (TOP) mRNAs by insulin Thomais Markou, Andrew K Marshall, Timothy E Cullingford, El L Tham, Peter H Sugden and Angela Clerk* Abstract Background: Changes in cellular phenotype result from underlying changes in mRNA transcription and translation. Endothelin-1 stimulates cardiomyocyte hypertrophy with associated changes in mRNA/protein expression and an increase in the rate of protein synthesis. Insulin also increases the rate of translation but does not promote overt cardiomyocyte hypertrophy. One mechanism of translational regulation is through 5' terminal oligopyrimidine tracts (TOPs) that, in response to growth stimuli, promote mRNA recruitment to polysomes for increased translation. TOP mRNAs include those encoding ribosomal proteins, but the full panoply remains to be established. Here, we used microarrays to compare the effects of endothelin-1 and insulin on the global transcriptome of neonatal rat cardiomyocytes, and on mRNA recruitment to polysomes (i.e. the translatome). Results: Globally, endothelin-1 and insulin (1 h) promoted >1.5-fold significant (false discovery rate < 0.05) changes in expression of 341 and 38 RNAs, respectively. For these transcripts with this level of change there was little evidence of translational regulation. However, 1336 and 712 RNAs had >1.25-fold significant changes in expression in total and/or polysomal RNA induced by endothelin-1 or insulin, respectively, of which ~35% of endothelin-1-responsive and ~56% of insulin-responsive transcripts were translationally regulated. -

Transcriptomic and Proteomic Analysis of Human Hepatic Stellate Cells Treated with Natural Taurine
1442 MOLECULAR MEDICINE REPORTS 7: 1442-1452, 2013 Transcriptomic and proteomic analysis of human hepatic stellate cells treated with natural taurine JIAN LIANG, XIN DENG, FA‑SHENG WU and YAN‑FANG TANG Ruikang Hospital of Guangxi Traditional Chinese Medical University, Nanning, Guangxi 530011, P.R. China Received September 24, 2012; Accepted February 14, 2013 DOI: 10.3892/mmr.2013.1389 Abstract. The aim of this study was to investigate the Introduction differential expression of genes and proteins between natural taurine (NTau)-treated hepatic stellate cells (HSCs) and Hepatic fibrosis refers to the excessive accumulation of extra- control cells as well as the underlying mechanism of NTau in cellular matrix (ECM) components in the liver. It occurs in inhibiting hepatic fibrosis. A microculture tetrazolium(MTT) most types of chronic liver diseases, including liver cirrhosis, assay was used to analyze the proliferation of NTau‑treated liver failure, and portal hypertension, and often requires liver HSCs. Flow cytometry was performed to compare the transplantation (1). The activation of hepatic stellate cells apoptosis rate between NTau-treated and non-treated HSCs. (HSCs) is an important event by which this cell type, which Proteomic analysis using a combination of 2-dimensional is otherwise quiescent, expresses α-smooth muscle actin gel electrophoresis (2DE) and mass spectrometry (MS) was (α‑SMA), assumes a myofibroblastic phenotype and synthe- conducted to identify the differentially expressed proteins. sizes fibrillar collagens (2,3). Therefore, the inhibition of Microarray analysis was performed to investigate the differ- HSC proliferation, the regulation of the HSC cell cycle, and ential expression of genes and real-time polymerase chain the facilitation of HSC apoptosis are important therapeutic reaction (PCR) was used to validate the results. -
Identification of the Genomic Breakpoints of a Novel Chromosome Translocation in Myelodysplastic Syndrome
IDENTIFICATION OF THE GENOMIC BREAKPOINTS OF A NOVEL CHROMOSOME TRANSLOCATION IN MYELODYSPLASTIC SYNDROME Sarah Sofia Mealhada Cardoso de Matos Dissertation for a Master’s Degree in Oncology Porto, 2011 Sarah Sofia Mealhada Cardoso de Matos IDENTIFICATION OF THE GENOMIC BREAKPOINTS OF A NOVEL CHROMOSOME TRANSLOCATION IN MYELODYSPLASTIC SYNDROME Dissertation for applying to a Master’s Degree in Oncology – Specialization in Molecular Oncology submitted to the Institute of Biomedical Sciences Abel Salazar – University of Porto Supervisor: Manuel António Rodrigues Teixeira, MD, PhD Guest Full Professor at the Department of Pathology and Molecular Immunology of the Institute of Biomedical Sciences Abel Salazar – University of Porto Director of the Genetics Department and Research Centre of the Portuguese Oncology Institute – Porto Co-supervisors: Nuno Cerveira, PhD Superior Health Technician at the Genetics Department of the Portuguese Oncology Institute – Porto Cecília Correia, MSc Superior Health Technician at the Genetics Department of the Portuguese Oncology Institute – Porto “Nothing in life is to be feared, it is only to be understood.” — Marie Curie AKNOWLEDGEMENTS First of all, I would like to express my gratitude to my supervisor, Professor Manuel Teixeira for guiding me through the process of completing this thesis: for his supervision, time and advice from the very early stage of this research. Thank you for the diligence in the reading and revision of this thesis. To Professor Carlos Lopes, as Director of the Oncology Master’s Programme, and to all other lecturers who participated in this course and shared with us their knowledge and experience in Oncology. I also want to thank my co-supervisor Nuno Cerveira for teaching me and spending countless hours of his time with me. -

A Master Autoantigen-Ome Links Alternative Splicing, Female Predilection, and COVID-19 to Autoimmune Diseases
bioRxiv preprint doi: https://doi.org/10.1101/2021.07.30.454526; this version posted August 4, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. A Master Autoantigen-ome Links Alternative Splicing, Female Predilection, and COVID-19 to Autoimmune Diseases Julia Y. Wang1*, Michael W. Roehrl1, Victor B. Roehrl1, and Michael H. Roehrl2* 1 Curandis, New York, USA 2 Department of Pathology, Memorial Sloan Kettering Cancer Center, New York, USA * Correspondence: [email protected] or [email protected] 1 bioRxiv preprint doi: https://doi.org/10.1101/2021.07.30.454526; this version posted August 4, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. Abstract Chronic and debilitating autoimmune sequelae pose a grave concern for the post-COVID-19 pandemic era. Based on our discovery that the glycosaminoglycan dermatan sulfate (DS) displays peculiar affinity to apoptotic cells and autoantigens (autoAgs) and that DS-autoAg complexes cooperatively stimulate autoreactive B1 cell responses, we compiled a database of 751 candidate autoAgs from six human cell types. At least 657 of these have been found to be affected by SARS-CoV-2 infection based on currently available multi-omic COVID data, and at least 400 are confirmed targets of autoantibodies in a wide array of autoimmune diseases and cancer. -

Transcriptome Characterization of Human Mammary Cell Lines Expressing Different Levels of ERBB2 by Serial Analysis of Gene Expression
1441-1461 26/4/06 13:24 Page 1441 INTERNATIONAL JOURNAL OF ONCOLOGY 28: 1441-1461, 2006 Transcriptome characterization of human mammary cell lines expressing different levels of ERBB2 by serial analysis of gene expression MARIANA LOPES DOS SANTOS1, CAROLINA GONÇALVES PALANCH1, SIBELI SALAORNI1, WILSON ARAUJO DA SILVA Jr2 and MARIA APARECIDA NAGAI1 1Disciplina de Oncologia, Departamento de Radiologia da Faculdade de Medicina da Universidade de São Paulo, São Paulo; 2Departamento de Genética, Faculdade de Medicina de Ribeirão Preto-USP, Ribeirão Preto, Brazil Received October 31, 2005; Accepted December 27, 2005 Abstract. Over-expression of ERBB2, a member of the family Introduction of transmembrane receptor tyrosine kinases, occurs in 15-30% of primary breast tumors and is associated with poor prognosis The ERBB2 gene, also known as HER2/NEU, encodes a and chemoresistance to a variety of anticancer drugs. In this 185-kDa transmembrane glycoprotein with intrinsic tyrosine study, aiming to identify differentially-expressed genes kinase activity that belongs to the epidermal growth factor involved in erbB2-mediated transformation of the breast, we receptor (EGFR) family (1). This family of receptor tyrosine generated SAGE libraries from two human mammary cell kinases (RTKs) comprises four members, EGFR (ERBB1), lines, derived from normal luminal cells, expressing different ERBB2 (HER2), ERBB3 and ERBB4 that play important roles levels of erbB2. The parental cell line HB4a expresses basal in several signal transduction pathways regulating cell prolifer- levels and the C5.2 expresses high levels of erbB2. A total of ation, differentiation, cytoskeletal rearrangement and survival 161,632 tags was generated by sequencing, 81,684 from HB4a (1,2).